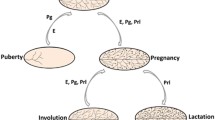
Abstract
The presence of long-lived lineage restricted progenitor and multipotent progenitor cells in adult mouse mammary gland for cancer development is compelling. Mammary cancers are phenotypically diverse This might be explained by transformation of long-lived, lineage-limited progenitor subpopulations. Mammary multipotent epithelial stem cells and their environmental niches must be considered, since their niche(s), once empty might be occupied by lineage-limited progenitors that are proximal. The existence of premalignant mammary populationst that manifest characteristics of lineage limitation argues strongly for this proposition.
Similar content being viewed by others
References
Young LJT, et al. The influence of host and tissue age on life span and growth rate of serially transplanted mouse mammary gland. Exp Gerontol. 1971;6(1):49–56.
Raafat A, et al. Effects of Age and Parity on Mammary Gland Lesions and Progenitor Cells in the FVB/N-RC Mice. PLoS One. 2012;7(8):e43624.
Cereser B, et al. Analysis of clonal expansions through the normal and premalignant human breast epithelium reveals the presence of luminal stem cells. J Pathol. 2018;244:61–70.
Wuidart A, et al. Quantitative lineage tracing strategies to resolve multipotency in tissue-specific stem cells. Genes Dev. 2016;30(11):1261–77.
Shackleton M, et al. Generation of a functional mammary gland from a single stem cell. Nature. 2006;439(7072):84–8.
Stingl J, et al. Purification and unique properties of mammary epithelial stem cells. Nature. 2006;439(7079):993–7.
Kordon EC, Smith GH. An entire functional mammary gland may comprise the progeny from a single cell. Development. 1998;125(10):1921–30.
Van Keymeulen A, et al. Distinct stem cells contribute to mammary gland development and maintenance. Nature. 2011;479(7372):189–U58.
Wang CH, et al. Lineage-Biased Stem Cells Maintain Estrogen-Receptor-Positive and -Negative Mouse Mammary Luminal Lineages. Cell Rep. 2017;18(12):2825–35.
Boulanger CA, Wagner KU, Smith GH. Parity-induced mouse mammary epithelial cells are pluripotent, self-renewing and sensitive to TGF-beta1 expression. Oncogene. 2005;24(4):552–60.
Kaanta AS, et al. Evidence for a multipotent mammary progenitor with pregnancy-specific activity. Breast Cancer Res. 2013;15(4):R65.
Booth BW, Boulanger CA, Smith GH. Alveolar progenitor cells develop in mouse mammary glands independent of pregnancy and lactation. J Cell Physiol. 2007;212(3):729–36.
Smith GH. Experimental mammary epithelial morphogenesis in an in vivo model: evidence for distinct cellular progenitors of the ductal and lobular phenotype. Breast Cancer Res Treat. 1996;39(1):21–31.
Welm BE, et al. Lentiviral transduction of mammary stem cells for analysis of gene function during development and cancer. Cell Stem Cell. 2008;2(1):90–102.
Mintz B, Slemmer G. Gene control of neoplasia i genotypic mosaicism in normal and preneoplastic mammary glands of allophenic mice. J Natl Cancer Inst. 1969;348(11):87–95.
Smith GH, Boulanger CA. Mammary stem cell repertoire: new insights in aging epithelial populations. Mech Ageing Dev. 2002;123(11):1505–19.
Callahan R, Smith GH. Common integration sites for MMTV in viral induced mouse mammary tumors. J Mammary Gland Biol Neoplasia. 2008;13(3):309–21.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, G.H., Medina, D. Does the Mouse Mammary Gland Arise from Unipotent or Multipotent Mammary Stem/Progenitor Cells?. J Mammary Gland Biol Neoplasia 23, 1–3 (2018). https://doi.org/10.1007/s10911-018-9394-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10911-018-9394-2