Abstract
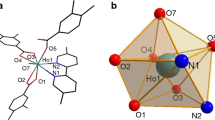
Rare earth (RE) alkoxides of Y, La, Pr, and Gd were synthesized using three tertiary alcohols namely 2,3-dimethyl-2-butanol, 2,2-dimethyl-3-ethyl-3-pentanol, and 3-isopropyl-2,4-dimethyl-pentan-3-ol. 1H and 13C NMR, elemental analysis (CHN) and MS were used to characterize the complexes. New crystal structures of dimeric [Y(OCEt2 tBu)3]2, [La(OCEt2 tBu)3]2, [Gd(OCiPr3)3]2, and [La(OCiPr3)3]2 were solved. Thermal properties of these compounds were studied with TG/SDTA and vacuum sublimation experiments. With all the cations Y, La, Pr, and Gd, the compounds with smallest ligand iPrMe2CO− seem to be the most volatile. The compounds with larger alkoxo ligands have lower volatility but their thermal stability is better. Atomic layer deposition experiments showed that RE oxide thin films could be grown from these alkoxides using water as the oxygen precursor.











Similar content being viewed by others
References
Leskelä M, Ritala M. Rare-earth oxide thin films as gate oxides in MOSFET transistors. J Solid State Chem. 2003;171:170.
Leskelä M, Kukli K, Ritala M. Rare-earth oxide thin films for gate dielectrics in microelectronics. J Alloys Comp. 2006;418:27.
Leskelä M, Ritala M. Atomic layer deposition chemistry: recent developments and future challenges. Angew Chem Int Ed. 2003;42:5548.
Päiväsaari J, Niinistö J, Myllymäki P, Dezelah IV C, Winter CH, Putkonen M, Nieminen M, Niinistö L. Atomic layer deposition of rare earth oxides. In: Fanciulli M, Scarel G, editors. Topics in applied physics, vol. 106. Berlin, Heidelberg: Springer-Verlag; 2007. p. 15–32.
Aspinall HC. Requirements of precursors for MOCVD and ALD of rare earth oxides. In: Fanciulli M, Scarel G, editors. Topics in applied physics, vol. 106. Berlin, Heidelberg: Springer-Verlag; 2007. p. 33–72.
Niinistö J, Petrova N, Putkonen M, Niinistö L, Arstila K, Sajavaara T. Gadolinium oxide thin films by atomic layer deposition. J Cryst Growth. 2005;285:191.
Nieminen M, Putkonen M, Niinistö L. Formation and stability of lanthanum oxide thin films deposited from beta-diketonate precursor. Appl Surf Sci. 2001;174:155.
Potter RJ, Chalker PR, Manning TD, Aspinall HC, Loo YF, Jones AC, Smith LM, Critchlow GW, Schumacher M. Deposition of HfO2, Gd2O3and PrOx by liquid injection ALD techniques. Chem Vap Deposition. 2005;11:159.
Lim BS, Rahtu A, Gordon RG. Atomic layer depostion of transition metals. Nat Mater. 2003;2:729.
Lim BS, Rahtu A, deRouffignac P, Gordon RG. Atomic layer deposition of lanthanum aluminium oxide nano-laminates for electrical applications. Appl Phys Lett. 2004;84:3957.
Päiväsaari J, Dezelah CL, Back D, El-Kadri HM, Heeg MJ, Putkonen M, Niinistö L, Winter CH. Synthesis, structure, and properties of volatile lanthanide complexes containing amidinate ligands: application for Er2O3 thin film growth byatomic layer deposition. J Mater Chem. 2005;15:4224.
Putkonen M, Niinitö L. Organometallic precursors for atomic layer deposition in CVD precursors. Top Organomet Chem. 2005;9:125.
Bradley DC, Mehotra RC, Gaur DP. Metal alkoxides. London: Academic Press; 1978.
Poncelet O, Sartain WJ, Hubert-Pfalzgraf LG, Folting K, Caulton KG. Chemistry of yttrium triisopropoxide revisited—characterization and crystal-structure of Y5(μ5-O)(μ3-OiPr)4(μ2-OiPr)4(OiPr)5. Inorg Chem. 1989;28:263.
Bradley DC, Chudzynska H, Frigo DM, Hammond ME, Hursthouse MB, Mazid MA. Pentanuclear oxoalkoxide clusters of scandium, yttrium, indium and ytterbium, x-ray crystal-structures of [M5(μ5-O)(μ3-OPri)4 (μ2-OPri)4(OPri)5] (M = In, Yb). Polyhedron. 1990;9:719.
Helgesson G, Jagner S, Poncelet O, Hubert-Pfalzgraf LG. Synthesis and molecular-structure of Nd5(μ5-O)(μ3-OR)2(μ2-OR)6(OR)5(ROH)2 (R = Pri)—the 1st example of a trigonal bipyramidal metal oxoalkoxide. Polyhedron. 1991;10:1559.
Bradley DC, Chudzynska H, Hursthouse MB, Motevalli M. Volatile tris-tertiary-alkoxides of yttrium and lanthanum—the x-ray crystal-structure of [La3(OBu-tert)9(Bu-tertOH)2]. Polyhedron. 1991;10:1049.
Anwander R. Routes to monomeric lanthanide alkoxides. In: Herrmann WA, editor. Topics in current chemistry, vol. 173. Berlin: Springer; 1996. p. 149.
Kukli K, Hatanpää T, Ritala M, Leskelä M. Atomic layer deposition of gadolinium oxide films. Chem Vap Deposition. 2007;13:546.
Buhler JD. Reaction of lithium alkyls with aldehydes and ketones—general study. J Org Chem. 1973;38:904.
Imamoto T, Takiyama N, Nakamura K, Hatajima T, Kamiya Y. Reactions of carbonyl-compounds with Grignard-reagents in the presence of cerium chloride. J Am Chem Soc. 1989;111:4392.
Bradley DC, Ghotra JS, Hart FA. Low coordination numbers in lanthanide and actinide compounds. 1. Preparation and characterization of tris[bis(trimethylsilyl)-amido]lanthanides. J Chem Soc Dalton Trans 1973:1021.
Kottke T, Stalke D. Crystal handling at low-temperatures. J Appl Crystallogr. 1993;26:615.
Sheldrick GM. SADABS, a software for empirical absorption correction. Gottingen, Germany: University of Gottingen; 2000.
Sheldrick GM. A short history of SHELX. Acta Crystallogr Sect A. 2008;64:112.
SHELXTL Reference Manual, Version 5.1; Bruker AXS, Analytical Instrumentation, Madison, Wisconsin, USA; 2000.
Suntola T. Atomic layer epitaxy. Thin Solid Films. 1992;216:84.
Ylilammi M, Ranta-aho T. Optical determination of the film thicknesses in multilayer thin-film structures. Thin Solid Films. 1993;232:56.
Shannon RD. Revised effective ionic-radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr Sect A. 1976;32:751.
Boyle TJ, Ottley LAM. Advances in structurally characterized lanthanide alkoxide, aryloxide, and silyloxide compounds. Chem Rev. 2008;108:1896.
Stecher HA, Sen A, Rheingold AL. Synthesis, structure, and reactivity of cerium(III) alkoxides. 2. Thermal-decomposition of Ce(OCtBu3)3 and the structure of [Ce(OCHtBu2)3]2. Inorg Chem. 1989;28:3280.
Evans WJ, Golden RE, Ziller JW. A comparative synthetic and structural study of triphenylmethoxide and triphenylsiloxide complexes of the early lanthanides, including X-ray crystal-structures of La2(OCPh3)6 and Ce2(OSiPh3)6. Inorg Chem. 1991;30:4963.
Sen A, Stecher HA, Rheingold AL. Synthesis, structure, and reactivity of homoleptic cerium(IV) and cerium(III) alkoxides. Inorg Chem. 1992;31:473.
Gromada J, Morteux A, Chenal T, Ziller JW, Leising F, Carpentier J-F. Neodymium alkoxides: synthesis, characterization and their combinations with dialkylmagnesiums as unique systems for polymerization and block copolymerization of ethylene and methyl methacrylate. Chem Eur J. 2002;8:3773.
Anwander R, Munch FC, Priemeier T, Scherer W, Runte O, Hermann WA. Volatile donor-functionalized alkoxy derivatives of lutetium and their structural characterization. Inorg Chem. 1997;36:3545.
Allen FH. The Cambridge Structural Database: a quarter of a million crystal structures and rising. Acta Crystallogr B. 2002;58:380–8.
Dietrich HM, Meermann C, Tornroos KW, Anwander R. Sounding out the reactivity of trimethylyttrium. Organometallics. 2006;25:4316.
Aspinall HC, Bickley JF, Gaskell JM, Jones AC, Labat G, Chalker PR, Williams PA. Precursors for MOCVD and ALD of rare earth oxides-complexes of the early lanthanides with a donor-functionalized alkoxide ligand. Inorg Chem. 2007;46:5852.
Kritikos M, Moustiakimov M, Wijk M, Westin G. Synthesis, structure and characterisation of Ln5O(OPri)13 with Ln = Nd, Gd or Er. J Chem Soc Dalton Trans 2001:1931.
Daniel SD, Lehn J-SM, van der Heide P, Wang Y, Hoffman DM. Synthesis of cyclopentadienyl gadolinium and samarium alkoxide complexes. Inorg Chim Acta. 2006;359:257.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hatanpää, T., Kukli, K., Ritala, M. et al. Crystal structures and thermal properties of some rare earth alkoxides with tertiary alcohols. J Therm Anal Calorim 105, 61–71 (2011). https://doi.org/10.1007/s10973-011-1546-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-011-1546-1