Abstract
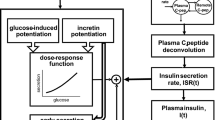
A proof of concept study was conducted to investigate the safety and tolerability of a novel oral glucokinase activator, LY2599506, during multiple dose administration to healthy volunteers and subjects with Type 2 diabetes mellitus (T2DM). To analyze the study data, a previously established semi-mechanistic integrated glucose-insulin model [1–5] was extended to include characterization of glucagon dynamics. The model captured endogenous glucose and insulin dynamics, including the amplifying effects of glucose on insulin production and of insulin on glucose elimination, as well as the inhibitory influence of glucose and insulin on hepatic glucose production. The hepatic glucose production in the model was increased by glucagon and glucagon production was inhibited by elevated glucose concentrations. The contribution of exogenous factors to glycemic response, such as ingestion of carbohydrates in meals, was also included in the model. The effect of LY2599506 on glucose homeostasis in subjects with T2DM was investigated by linking a one-compartment, pharmacokinetic model to the semi-mechanistic, integrated glucose-insulin-glucagon system. Drug effects were included on pancreatic insulin secretion and hepatic glucose production. The relationships between LY2599506, glucose, insulin, and glucagon concentrations were described quantitatively and consequently, the improved understanding of the drug-response system could be used to support further clinical study planning during drug development, such as dose selection.








Similar content being viewed by others
References
Jauslin PM, Silber HE, Frey N, Gieschke R, Simonsson US, Jorga K, Karlsson MO (2007) An integrated glucose-insulin model to describe oral glucose tolerance test data in type 2 diabetics. J Clin Pharmacol 47(10):1244–1255
Silber HE, Jauslin PM, Frey N, Gieschke R, Simonsson US, Karlsson MO (2007) An integrated model for glucose and insulin regulation in healthy volunteers and type 2 diabetic patients following intravenous glucose provocations. J Clin Pharmacol 47(9):1159–1171
Silber HE, Jauslin PM, Frey N, Karlsson MO (2010) An integrated model for the glucose-insulin system. Basic Clin Pharmacol Toxicol 106(3):189–194
Jauslin PM, Frey N, Karlsson MO (2011) Modeling of 24-hour glucose and insulin profiles of patients with type 2 diabetes. J Clin Pharmacol 51(2):153–164
Jauslin PM, Karlsson MO, Frey N (2011) Identification of the mechanism of action of a glucokinase activator from oral glucose tolerance test data in type 2 diabetic patients based on an integrated glucose-insulin model. J Clin Pharmacol 52(12):1861–1871
de Winter W, DeJongh J, Post T, Ploeger B, Urquhart R, Moules I, Eckland D, Danhof M (2006) A mechanism-based disease progression model for comparison of long-term effects of pioglitazone, metformin and gliclazide on disease processes underlying Type 2 Diabetes Mellitus. J Pharmacokinet Pharmacodyn 33(3):313–343
Landersdorfer CB, Jusko WJ (2008) Pharmacokinetic/pharmacodynamic modelling in diabetes mellitus. Clin Pharmacokinet 47(7):417–448
Gao W, Jusko WJ (2012) Target-mediated pharmacokinetic and pharmacodynamic model of exendin-4 in rats, monkeys and man. Drug Metab Dispos 40(5):990–997
Landersdorfer CB, He YL, Jusko WJ (2012) Mechanism-based population modelling of the effects of vildagliptin on GLP-1, glucose and insulin in patients with type 2 diabetes. Br J Clin Pharmacol 73(3):373–390
Dahl SG, Aarons L, Gundert-Remy U, Karlsson MO, Schneider YJ, Steimer JL, Troconiz IF (2010) Incorporating physiological and biochemical mechanisms into pharmacokinetic-pharmacodynamic models: a conceptual framework. Basic Clin Pharmacol Toxicol 106(1):2–12
Matschinsky FM (2009) Assessing the potential of glucokinase activators in diabetes therapy. Nat Rev Drug Discov 8(5):399–416
Zhang L, Beal SL, Sheiner LB (2003) Simultaneous vs. sequential analysis for population PK/PD data I: best-case performance. J Pharmacokinet Pharmacodyn 30(6):387–404
Zhang L, Beal SL, Sheinerz LB (2003) Simultaneous vs. sequential analysis for population PK/PD data II: robustness of methods. J Pharmacokinet Pharmacodyn 30(6):405–416
Sharma A, Jusko WJ (1998) Characteristics of indirect pharmacodynamic models and applications to clinical drug responses. Br J Clin Pharmacol 45(3):229–239
Bauer R (2011) NONMEM User’s guide, introduction to NONMEM 7.2.0. ICON Development solutions, Ellicott City, MD
Yano Y, Beal SL, Sheiner LB (2001) Evaluating pharmacokinetic/pharmacodynamic models using the posterior predictive check. J Pharmacokinet Pharmacodyn 28(2):171–192
Cherrington AD (1999) Banting Lecture, 1997.Control of glucose uptake and release by the liver in vivo. Diabetes 48(5):1198–1214
Ramnanan CJ, Edgerton DS, Kraft G, Cherrington AD (2011) Physiologic action of glucagon on liver glucose metabolism. Diabetes Obes Metab 13(Suppl 1):118–125
Meyer C, Stumvoll M, Nadkarni V, Dostou J, Mitrakou A, Gerich J (1998) Abnormal renal and hepatic glucose metabolism in type 2 diabetes mellitus. J Clin Invest 102(3):619–624
Meyer C, Woerle HJ, Dostou JM, Welle SL, Gerich JE (2004) Abnormal renal, hepatic, and muscle glucose metabolism following glucose ingestion in type 2 diabetes. Am J Physiol Endocrinol Metab 287(6):E1049–E1056
Bue-Valleskey JM, Schneck KB, Sinha VP, Wondmagegnehu ET, Kapitza C, Miller JW (2011) LY2599506, a novel glucokinase activator (GKA), improves fasting and postprandial glucose in patients wtih type 2 diabetes mellitus (T2DM) [abstract]. Diabetes 60(Suppl 1):A272
Bue-Valleskey JM, Schneck KB, Sinha VP, Wondmagegnehu ET, Kapitza C, Miller JW (2011) LY2599506, a novel glucokinase activator (GKA), improves fasting and postprandial glucose in patients wtih Type 2 diabetes mellitus. In: 47th EASD Annual Meeting, Lisbon, 12–16 September 2011. European Association for the study of diabetes
Walker JN, Ramracheya R, Zhang Q, Johnson PR, Braun M, Rorsman P (2011) Regulation of glucagon secretion by glucose: paracrine, intrinsic or both? Diabetes Obes Metab 13(Suppl 1):95–105
Postic C, Shiota M, Magnuson MA (2001) Cell-specific roles of glucokinase in glucose homeostasis. Recent Prog Horm Res 56:195–217
Postic C, Shiota M, Niswender KD, Jetton TL, Chen Y, Moates JM, Shelton KD, Lindner J, Cherrington AD, Magnuson MA (1999) Dual roles for glucokinase in glucose homeostasis as determined by liver and pancreatic beta cell-specific gene knock-outs using Cre recombinase. J Biol Chem 274(1):305–315
Bauer R (2008) S-ADAPT/MCPEM User’s Guide (version 1.56). Berkeley, CA
Hamren B, Bjork E, Sunzel M, Karlsson M (2008) Models for plasma glucose, HbA1c, and hemoglobin interrelationships in patients with type 2 diabetes following tesaglitazar treatment. Clin Pharmacol Ther 84(2):228–235
Acknowledgments
The authors would like to thank Mr. Michael Heathman for assistance with the implementation of models and simulations and Dr. Brian Topp for discussions on physiological aspects of glucagon. Dr. Jeffrey Miller was a significant contributor to the design of the study.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Glucose submodel equations
GA is glucose in the absorption compartment, ka is first-order absorption, GC and GP are the glucose in the central and peripheral compartments, GPROD is endogenous glucose production, Q is intercompartmental clearance, VG and VP are the central and peripheral volumes of distribution, CLGI is insulin-dependent clearance and CLG is insulin-independent clearance, GSS is glucose in the central compartment at steady-state, GE is the glucose effect compartment, kGE is the equilibration rate constant to and from the glucose effect compartment, IE is the insulin effect compartment, ISS is insulin concentration at steady-state.
Insulin submodel equations
ISEC is endogenous insulin secretion, CLI is insulin clearance, VI is the volume of distribution of insulin, ISEC,0 is baseline insulin secretion, GCM is the fraction of insulin secretion regulated by the glucose concentration in the effect compartment, IPRG is the estimated power in the relationship, IABSG is the fraction of insulin secretion triggered by the incretin effect following glucose absorption from the gut, Sincr is the slope of a linear relationship, kIE is the equilibration rate constant to and from the insulin effect compartment.
Rights and permissions
About this article
Cite this article
Schneck, K.B., Zhang, X., Bauer, R. et al. Assessment of glycemic response to an oral glucokinase activator in a proof of concept study: application of a semi-mechanistic, integrated glucose-insulin-glucagon model. J Pharmacokinet Pharmacodyn 40, 67–80 (2013). https://doi.org/10.1007/s10928-012-9287-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-012-9287-8