Abstract
Purpose
Non-pulmonary vein (non-PV) triggers and left atrial (LA) scars perpetuate atrial fibrillation (AF) and limit the success rate of catheter ablation. In order to understand the genetic basis of these risk factors, we examined the association of selected single nucleotide polymorphisms (SNPs) with scar and non-PV triggers.
Methods
Four hundred AF patients (67 % male, 62 ± 12 years, LA size 45.3 ± 7 mm, 64 % non-paroxysmal) undergoing catheter ablation were prospectively enrolled. DNA extractions for 16 AF-related SNPS from blood samples were performed of which 371 DNA samples were available for genotyping. Multivariate logistic regression analysis was used for assessing predictive role of individual SNP, and logistic kernel-machine approach was applied to test the cumulative effect of multiple SNPs as a group with non-PV triggers and LA scar. False discovery rate (FDR) was computed for all candidate SNPs to address multiple testing.
Results
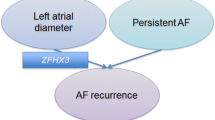
SNPs rs6599230 and rs6843082 were inversely associated (OR 0.68, p = 0.04, and 0.62, p = 0.01, respectively) whereas rs1448817 (OR 1.74, p = 0.04) and rs7193343 (OR 1.66, p = 0.02) predicted higher risk of non-PV triggers. Genotypes for rs6599230 and rs6843082 conferred 51 % reduction in the odds for non-PV triggers (combined OR 0.49, p = 0.019), while rs1448817 and rs7193343 demonstrated a combined OR of 1.93, p = 0.025. FDR was controlled at 16 % to adjust for multiple testing. For LA scar, inverse association was observed with rs1448817 (OR 0.29, p = 0.006), rs17042171 (OR 0.27, p = 0.032), rs3807989 (OR 0.54, p = 0.017), and rs6843082 (OR 0.56, p = 0.009). Two SNPs were associated with increased scar risk: rs17375901 (OR 3.68, p = 0.03) and rs7193343 (OR 1.74, p = 0.037). For global association of SNPs with left atrial scar FDR was controlled at ≤10 % to adjust for multiple testing.
Conclusions
This study has a strong clinical significance as it provides important insights into the molecular basis of pertinent therapeutic targets. Our findings demonstrate that the presence of certain genetic polymorphisms increases the risk of scar and non-PV triggers in AF patients. Therefore, PVAI alone will not be enough to eliminate the arrhythmia and the operators may need to identify and isolate the non-PV foci to maximize procedural success in patients carrying these risk variants.
Clinical trial registration
clinicaltrials.gov (NCT01751607)


Similar content being viewed by others
References
Colilla, S., Crow, A., Petkun, W., Singer, D. E., Simon, T., & Liu, X. (2013). Estimates of current and future incidence and prevalence of atrial fibrillation in the U.S. Adult population. American Journal of Cardiology, 112(8), 1142–1147.
Jaïs, P., Cauchemez, B., Macle, L., et al. (2008). Catheter ablation versus antiarrhythmic drugs for atrial fibrillation: the A4 study. Circulation, 118(24), 2498–2505.
Mont, L., Bisbal, F., Hernández-Madrid, A., et al. (2014). SARA investigators. Catheter ablation vs. antiarrhythmic drug treatment of persistent atrial fibrillation: a multicentre, randomized, controlled trial (SARA study). European Heart Journal, 35(8), 501–507.
Bhargava, M., Di Biase, L., Mohanty, P., et al. (2009). Impact of type of atrial fibrillation and repeat catheter ablation on long-term freedom from atrial fibrillation: results from a multicenter study. Heart Rhythm, 6(10), 1403–1412.
Di Biase, L., Burkhardt, J. D., Mohanty, P., et al. (2010). Left atrial appendage: an underrecognized trigger site of atrial fibrillation. Circulation, 122(2), 109–118.
Mohanty, S., Mohanty, P., Di Biase, L., et al. (2012). Impact of metabolic syndrome on procedural outcomes in patients with atrial fibrillation undergoing catheter ablation. Journal of the American College of Cardiology, 59(14), 1295–1301.
Verma, A., Wazni, O. M., Marrouche, N. F., et al. (2005). Pre-existent left atrial scarring in patients undergoing pulmonary vein antrum isolation: an independent predictor of procedural failure. Journal of the American College of Cardiology, 45(2), 285–292.
Tucker, N. R., & Ellinor, P. T. (2014). Emerging directions in the genetics of atrial fibrillation. Circulation Research, 114(9), 1469–1482.
Everett, B. M., Cook, N. R., Conen, D., Chasman, D. I., Ridker, P. M., & Albert, C. M. (2013). Novel genetic markers improve measures of atrial fibrillation risk prediction. European Heart Journal, 34(29), 2243–2251.
Melander, O., Shiffman, D., Smith, J. G., Sjögren, M., Louie, J., Catanese, J. J., Engström, G., Devlin, J., & Kathiresan, S. (2014). A multi-locus genetic risk score is associated with incident atrial fibrillation and ischemic stroke among 27,471 participants from a prospective cohort study. Journal of the American College of Cardiology, 63(12_S) (ABSTRACT).
Di Biase, L., Burkhardt, J. D., Santangeli, P., et al. (2014). Periprocedural stroke and bleeding complications in patients undergoing catheter ablation of atrial fibrillation with different anticoagulation management: results from the Role of Coumadin in Preventing Thromboembolism in Atrial Fibrillation (AF) Patients Undergoing Catheter Ablation (COMPARE) randomized trial. Circulation, 129(25), 2638–2644.
Valles, E., Fan, R., Roux, J. F., Liu, C. F., Harding, J. D., Dhruvakumar, S., et al. (2008). Localization of atrial fibrillation triggers in patients undergoing pulmonary vein isolation: importance of the carina region. Journal of the American College of Cardiology, 52(17), 1413–1420.
Di Biase, L., Santangeli, P., Burkhardt, J. D., & Natale, A. (2013). Which is the best approach for ablation of long-standing persistent atrial fibrillation? Pacing and Clinical Electrophysiology, 36(10), 1198–1201.
Corrado, A., Bonso, A., Madalosso, M., Rossillo, A., Themistoclakis, S., Di Biase, L., Natale, A., & Raviele, A. (2010). Impact of systematic isolation of superior vena cava in addition to pulmonary vein antrum isolation on the outcome of paroxysmal, persistent, and permanent atrial fibrillation ablation: results from a randomized study. Journal of Cardiovascular Electrophysiology, 21(1), 1–5.
Wilber, D. J. (2012). Fibroblasts, focal triggers, and persistent atrial fibrillation: is there a connection? Circulation. Arrhythmia and Electrophysiology, 5(2), 249–251.
Rohr, S. (2012). Arrhythmogenic implications of fibroblast-myocyte interactions. Circulation. Arrhythmia and Electrophysiology, 5(2), 442–452.
Manabe, I., Shindo, T., & Nagai, R. (2002). Gene expression in fibroblasts and fibrosis: involvement in cardiac hypertrophy. Circulation Research, 91, 1103–1113.
Sellers, T. A., Huang, Y., Cunningham, J., et al. (2008). Association of single nucleotide polymorphisms in glycosylation genes with risk of epithelial ovarian cancer. Cancer Epidemiology, Biomarkers and Prevention, 17(2), 397–404.
Gudbjartsson, D. F., Holm, H., Gretarsdottir, S., et al. (2009). A sequence variant in ZFHX3 on 16q22 associates with atrial fibrillation and ischemic stroke. Nature Genetics, 41(8), 876–878.
Liu, Y., Ni, B., Lin, Y., Chen, X. G., Fang, Z., Zhao, L., Hu, Z., & Zhang, F. (2014). Genetic polymorphisms in ZFHX3 Are associated with atrial fibrillation in a Chinese Han population. PloS One, 9(7), e101318.
Benjamin, E. J., Rice, K. M., Arking, D. E., et al. (2009). Variants in ZFHX3 are associated with atrial fibrillation in individuals of European ancestry. Nature Genetics, 41(8), 879–881.
Nojiri, S., Joh, T., Miura, Y., Sakata, N., Nomura, T., Nakao, H., Sobue, S., Ohara, H., Asai, K., & Ito, M. (2004). ATBF1 enhances the suppression of STAT3 signaling by interaction with PIAS3. Biochemical and Biophysical Research Communications, 314(1), 97–103.
Tsai, C. T., Lin, J. L., Lai, L. P., Lin, C. S., & Huang, S. K. (2008). Membrane translocation of small GTPase Rac1 and activation of STAT1 and STAT3 in pacing-induced sustained atrial fibrillation. Heart Rhythm, 5(9), 1285–1293.
Li, P., Wang, D., Lucas, J., Oparil, S., Xing, D., Cao, X., Novak, L., Renfrow, M. B., & Chen, Y. F. (2008). Atrial natriuretic peptide inhibits transforming growth factor beta-induced Smad signaling and myofibroblast transformation in mouse cardiac fibroblasts. Circulation Research, 102(2), 185–192.
Delaney, J. T., Jeff, J. M., Brown, N. J., Pretorius, M., Okafor, H. E., Darbar, D., Roden, D. M., & Crawford, D. C. (2012). Characterization of genome-wide association-identified variants for atrial fibrillation in African Americans. PloS One, 7(2), e32338.
Ellinor, P. T., Lunetta, K. L., Glazer, N. L., Pfeufer, A., et al. (2010). Common variants in KCNN3 are associated with lone atrial fibrillation. Nature Genetics, 42(3), 240–244.
Mohanty, S., Santangeli, P., Bai, R., Di Biase, L., Mohanty, P., Pump, A., & Natale, A. (2013). Variant rs2200733 on chromosome 4q25 confers increased risk of atrial fibrillation: evidence from a meta-analysis. Journal of Cardiovascular Electrophysiology, 24(2), 155–161.
Lozano-Velasco, E., Chinchilla, A., Martínez-Fernández, S., Hernández-Torres, F., Navarro, F., Lyons, G. E., Franco, D., & Aránega, A. E. (2011). Pitx2c modulates cardiac-specific transcription factors networks in differentiating cardiomyocytes from murine embryonic stem cells. Cells, Tissues, Organs, 194(5), 349–362.
Maekawa, K., Saito, Y., Ozawa, S., Adachi-Akahane, S., Kawamoto, M., Komamura, K., Shimizu, W., Ueno, K., Kamakura, S., Kamatani, N., Kitakaze, M., & Sawada, J. (2005). Genetic polymorphisms and haplotypes of the human cardiac sodium channel alpha subunit gene (SCN5A) in Japanese and their association with arrhythmia. Annals of Human Genetics, 69(Pt 4), 413–428.
Balouch, M. A., Kolek, M. J., & Darbar, D. (2014). Improved understanding of the pathophysiology of atrial fibrillation through the lens of discrete pathological pathways. Global Cardiology Science & Practice, 1, 24–36.
Butler, A. M., Yin, X., Evans, D. S., et al. (2012). Novel loci associated with PR interval in a genome-wide association study of 10 African American cohorts. Circulation. Cardiovascular Genetics, 5(6), 639–646.
Christley, R. M. (2010). Power and error: increased risk of false positive results in underpowered studies. The Open Epidemiology Journal, 3, 16–19.
Di Biase, L., Santangeli, P., & Natale, A. (2013). How to ablate long-standing persistent atrial fibrillation? Current Opinion in Cardiology, 1, 26–35.
Shoemaker, M. B., Bollmann, A., Lubitz, S. A., Ueberham, L., Saini, H., Montgomery, J., Edwards, T., Yoneda, Z., Sinner, M. F., Arya, A., Sommer, P., Delaney, J., Goyal, S. K., Saavedra, P., Kanagasundram, A., Whalen, S. P., Roden, D. M., Hindricks, G., Ellis, C. R., Ellinor, P. T., Darbar, D., & Husser, D. (2015). Common genetic variants and response to atrial fibrillation ablation. Circulation. Arrhythmia and Electrophysiology, 8(2), 296–302.
Acknowledgments
This study was partly funded by St. David’s Medical Center, Austin, TX.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Luigi Di Biase is a consultant for Hansen Medical, St. Jude Medical and Biosense Webster, Atricure and EpiEP. Dr. Natale received honoraria from Boston Scientific, Biosense Webster, Janssen, St. Jude Medical, Biotronik and Medtronic.
Additional information
Dr. Mohanty and Ms. Hall made equivalent contributions in the drafting of the manuscript, and are co-primary authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 251 kb)
Rights and permissions
About this article
Cite this article
Mohanty, S., Hall, A.W., Mohanty, P. et al. Novel association of polymorphic genetic variants with predictors of outcome of catheter ablation in atrial fibrillation: new directions from a prospective study (DECAF). J Interv Card Electrophysiol 45, 7–17 (2016). https://doi.org/10.1007/s10840-015-0069-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-015-0069-2