Abstract
Purpose
High progesterone is associated with low implantation rate. Our previous study demonstrated that DNA methylation in endometrium was increased in women with high progesterone in IVF cycles. However, the DNA methylation status is still not yet confirmed, and how it affects endometrial receptivity in high progesterone is still unknown. Current study investigated the effects of high progesterone on DNA methylation and gene expression of adhesion molecules on endometrium during implantation window.
Methods
A cohort study included 20 women with high progesterone (HP) and 20 with normal progesterone (NP) on the day of human chorionic gonadotropin (hCG) administration after controlled ovarian hyperstimulation in IVF cycle. Endometrial tissues were collected on the 7th day after hCG administration. Immunohistochemical staining of DNA methyltransferases (DNMT1 and DNMT3B) and adhesion molecules (MUC1, CDH1 and CTNNB1) were performed. Methylation of MUC1, CDH1, and CTNNB1 promoter regions was detected by Sequenom MassARRAY or bisulfite sequencing PCR. RT-qPCR was used to quantify mRNA expression levels, and correlation of methylation and gene expression level of the adhesion molecules were determined.
Results
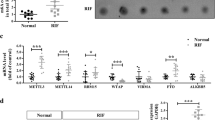
DNMT3B, but not DNMT1, in nucleus of luminal and glandular epithelial cells in HP group was significantly higher than that in NP group. Promoter regions of CDH1 and CTNNB1, but not MUC1, in endometrium of HP group were hypermethylated. Protein and mRNA expression of MUC1, CDH1, and CTNNB1 in endometrium of HP group was significantly lower than that in NP group. Level of DNA methylation was negatively correlated with the gene expression of CDH1 and CTNNB1, but not MUC1.
Conclusions
DNA hypermethylation and low expression of adhesion molecules on endometrium were associated with high progesterone during implantation window, which may contribute to the underlying epigenetic mechanism in the failure of IVF treatment.





Similar content being viewed by others
References
Schoolcraft W, Sinton E, Schlenker T, Huynh D, Hamilton F, Meldrum DR. Lower pregnancy rate with premature luteinization during pituitary suppression with leuprolide acetate. Fertil Steril. 1991;55(3):563–6.
Venetis CA, Kolibianakis EM, Papanikolaou E, Bontis J, Devroey P, Tarlatzis BC. Is progesterone elevation on the day of human chorionic gonadotropin administration associated with the probability of pregnancy in in vitro fertilization? A systematic review and meta-analysis. Hum Reprod Update. 2007;13(4):343–55. https://doi.org/10.1093/humupd/dmm007.
Bosch E, Labarta E, Crespo J, Simon C, Remohi J, Jenkins J, et al. Circulating progesterone levels and ongoing pregnancy rates in controlled ovarian stimulation cycles for in vitro fertilization: analysis of over 4000 cycles. Hum Reprod. 2010;25(8):2092–100. https://doi.org/10.1093/humrep/deq125.
Venetis CA, Kolibianakis EM, Bosdou JK, Tarlatzis BC. Progesterone elevation and probability of pregnancy after IVF: a systematic review and meta-analysis of over 60 000 cycles. Hum Reprod Update. 2013;19(5):433–57. https://doi.org/10.1093/humupd/dmt014.
Kolibianakis EM, Venetis CA, Bontis J, Tarlatzis BC. Significantly lower pregnancy rates in the presence of progesterone elevation in patients treated with GnRH antagonists and gonadotropins: a systematic review and meta-analysis. Curr Pharm Biotechnol. 2012;13(3):464–70.
Huang CC, Lien YR, Chen HF, Chen MJ, Shieh CJ, Yao YL, et al. The duration of pre-ovulatory serum progesterone elevation before hCG administration affects the outcome of IVF/ICSI cycles. Hum Reprod. 2012;27(7):2036–45. https://doi.org/10.1093/humrep/des141.
Kilicdag EB, Haydardedeoglu B, Cok T, Hacivelioglu SO, Bagis T. Premature progesterone elevation impairs implantation and live birth rates in GnRH-agonist IVF/ICSI cycles. Arch Gynecol Obstet. 2010;281(4):747–52. https://doi.org/10.1007/s00404-009-1248-0.
Legro RS, Ary BA, Paulson RJ, Stanczyk FZ, Sauer MV. Premature luteinization as detected by elevated serum progesterone is associated with a higher pregnancy rate in donor oocyte in-vitro fertilization. Hum Reprod. 1993;8(9):1506–11.
Labarta E, Martinez-Conejero JA, Alama P, Horcajadas JA, Pellicer A, Simon C, et al. Endometrial receptivity is affected in women with high circulating progesterone levels at the end of the follicular phase: a functional genomics analysis. Hum Reprod. 2011;26(7):1813–25. https://doi.org/10.1093/humrep/der126.
Van Vaerenbergh I, Fatemi HM, Blockeel C, Van Lommel L, In't Veld P, Schuit F, et al. Progesterone rise on HCG day in GnRH antagonist/rFSH stimulated cycles affects endometrial gene expression. Reprod BioMed Online. 2011;22(3):263–71. https://doi.org/10.1016/j.rbmo.2010.11.002.
Haouzi D, Bissonnette L, Gala A, Assou S, Entezami F, Perrochia H, et al. Endometrial receptivity profile in patients with premature progesterone elevation on the day of HCG administration. Biomed Res Int. 2014;2014:951937. https://doi.org/10.1155/2014/951937.
Li R, Qiao J, Wang L, Li L, Zhen X, Liu P, et al. MicroRNA array and microarray evaluation of endometrial receptivity in patients with high serum progesterone levels on the day of hCG administration. Reprod Biol Endocrinol. 2011;9:29. https://doi.org/10.1186/1477-7827-9-29.
Seppala M, Taylor RN, Koistinen H, Koistinen R, Milgrom E. Glycodelin: a major lipocalin protein of the reproductive axis with diverse actions in cell recognition and differentiation. Endocr Rev. 2002;23(4):401–30. https://doi.org/10.1210/er.2001-0026.
Lessey BA. Assessment of endometrial receptivity. Fertil Steril. 2011;96(3):522–9. https://doi.org/10.1016/j.fertnstert.2011.07.1095.
Brayman M, Thathiah A, Carson DD. MUC1: a multifunctional cell surface component of reproductive tissue epithelia. Reprod Biol Endocrinol. 2004;2:4. https://doi.org/10.1186/1477-7827-2-4.
Achache H, Revel A. Endometrial receptivity markers, the journey to successful embryo implantation. Hum Reprod Update. 2006;12(6):731–46. https://doi.org/10.1093/humupd/dml004.
Xiong Y, Wang J, Liu L, Chen X, Xu H, Li TC, et al. Effects of high progesterone level on the day of human chorionic gonadotropin administration in in vitro fertilization cycles on epigenetic modification of endometrium in the peri-implantation period. Fertil Steril. 2017;108(2):269–76.e1. https://doi.org/10.1016/j.fertnstert.2017.06.004.
Ziller MJ, Muller F, Liao J, Zhang Y, Gu H, Bock C, et al. Genomic distribution and inter-sample variation of non-CpG methylation across human cell types. PLoS Genet. 2011;7(12):e1002389. https://doi.org/10.1371/journal.pgen.1002389.
Smith ZD, Meissner A. DNA methylation: roles in mammalian development. Nat Rev Genet. 2013;14(3):204–20. https://doi.org/10.1038/nrg3354.
Okano M, Bell DW, Haber DA, Li E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell. 1999;99(3):247–57.
Li E, Bestor TH, Jaenisch R. Targeted mutation of the DNA methyltransferase gene results in embryonic lethality. Cell. 1992;69(6):915–26.
Liu L, Zhao L, Li TC, Zhu H, Lin X, Jin X, et al. Comparison of progesterone measurement on day of, and day after, HCG administration in IVF-embryo transfer cycles. Reprod BioMed Online. 2015;30(2):157–65. https://doi.org/10.1016/j.rbmo.2014.10.017.
Feng S, Jacobsen SE, Reik W. Epigenetic reprogramming in plant and animal development. Science. 2010;330(6004):622–7. https://doi.org/10.1126/science.1190614.
Jeltsch A, Jurkowska RZ. New concepts in DNA methylation. Trends Biochem Sci. 2014;39(7):310–8. https://doi.org/10.1016/j.tibs.2014.05.002.
Grady WM, Willis J, Guilford PJ, Dunbier AK, Toro TT, Lynch H, et al. Methylation of the CDH1 promoter as the second genetic hit in hereditary diffuse gastric cancer. Nat Genet. 2000;26(1):16–7. https://doi.org/10.1038/79120.
Li Y, Ran R, Guan Y, Zhu X, Kang S. Aberrant methylation of the E-cadherin gene promoter region in the endometrium of women with uterine fibroids. Reprod Sci. 2016;23(8):1096–102. https://doi.org/10.1177/1933719116630415.
Yamada N, Nishida Y, Tsutsumida H, Hamada T, Goto M, Higashi M, et al. MUC1 expression is regulated by DNA methylation and histone H3 lysine 9 modification in cancer cells. Cancer Res. 2008;68(8):2708–16. https://doi.org/10.1158/0008-5472.can-07-6844.
Hey NA, Graham RA, Seif MW, Aplin JD. The polymorphic epithelial mucin MUC1 in human endometrium is regulated with maximal expression in the implantation phase. J Clin Endocrinol Metab. 1994;78(2):337–42. https://doi.org/10.1210/jcem.78.2.8106621.
Thathiah A, Carson DD. MT1-MMP mediates MUC1 shedding independent of TACE/ADAM17. Biochem J. 2004;382(Pt 1):363–73. https://doi.org/10.1042/bj20040513.
Meseguer M, Aplin JD, Caballero-Campo P, O'Connor JE, Martin JC, Remohi J, et al. Human endometrial mucin MUC1 is up-regulated by progesterone and down-regulated in vitro by the human blastocyst. Biol Reprod. 2001;64(2):590–601. https://doi.org/10.1095/biolreprod64.2.590.
Hoffman LH, Olson GE, Carson DD, Chilton BS. Progesterone and implanting blastocysts regulate Muc1 expression in rabbit uterine epithelium. Endocrinology. 1998;139(1):266–71. https://doi.org/10.1210/endo.139.1.5750.
Simon C, Martin JC, Pellicer A. Paracrine regulators of implantation. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14(5):815–26. https://doi.org/10.1053/beog.2000.0121.
Acknowledgements
This work was supported by the Direct Grant for Research 2016/2017 (2016.058) to Y.X., H.X., and C C.W and Shenzhen Science Project Funding JCYJ20160427113429186 to T.Z. and Postdoctoral International Exchange Program to Y.X.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict(s) of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The paper was selected to present at the 34th annual meeting, European Society of Human Reproduction and Embryology, Barcelona, Spain, July 1–4, 2018.
Rights and permissions
About this article
Cite this article
Xiong, Y., Hu, L., Zhang, T. et al. Effects of high progesterone in in-vitro fertilization cycle on DNA methylation and gene expression of adhesion molecules on endometrium during implantation window. J Assist Reprod Genet 37, 33–43 (2020). https://doi.org/10.1007/s10815-019-01623-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-019-01623-6