Abstract
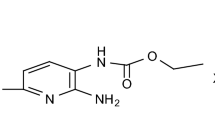
Melting and crystallization of metacetamol (C8H9NO2, N-(3-hydroxyphenyl)acetamide, structural isomer of paracetamol) were measured using DSC cycling heating–cooling between room temperature and 165 °C with constant heating rate of 6 °C min−1 and variable cooling rate ranging from 3 to 24 °C min−1. The selection of the cooling rate allows us to control the crystallization of metacetamol into one of its two polymorphs, I or II. Pure form II of metacetamol (recently discovered) is crystallized after the cooling rate of 6 °C min−1. Increase or decrease in the cooling rate suppresses the crystallization of the form II and produces the form I with remnant amorphous phase and a small impurity of the form II. The melting points and enthalpies of fusion are 420 K and 26.0 ± 1.3 kJ mol−1 for the form I and 399 K and 21.3 ± 1.1 kJ mol−1 for the form II, respectively.







Similar content being viewed by others
References
Haisa M, Kashino S, Kawai R, Maeda H. The monoclinic form of p-hydroxyacetanilide. Acta Cryst. 1976;B32:1283–5.
Haisa M, Kashino S, Maeda H. The orthorhombic form of p-hydroxyacetanilide. Acta Cryst. 1974;B30:2510–2.
Perrin M-A, Neumann MA, Elmaleh H, Zaske L. Crystal structure determination of the elusive paracetamol form III. Chem Commun. 2009;3181–3.
Smith SJ, Bishop MM, Montgomery JM, Hamilton TP, Vohra YK. Polymorphism in paracetamol: evidence of additional forms IV and V at high pressure. J Phys Chem A. 2014;118:6068–77.
McGregor L, Rychkov DA, Coster PL, Day S, Drebushchak VA, Achkasov AF, Nichol GS, Pulham CR, Boldyreva EV. A new polymorph of metacetamol. CrystEngComm. 2015;17:6183–92.
Burger A, Ramberger R. On the polymorphism of pharmaceuticals and other molecular crystals. II. Applicability of thermodynamic rules. Mikrochim Acta. 1979;72:273–316.
Di Martino P, Conflant P, Drache M, Huvenne J-P, Guyot-Hermann A-M. Preparation and physical characterization of forms II and III of paracetamol. J Therm Anal. 1997;48:447–58.
Di Martino P, Palmieri GF, Martelli S. Molecular mobility of the paracetamol amorphous form. Chem Pharm Bull. 2000;48:1105–8.
Politov AA, Kostrovskii VG, Boldyrev VV. Conditions of preparation and crystallization of amorphous paracetamol. Russ J Phys Chem. 2001;75:1903–11.
Boldyreva EV, Drebushchak VA, Paukov IE, Kovalevskaya YA, Drebushchak TN. DSC and adiabatic calorimetry study of the polymorphs of paracetamol. J Therm Anal Calorim. 2004;77:607–23.
Espeau Ph, Céolin R, Tamarit J-L, Perrin M-A, Gauchi J-P, Leveiller F. Polymorphism of paracetamol: relative stabilities of the monoclinic and orthorhombic phases inferred from topological pressure-temperature and temperature-volume phase diagrams. J Pharm Sci. 2005;94:524–39.
Drebushchak VA. Calibration coefficient of a heat-flow DSC; Part II. Optimal calibration procedure. J Therm Anal Calorim. 2005;79:213–8.
Boldyreva EV, Arkhipov SG, Drebushchak TN, Drebushchak VA, Losev EA, Matvienko AA, Minkov VS, Rychkov DA, Seryotkin YV, Stare J, Zakharov BA. Isoenergetic polymorphism: the puzzle of tolazamide as a case study. Chem Eur J. 2015;21:15395–404.
Avrami M. Kinetics of phase change. II. Transformation-time relations for random distribution of nuclei. J Chem Phys. 1940;8:212–24.
Cahn JW, Hillig WB, Sears GW. The molecular mechanism of solidification. Acta Metall. 1964;12:1421–39.
Jackson KA, Uhlmann DR, Hunt JD. On the nature of crystal growth from the melt. J Cryst Growth. 1967;1:1–36.
Kidyarov BI, Bolkhovityanov YuB, Demyanov EA. Statistical investigation of the kinetics of crystal nucleation in melt. Zhurnal Phys Khim. 1970;44:668–72 (in Russian).
Langer JS. Instabilities and pattern formation in crystal growth. Rev Mod Phys. 1980;52:1–28.
Andronis V, Zografi G. Crystal nucleation and growth of indomethacin polymorphs from the amorphous state. J Non Cryst Solids. 2000;271:236–48.
Vali G. Repeatability and randomness in heterogeneous freezing nucleation. Atmos Chem Phys. 2008;8:5017–31.
Bach A, Fischer D, Jansen M. Metastable phase formation of indium monochloride from an amorphous feedstock. Z Anorg Allg Chem. 2013;639:465–7.
Pishchur DP, Drebushchak VA. Recommendations on DSC calibration. How to escape the transformation of a random error into the systematic error. J Therm Anal Calorim. 2016;124:951–8.
Squires GL. Practical physics. 4th ed. Cambridge: Cambridge University Press; 2001.
Acknowledgements
The experiments were supported by a Year Abroad Program of the School of Chemistry, University of Edinburgh (LMcG), Russian Ministry of Education and Science, project 1828 and a project No И-37 of the Siberian Branch of Russian Academy of Sciences (DR). Denis Rychkov and Lindsay McGregor thank professors Elena Boldyreva and Colin R. Pulham for supervision. VAD acknowledges that his work was supported by state Assignment Project No. 0330-2016-004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drebushchak, V.A., McGregor, L. & Rychkov, D.A. Cooling rate “window” in the crystallization of metacetamol form II. J Therm Anal Calorim 127, 1807–1814 (2017). https://doi.org/10.1007/s10973-016-5954-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-5954-0