Abstract
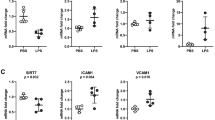
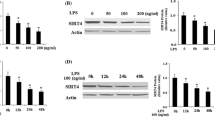
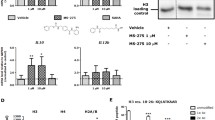
Inflammatory response in the pulmonary endothelium drives the pathogenesis of acute lung injury and sepsis. Sirtuin 6 (SIRT6), a member of class III NAD+-dependent deacetylases belonging to the sirtuin family, regulates senescence, metabolism, and inflammation and extends lifespan in mice and model organisms. However, the role of SIRT6 in pulmonary endothelial inflammation is unknown. Thus, we hypothesized that SIRT6 suppresses inflammatory response in human lung microvascular cells (HLMEC) and ensues monocyte adhesion to endothelial cells. Primary HLMECs were treated with control or SIRT6 adenovirus or SIRT6 agonist, with or without lipopolysaccharide (LPS) treatment. We observed that treatment with LPS did not affect the protein expression of SIRT6 in HLMECs. However, adenovirus-mediated SIRT6 overexpression attenuated LPS-induced VCAM1 gene and protein expression, followed by decreased monocyte adhesion to endothelial cells. Similarly, activation of SIRT6 by a recently reported SIRT6 activator UBCS039, but not the regioisomer negative control compound UBCS060, ameliorated LPS-induced VCAM1 mRNA and protein expression as well as monocyte adhesion. Moreover, luciferase assay revealed that SIRT6 adenovirus decreased the activity of NF-κB, the master regulator of vascular inflammation. Taken together, these results indicate that molecular and pharmacological activation of SIRT6 protects against lung microvascular inflammation via suppressing NF-κB activation, implicating the therapeutic potential of the SIRT6 activators for lung disorders associated with microvascular inflammation.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Seeley, E.J., P. Rosenberg, and M.A. Matthay. 2013. Calcium flux and endothelial dysfunction during acute lung injury: A STIMulating target for therapy. The Journal of clinical investigation 123: 1015–1018.
Xu, S., I. Ilyas, P.J. Little, et al. 2021. Endothelial dysfunction in atherosclerotic cardiovascular diseases and beyond: From mechanism to pharmacotherapies. Pharmacological reviews 73: 924–967.
Liu, G., H. Chen, H. Liu, W. Zhang, and J. Zhou. 2021. Emerging roles of SIRT6 in human diseases and its modulators. Medicinal Research Reviews 41: 1089–1137.
Xu, S., P. Bai, and Z.G. Jin. 2016. Sirtuins in cardiovascular health and diseases. Trends in endocrinology and metabolism: TEM 27: 677–678.
Zhao, Y., X. Jia, X. Yang, et al. 2022. Deacetylation of Caveolin-1 by Sirt6 induces autophagy and retards high glucose-stimulated LDL transcytosis and atherosclerosis formation. Metabolism: Cinical and Experimental 155162.
Taylor, J.R., J.G. Wood, E. Mizerak, et al. 2022. Sirt6 regulates lifespan in Drosophila melanogaster. Proceedings of the National Academy of Sciences of the United States of America 119.
Kanfi, Y., S. Naiman, G. Amir, et al. 2012. The sirtuin SIRT6 regulates lifespan in male mice. Nature 483: 218–221.
Li, X., L. Liu, T. Li, et al. 2021.SIRT6 in senescence and aging-related cardiovascular diseases. Frontiers in Cell and Developmental Biology 9: 641315.
Xu, S., M. Yin, M. Koroleva, et al. 2016. SIRT6 protects against endothelial dysfunction and atherosclerosis in mice. Aging 8: 1064–82.
Chang, A.R., C.M. Ferrer, and R. Mostoslavsky. 2020. SIRT6, a mammalian deacylase with multitasking abilities. Physiological Reviews 100: 145–169.
Xu, S., Y. Xu, P. Liu, et al. 2019. The novel coronary artery disease risk gene JCAD/KIAA1462 promotes endothelial dysfunction and atherosclerosis. European Heart Journal 40: 2398–2408.
Liu, R., H. Liu, Y. Ha, R.G. Tilton, and W. Zhang. 2014. Oxidative stress induces endothelial cell senescence via downregulation of Sirt6. BioMedical Research International 2014: 902842.
Iachettini, S., D. Trisciuoglio, D. Rotili, et al. 2018. Pharmacological activation of SIRT6 triggers lethal autophagy in human cancer cells. Cell Death and Disease 9: 996.
You, W., D. Rotili, T.M. Li, et al. 2017. Structural basis of sirtuin 6 activation by synthetic small molecules. Angewandte Chemie (International ed in English) 56: 1007–1011.
Lappas, M. 2012. Anti-inflammatory properties of sirtuin 6 in human umbilical vein endothelial cells. Mediators of Inflammation 2012: 597514.
Qin, Y., L. Cao, and L. Hu. 2019. Sirtuin 6 mitigated LPS-induced human umbilical vein endothelial cells inflammatory responses through modulating nuclear factor erythroid 2-related factor 2. Journal of Cellular Biochemistry.
Wang, L., Y. Cao, B. Gorshkov, et al. 2019. Ablation of endothelial Pfkfb3 protects mice from acute lung injury in LPS-induced endotoxemia. Pharmacological Research 146: 104292.
Daci, A., L. Da Dalt, R. Alaj, et al. 2020. Rivaroxaban improves vascular response in LPS-induced acute inflammation in experimental models. PLoS ONE 15: e0240669.
Lee, B.K., W.J. Lee, and Y.S. Jung. 2017.Chrysin attenuates VCAM-1 expression and monocyte adhesion in lipopolysaccharide-stimulated brain endothelial cells by preventing NF-kappaB signaling. International Journal of Molecular Sciences 18.
Mambetsariev, I., Y. Tian, T. Wu, et al. 2014. Stiffness-activated GEF-H1 expression exacerbates LPS-induced lung inflammation. PLoS ONE 9: e92670.
Jang, H.Y., S. Gu, S.M. Lee, and B.H. Park. 2016. Overexpression of sirtuin 6 suppresses allergic airway inflammation through deacetylation of GATA3. The Journal of Allergy and Clinical Immunology 138: 1452-1455.e13.
Wang, J., Y. Cai, and Z. Sheng. 2020. Experimental studies on the protective effects of the overexpression of lentivirus-mediated sirtuin 6 on radiation-induced lung injury. Advances in Clinical and Experimental Medicine : Official Organ Wroclaw Medical University 29: 873–877.
Jiao, F., Z. Zhang, H. Hu, Y. Zhang, and Y. Xiong. 2022. SIRT6 activator UBCS039 inhibits thioacetamide-induced hepatic injury in vitro and in vivo. Frontiers in Pharmacology 13: 837544.
Zou, Y., J. Zhang, J. Xu, et al. 2021. SIRT6 inhibition delays peripheral nerve recovery by suppressing migration, phagocytosis and M2-polarization of macrophages. Cell & Bioscience 11: 210.
Jin, Z., B. Wang, L. Ren, et al. 2023. 20-Hydroxyecdysone inhibits inflammation via SIRT6-mediated NF-kappaB signaling in endothelial cells. Biochimica et Biophysica Acta Molecular Cell Research 1870: 119460.
Li, P., Y. Jin, F. Qi, et al. 2018. SIRT6 acts as a negative regulator in dengue virus-induced inflammatory response by targeting the DNA binding domain of NF-kappaB p65. Frontiers in Cellular and Infection Microbiology 8: 113.
Funding
This study was partly supported by grants from Guangdong Basic and Applied Basic Research Foundation (no. 2021A1515011001 to JPW), from FISR2019_00374 (MeDyCa) (to AM), and from USA National Institute of Health (HL130167 and HL141171 to ZGJ).
Author information
Authors and Affiliations
Contributions
SWX and ZGJ participated in research design. JPW, JQL, AM, CS, and WZ conducted experiments. JPW, SWX, and ZGJ performed data analysis. JPW and SWX contributed to the writing of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Luo, J., Rotili, D. et al. SIRT6 Protects Against Lipopolysaccharide-Induced Inflammation in Human Pulmonary Lung Microvascular Endothelial Cells. Inflammation 47, 323–332 (2024). https://doi.org/10.1007/s10753-023-01911-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01911-5