Abstract
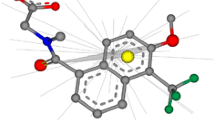
The identification of new, potent and selective inhibitors of important protein kinase targets is a major goal of drug discovery. Here we analyze the crystal structures of 55 protein kinase complexes with Type II inhibitors and find they adopt a conserved twisted V-shape, with an angle of 121 ± 8° and twist of 78 ± 8°. The tightly conserved twist appears important in ensuring ligands curve around the protein backbone and towards the deep pocket. From this, we develop predictive pharmacophore- and shape-based screens to identify Type II inhibitors from a database which also contains Type I inhibitors as decoys. Both approaches exhibit a good level of discrimination for Type II molecules. The most effective pharmacophore model requires six features and three excluded volume regions. Shape-based screening using ROCS generally performs at least as well as pharmacophore approaches. There is only a moderate dependence of shape-based or pharmacophore-based screens on the underlying conformer generator (MOE, Macromodel, Omega and SPE), as well as on ligand linkage chemistry (amide and urea). Finally, we apply our approach to retrieval of Type II inhibitors from a modified version of the DUD database, containing over 104,000 compounds. We observe good enrichment, providing further evidence that the in silico screens developed here will constitute useful guides for identification of small molecule inhibitors targetting protein kinases in their inactive conformational state.









Similar content being viewed by others
References
Zhang JM, Yang PL, Gray NS (2009) Nat Rev Cancer 9:28
Noble MEM, Endicott JA, Johnson LN (2004) Science 303:1800
Hanks SK, Quinn AM, Hunter T (1988) Science 241:42
Scapin G (2002) Drug Discov Today 7:601
Backes AC, Zech B, Felber B, Klebl B, Muller G (2008) Expert Opin Drug Discov 3:1427
Kufareva I, Abagyan R (2008) J Med Chem 51:7921
Backes AC, Zech B, Felber B, Klebl B, Muller G (2008) Expert Opin Drug Discov 3:1409
Liu Y, Gray NS (2006) Nat Chem Biol 2:358
Seeliger MA, Ranjitkar P, Kasap C, Shan YB, Shaw DE, Shah NP, Kuriyan J, Maly DJ (2009) Cancer Res 69:2384
Neumann L, Ritscher A, Muller G, Hafenbradl D (2009) J Comp Aided Mol Des 23:501
Pargellis C, Tong L, Churchill L, Cirillo PF, Gilmore T, Graham AG, Grob PM, Hickey ER, Moss N, Pav S, Regan J (2002) Nat Struct Biol 9:268
Zuccotto F, Ardini E, Casale E, Angiolini M (2010) J Med Chem 53:2681
McGregor MJ (2007) J Chem Inf Modell 47:2374
Xie QQ, Xie HZ, Ren JX, Li LL, Yang SY (2009) J Mol Graph Modell 27:751
Okram B, Nagle A, Adrian FJ, Lee C, Ren P, Wang X, Sim T, Xie YP, Wang X, Xia G, Spraggon G, Warmuth M, Liu Y, Gray NS (2006) Chem Biol 13:779
Nagar B, Bornmann WG, Pellicena P, Schindler T, Veach DR, Miller WT, Clarkson B, Kuriyan J (2002) Cancer Res 62:4236
Wan PTC, Garnett MJ, Roe SM, Lee S, Niculescu-Duvaz D, Good VM, Jones CM, Marshall CJ, Springer CJ, Barford D, Marais R (2004) Cell 116:855
Weisberg E, Manley PW, Breitenstein W, Bruggen J, Cowan-Jacob SW, Ray A, Huntly B, Fabbro D, Fendrich G, Hall-Meyers E, Kung AL, Mestan J, Daley GQ, Callahan L, Catley L, Cavazza C, Mohammed A, Neuberg D, Wright RD, Gilliland DG, Griffin JD (2005) Cancer Cell 7:129
Deak HL, Newcomb JR, Nunes JJ, Boucher C, Cheng AC, DiMauro EF, Epstein LF, Gallant P, Hodous BL, Huang X, Lee JH, Patel VF, Schneider S, Turci SM, Zhu XT (2008) Bioorg Med Chem Lett 18:1172
Araujo J, Mathew R, Armstrong AJ, Braud EL, Posadas E, Lonberg M, Gallick G, Trudel GC, Paliwal P, Logothetis CJ (2009) EJC Suppl 7:415
Oslob JD, Romanowski MJ, Allen DA, Baskaran S, Bui M, Elling RA, Flanagan WM, Fung AD, Hanan EJ, Harris S, Heumann SA, Hoch U, Jacobs JW, Lam J, Lawrence CE, McDowell RS, Nannini MA, Shen W, Silverman JA, Sopko MM, Tangonan BT, Teague J, Yoburn JC, Yu CH, Zhong M, Zimmerman KM, O’Brien T, Lew W (2008) Bioorg Med Chem Lett 18:4880
Rush TS, Grant JA, Mosyak L, Nicholls A (2005) J Med Chem 48:1489
Chemical Computing Group, Montreal, QC, Canada, 2003
Schrodinger; Schrodinger LLC (2005). http://www.schrodinger.com.
Omega; OpenEye Scientific Software (1997). http://www.eyesopen.com/.
SPE, Johnson and Johnson (2009)
Agrafiotis DK, Gibbs A, Zhu FQ, Izrailev S, Martin E (2006) Aust J Chem 59:874
Halgren TA (1999) J Comput Chem 20:720
Halgren TA (1996) J Comput Chem 17:490
Halgren TA (1996) J Comput Chem 17:520
Halgren TA (1996) J Comput Chem 17:553
Halgren TA, Nachbar RB (1996) J Comput Chem 17:587
Halgren TA (1996) J Comput Chem 17:616
Tresadern G, Agrafiotis DK (2009) J Chem Inf Modell 49:2786
Kirchmair J, Distinto S, Markt P, Schuster D, Spitzer GM, Liedl KR, Wolber G (2009) J Chem Inf Modell 49:678
Needleman SB, Wunsch CD (1970) J Mol Biol 48:443
Pipeline Pilot, Accelrys, Inc., San Diego, USA, 2010
Kirchmair J, Ristic S, Eder K, Markt P, Wolber G, Laggner C, Langer T (2007) J Chem Inf Modell 47:2182
Gribskov M, Robinson NL (1996) Comput Chem 20:25
O’Hare T, Shakespeare WC, Zhu XT, Eide CA, Rivera VM, Wang F, Adrian LT, Zhou TJ, Huang WS, Xu QH, Metcalf CA, Tyner JW, Loriaux MM, Corbin AS, Wardwell S, Ning YY, Keats JA, Wang YH, Sundaramoorthi R, Thomas M, Zhou D, Snodgrass J, Commodore L, Sawyer TK, Dalgarno DC, Deininger MWN, Druker BJ, Clackson T (2009) Cancer Cell 16:401
Potashman MH, Bready J, Coxon A, DeMelfi TM, DiPietro L, Doerr N, Elbaum D, Estrada J, Gallan P, Germain J, Gu Y, Harmange JC, Kaufman SA, Kendall R, Kim JL, Kumar GN, Long AM, Neervannan S, Patel VF, Polverino A, Rose P, van der Plas S, Whittington D, Zanon R, Zhao HL (2007) J Med Chem 50:4351
Bonnet P, Agrafiotis DK, Zhu FQ, Martin E (2009) J Chem Inf Modell 49:2242
Hodous BL, Geuns-Meyer SD, Hughes PE, Albrecht BK, Bellon S, Bready J, Caenepeel S, Cee VJ, Chaffee SC, Coxon A, Emery M, Fretland J, Gallant P, Gu Y, Hoffman D, Johnson RE, Kendall R, Kim JL, Long AM, Morrison M, Olivieri PR, Patel VF, Polverino A, Rose P, Tempest P, Wang L, Whittington DA, Zhao HL (2007) J Med Chem 50:611
Gill AL, Frederickson M, Cleasby A, Woodhead SJ, Carr MG, Woodhead AJ, Walker MT, Congreve MS, Devine LA, Tisi D, O’Reilly M, Seavers LCA, Davis DJ, Curry J, Anthony R, Padova A, Murray CW, Carr RAE, Jhoti H (2005) J Med Chem 48:414
Miyazaki Y, Matsunaga S, Tang J, Maeda Y, Nakano M, Philippe RJ, Shibahara M, Liu W, Sato H, Wang LP, Nolte RT (2005) Bioorg Med Chem Lett 15:2203
Grant JA, Gallardo MA, Pickup BT (1996) J Comput Chem 17:1653
Agrafiotis DK, Gibbs AC, Zhu FQ, Izrailev S, Martin E (2007) J Chem Inf Modell 47:1067
Getlik M, Gruêtter C, Simard JR, Kluêter S, Rabiller M, Rode HB, Robubi A, Rauh D (2009) J Med Chem 52:3915
Simard JR, Getlik Mê, Gruêtter C, Pawar V, Wulfert S, Rabiller M, Rauh D (2009) J Am Chem Soc 131:13286
Berger DM, Torres N, Dutia M, Powell D, Ciszewski G, Gopalsamy A, Levin JI, Kim KH, Xu W, Wilhelm J, Hu Y, Collins K, Feldberg L, Kim S, Frommer E, Wojciechowicz D, Mallon R (2009) Bioorg Med Chem Lett 19:6519
Han S, Mistry A, Chang JS, Cunningham D, Griffor M, Bonnette PC, Wang H, Chrunyk BA, Aspnes GE, Walker DP, Brosius AD, Buckbinder L (2009) J Bio Chem 21:13193
Huang N, Shoichet BK, Irwin JJ (2006) J Med Chem 49:6789
Jahn A, Hinselmann G, Fechner N, Zell A (2009) J Cheminform 1:14
Acknowledgments
This work was funded by the Biotechnology and Biological Sciences Research Council and Johnson & Johnson Pharmaceutical Research & Development. The authors would like to thank Dr. Trevor Howe and Dr. Berthold Wroblowski for their valuable comments; and Andrew Henry from Chemical Computing Group for his support in MOE scripting.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mucs, D., Bryce, R.A. & Bonnet, P. Application of shape-based and pharmacophore-based in silico screens for identification of Type II protein kinase inhibitors. J Comput Aided Mol Des 25, 569–581 (2011). https://doi.org/10.1007/s10822-011-9442-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-011-9442-0