Abstract
Purpose
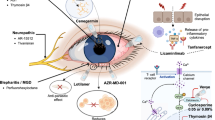
To report our experience with the 2% cyclosporin A (CsA) in a series of challenging inflammatory ocular surface diseases due to different etiologies.
Methods
The records of patients who received topical 2% CsA for different indications were reviewed retrospectively. Demographic characteristics, indications for treatment, patient symptoms and clinical findings were recorded.
Results
Fifty-two eyes of 52 patients were included. Mean age was 43.2 ± 14.3 (11–66) years with a F/M ratio of 34/18. Indications included pediatric acne rosacea (n = 4), adenoviral corneal subepithelial infiltrates (n = 12), filamentary keratitis (n = 14), pterygium recurrence (n = 15), herpetic marginal keratitis (n = 2) and graft versus host disease (n = 5 patients). Mean duration of treatment was 7.3 ± 2.8 (3–10) months. Forty-three (83%) patients reported favorable outcome with improvement in symptoms after a mean time of 4.4 ± 2.7 (2–6) months.
Conclusions
Topical 2% CsA may address the needs of different cases with ocular surface inflammation, as a safe option for long-term therapy.





Similar content being viewed by others
References
Utine CA, Stern M, Akpek EK (2010) Clinical review: topical ophthalmic use of cyclosporin A. Ocul Immunol Inflamm 18(5):352–361. https://doi.org/10.3109/09273948.2010.498657
Donnenfeld E, Pflugfelder SC (2009) Topical ophthalmic cyclosporine: pharmacology and clinical uses. Surv Ophthalmol 54:321–338
Boboridis KG, Konstas AGP (2018) Evaluating the novel application of cyclosporine 0.1% in ocular surface disease. Expert Opin Pharmacother 19:1027–1039
Leonardi A, Van Setten G, Amrane M, Amrane M, Ismail D, Garrigue JS et al (2016) Efficacy and safety of 0.1% cyclosporine A cationic emulsion in the treatment of severe dry eye disease: a multicenter randomized trial. Eur J Ophthalmol 26:287–296
Malta JB, Soong HK, Shtein RM, Musch DC, Rhoades W, Sugar A et al (2010) Treatment of ocular graft-versus-host disease with topical cyclosporine 0.05%. Cornea 29:1392–6
Rao SN (2006) Treatment of herpes simplex virus stromal keratitis unresponsive to topical prednisolone 1% with topical cyclosporine 0.05%. Am J Ophthalmol 141:771–2
Foulks GN (2006) Topical cyclosporine for treatment of ocular surface disease. Int Ophthalmol Clin 46:105–122
Sacchetti M, Mantelli F, Lambiase A (2014) Systematic review of randomized clinical trials on topical cyclosporine A for the treatment of dry eye disease. Br J Ophthalmol 98:1016–1022
Baudouin C, Figueiredo FC, Messmer EM, Ismail D, Amrane M, Garrigue JS et al (2017) A randomized study of the efficacy and safety of 0.1% cyclosporine A cationic emulsion in treatment of moderate to severe dry eye. Eur J Ophthalmol 27:520–530
Doan S, Gabison E, Gatinel D, Duong MH, Abitbol O, Hoang-Xuan T (2006) Topical cyclosporine A in severe steroid-dependent childhood phlyctenular keratoconjunctivitis. Am J Ophthalmol 141:62–66
Heiligenhaus A, Steuhl KP (1999) Treatment of HSV-1 stromal keratitis with topical cyclosporin A: a pilot study. Graefes Arch Clin Exp Ophthalmol 237:435–438
Reinhard T, Sundmacher R (1999) Topical cyclosporin A in Thygeson’s superficial punctate keratitis. Graefes Arch Clin Exp Ophthalmol 237:109–112
Hingorani M, Moodaley L, Calder VL, Buckley RJ, Lightman S (1998) A randomized, placebo-controlled trial of topical cyclosporin A in steroid dependent atopic keratoconjunctivitis. Ophthalmology 105:1715–1720
Pucci N, Novembre E, Cianferoni A, Lombardi E, Bernardini R, Caputo R et al (2002) Efficacy and safety of cyclosporine eyedrops in vernal keratoconjunctivitis. Ann Allergy Asthma Immunol 89:298–303
Cetinkaya A, Akova YA, Dursun D, Pelit A (2004) Topical cyclosporine in the management of shield ulcers. Cornea 23:194–200
Deshmukh R, Ting DSJ, Elsahn A, Mohammed I, Said DG, Dua HS (2022) Real-world experience of using ciclosporin-A 0.1% in the management of ocular surface inflammatory diseases. Br J Ophthalmol. 106(8):1087–1092
Pflugfelder SC (2004) Antiinflammatory therapy for dry eye. Am J Ophthalmol 137:337–342
Kashani S, Mearza AA (2008) Uses and safety profile of cyclosporin in ophthalmology. Expert Opin Drug Saf 7:79–89
Aragona P (2014) Topical cyclosporine: are all indications justified? Br J Ophthalmol 98(8):1001–1002
Perry HD, Doshi-Carnevale S, Donnenfeld ED, Solomon R, Biser SA, Bloom AH (2006) Efficacy of commercially available topical cyclosporine A 0.05% in the treatment of meibomian gland dysfunction. Cornea 25:171–175
Rubin M, Rao SN (2006) Efficacy of topical cyclosporin 0.05% in the treatment of posterior blepharitis. J Ocul Pharmacol Ther 22:47–53
Schechter BA, Katz RS, Friedman LS (2009) Efficacy of topical cyclosporine for the treatment of ocular rosacea. Adv Ther 26:651–659
van Zuuren EJ, Kramer S, Carter B, Graber MA, Fedorowicz Z (2011) Interventions for rosacea. Cochrane Database Syst Rev 3:Cd003262
Rousta ST (2017) Pediatric blepharokeratoconjunctivitis: is there a “right” treatment? Curr Opin Ophthalmol 28:449–453
Albietz J, Sanfilippo P, Troutbeck R, Lenton LM (2003) Management of filamentary keratitis associated with aqueous-deficient dry eye. Optom Vis Sci 80:420–430
Marsh P, Pflugfelder SC (1999) Topical nonpreserved methylprednisolone therapy for keratoconjunctivitis sicca in Sjogren syndrome. Ophthalmology 106:811–816
Grinbaum A, Yassur I, Avni I (2001) The beneficial effect of diclofenac sodium in the treatment of filamentary keratitis. Arch Ophthalmol 119:926–927
Perry HD, Doshi-Carnevale S, Donnenfeld ED, Kornstein HS (2003) Topical cyclosporine A 0.5% as a possible new treatment for superior limbic keratoconjunctivitis. Ophthalmology 110:1578–1581
Jhanji V, Chan TC, Li EY, Agarwal K, Vajpayee RB (2015) Adenoviral keratoconjunctivitis. Surv Ophthalmol 60:435–443
Levinger E, Slomovic A, Sansanayudh W, Bahar I, Slomovic AR (2010) Topical treatment with 1% cyclosporine for subepithelial infiltrates secondary to adenoviral keratoconjunctivitis. Cornea 29:638–640
Okumus S, Coskun E, Tatar MG, Kaydu E, Yayuspayi R, Comez A et al (2012) Cyclosporine a 0.05% eye drops for the treatment of subepithelial infiltrates after epidemic keratoconjunctivitis. BMC Ophthalmol 18:12–42
Asena L, Şıngar Özdemir E, Burcu A, Ercan E, Çolak M, Altınörs DD (2017) Comparison of clinical outcome with different treatment regimens in acute adenoviral keratoconjunctivitis. Eye 31(5):781–787
Nuzzi R, Tridico F (2018) How to minimize pterygium recurrence rates: clinical perspectives. Clin Ophthalmol 12:2347–2362
Turan-Vural E, Torun-Acar B, Kivanc SA, Acar S (2011) The effect of topical 0.05% cyclosporine on recurrence following pterygium surgery. Clin Ophthalmol 5:881–5
Özülken K, Koç M, Ayar O, Hasiripi H (2012) Topical cyclosporine A administration after pterygium surgery. Eur J Ophthalmol 22(Suppl 7):5–10
Gunduz K, Ozdemir O (1997) Topical cyclosporin as an adjunct to topical acyclovir treatment in herpetic stromal keratitis. Ophthalmic Res 29:405–408
Funding
The authors declare that no funds, grants or other support were received during the preparation of this manuscript. The authors have no relevant financial or nonfinancial interests to disclose.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, data collection and analysis. The first draft of the manuscript was written by LA, MD and DA, MD commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was approved by the Institutional Review Board of Baskent University Faculty of Medicine and adhered to the tenets of the Declaration of Helsinki.
Informed consent
Informed consent was obtained for the off-label use of topical 2% cyclosporin A from all individual participants included in this retrospective study.
Consent for publication
The authors affirm that participants provided informed consent for publication of the images in Figs. 1, 2, 3, 4 and 5.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asena, L., Dursun Altınörs, D. Application of topical 2% cyclosporine A in inflammatory ocular surface diseases. Int Ophthalmol 43, 3943–3952 (2023). https://doi.org/10.1007/s10792-023-02796-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-023-02796-x