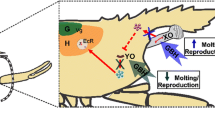
Abstract
Short-chain chlorinated paraffins (SCCPs) are ubiquitous, persistent chemicals whose toxicity and potential impacts on aquatic ecosystems have been overlooked for many years. This study aims to determine the impact of these chemicals on rotifers. As sexual reproduction is often more sensitive to toxicants than amictic reproduction, this study focuses on endpoints describing sexual reproduction and on the impact on the gene expression of three receptors associated with reproduction: retinoid-x-receptor (rxr), retinoid activated receptor (rar) and membrane-associated progesterone receptor (mapr). Brachionus calyciflorus was exposed to environmental concentrations of SCCPs (10,100 and 1000 ng/L) and the impact on sexual reproduction (mixis rate, fertilization rate, resting egg production and viability, inter-generational effects) was assessed. Variations in the gene expressions of rxr, rar and mapr were also studied through RT-qPCR analysis. Our results show that SCCPs increase the mixis rate but decrease the fertilization rate, leading to unaltered resting egg production. Exposure to SCCPs also affected the mRNA levels of the studied receptors in ways which could be linked to observed changes in mixis and fertilization rates.


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Andersen, R. A., 2005. Algal culturing techniques, Elsevier Academic Press, London:, 558.
Basconcillo, L., S. Backus, D. McGoldrick, D. Zaruk, E. Sverko & D. Muir, 2015. Current status of short- and medium chain polychlorinated n-alkanes in top predatory fish across Canada. Chemosphere. 127: 93–100. https://doi.org/10.1016/j.chemosphere.2015.01.016.
Bezchlebová, J., J. Černohlávková, K. Kobetičová, J. Lána, I. Sochová & J. Hofman, 2007. Effects of short-chain chlorinated paraffins on soil organisms. Ecotoxicology and Environmental Safety 67(2): 206–211. https://doi.org/10.1016/j.ecoenv.2006.12.015.
Castells, P., F. J. Santos & M. T. Galceran, 2004. Solid-phase extraction versus solid-phase microextraction for the determination of chlorinated paraffins in water using gas chromatography negative chemical ionisation mass spectrometry. Journal of Chromatography A 1025(2): 157–162. https://doi.org/10.1016/j.chroma.2003.10.069.
Chen, M. Y., X. J. Luo, X. L. Zhang, M. J. He, S. J. Chen & B. X. Mai, 2011. Chlorinated paraffins in sediments from the Pearl River Delta, South China: Spatial and temporal distributions and implication for processes. Environmental Science and Technology 45(23): 9936–9943. https://doi.org/10.1021/es202891a.
Cruciani, V., C. Iovine, J. P. Thomé & C. Joaquim-Justo, 2016. Impact of three phthalate esters on the sexual reproduction of the monogonont rotifer, Brachionus calyciflorus. Ecotoxicology 25: 192–200. https://doi.org/10.1007/s10646-015-1579-5.
Dahms, H. U., A. Hagiwara & J. S. Lee, 2011. Ecotoxicology, ecophysiology, and mechanistic studies with rotifers. Aquatic Toxicology 101(1): 1–12. https://doi.org/10.1016/J.AQUATOX.2010.09.006.
Dawson, M. I. & Z. Xia, 2012. The retinoid X receptors and their ligands. Biochimica Et Biophysica (BBA) Acta Molecular and Cell Biology of Lipids 1821(1): 21–56. https://doi.org/10.1016/J.BBALIP.2011.09.014.
EPA, 1985. In Weber, C. I. & W. H. Peltier (eds), Methods for measuring acute Ttxicity of effluents to freshwater and marine organisms. US Environmental Protection Agency, Washington.
Geissen, V., H. Mol, E. Klumpp, G. Umlauf, M. Nadal, M. van der Ploeg, S. E. A. T. M. van de Zee & C. J. Ritsema, 2015. Emerging pollutants in the environment: A challenge for water resource management. International Soil and Water Conservation Research 3(1): 57–65. https://doi.org/10.1016/j.iswcr.2015.03.002.
Geng, N., Y. Luo, R. Cao, X. Song, F. Li, F. Wang, Y. Gong, L. Xing, H. Zhang & J. Chen, 2021. Effect of short-chain chlorinated paraffins on metabolic profiling of male SD rats. Science of the Total Environment 750: 141404. https://doi.org/10.1016/j.scitotenv.2020.141404.
Germain, P., P. Chambon, G. Eichele, R. M. Evans, M. A. Lazar, M. Leid, A. R. De Lera, R. Lotan, D. J. Mangelsdorf & H. Gronemeyer, 2006. International union of pharmacology. LX. Retinoic Acid Receptors. Pharmacological Reviews 58(4): 712–725. https://doi.org/10.1124/PR.58.4.4.
Gismondi, E., H. M. Cauchie, V. Cruciani & C. Joaquim-Justo, 2019. Targeted impact of cyproterone acetate on the sexual reproduction of female rotifers. Ecotoxicology 28(6): 643–649. https://doi.org/10.1007/S10646-019-02063-9/FIGURES/3.
Houde, M., D. C. G. Muir, G. T. Tomy, D. M. Whittle, C. Teixeira & S. Moore, 2008. Bioaccumulation and trophic magnification of short- and medium-chain chlorinated paraffins in food webs from Lake Ontario and Lake Michigan. Environmental Science and Technology 42: 3893–3899. https://doi.org/10.1021/es703184s.
Jeong, C. B., H. S. Kim, H. M. Kang, Y. H. Lee, B. Zhou, J. Choe & J. S. Lee, 2017. Genome-wide identification of ATP-binding cassette (ABC) transporters and conservation of their xenobiotic transporter function in the monogonont rotifer (Brachionus koreanus). Comparative Biochemistry and Physiology Part D: Genomics and Proteomics 21: 17–26. https://doi.org/10.1016/j.cbd.2016.10.003.
Joaquim-Justo, C. & E. Gismondi, 2021. Expression variations of two retinoid signaling pathway receptors in the rotifer Brachionus calyciflorus exposed to three endocrine disruptors. Ecotoxicology 30(2): 343–350. https://doi.org/10.1007/s10646-020-02339-5.
Ke, L. X., Y. L. Xi, C. W. Zha & L. L. Dong, 2009. Effects of three organophosphorus pesticides on population growth and sexual reproduction of rotifer Brachionus calyciflorus Pallas. Acta Ecologica Sinica 29(3): 182–185. https://doi.org/10.1016/J.CHNAES.2009.07.008.
Kim, D. H., H. S. Kim, D. S. Hwang, H. J. Kim, A. Hagiwara, J. S. Lee & C. B. Jeong, 2017. Genome-wide identification of nuclear receptor (NR) genes and the evolutionary significance of the NR1O subfamily in the monogonont rotifer Brachionus spp. General and Comparative Endocrinology 252: 219–225. https://doi.org/10.1016/j.ygcen.2017.06.030.
Kim, H. S., B. Y. Lee, J. Han, C. B. Jeong, D. S. Hwang, M. C. Lee, H. M. Kang, D. H. Kim, H. J. Kim, S. Papakostas, S. A. J. Declerck, I. Y. Choi, A. Hagiwara, H. G. Park & J. S. Lee, 2018. The genome of the freshwater monogonont rotifer Brachionus calyciflorus. Molecular Ecology Resources 18(3): 646–655. https://doi.org/10.1111/1755-0998.12768.
Kim, D. H., E. Byeon, M. S. Kim, et al., 2022. The genome of the marine rotifer Brachionus manjavacas: Genome-wide identification of 310 G protein-coupled receptor (GPCR) genes. Marine Biotechnology. 24: 226–242. https://doi.org/10.1007/s10126-022-10102-6.
Kubickova, B., C. Ramwell, K. Hilscherova & M. N. Jacobs, 2021. Highlighting the gaps in hazard and risk assessment of unregulated endocrine active substances in surface waters: retinoids as a European case study. Environmental Sciences Europe 33(1): 1–38. https://doi.org/10.1186/S12302-020-00428-0.
Li, H., J. Fu, W. Pan, P. Wang, Y. Li, Q. Zhang, Y. Wang, A. Zhang, Y. Liang & G. Jiang, 2017. Environmental behaviour of short-chain chlorinated paraffins in aquatic and terrestrial ecosystems of Ny-Ålesund and London Island, Svalbard, in the Arctic. Science of the Total Environment 590–591: 163–170. https://doi.org/10.1016/j.scitotenv.2017.02.192.
Liu, L., Y. Li, M. Coelhan, H. M. Chan, W. Ma & L. Liu, 2016. Relative developmental toxicity of short-chain chlorinated paraffins in zebrafish (Danio rerio) embryos. Environmental Pollution 219: 1122–1130. https://doi.org/10.1016/j.envpol.2016.09.016.
Livak, K. J. & T. D. Schmittgen, 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4): 402–408. https://doi.org/10.1006/METH.2001.1262.
Lv, L., T. Snell, J. Yang, S. Li & W. Zhu, 2010. Effects of fenitrothion on life history parameters of the rotifer Brachionus calyciflorus. Journal of Freshwater Ecology 25(4): 589–598. https://doi.org/10.1080/02705060.2010.9664408.
Ma, X., H. Zhang, Z. Wang, Z. Yao, J. Chen & J. Chen, 2014. Bioaccumulation and trophic transfer of short chain chlorinated paraffins in a marine food web from Liaodong Bay, North China. Environmental Science and Technology 48(10): 5964–5971. https://doi.org/10.1021/es500940p.
Nicholls, C. R., C. R. Allchin & R. J. Law, 2001. Levels of short and medium chain length polychlorinated n-alkanes in environmental samples from selected industrial areas in England and Wales. Environmental Pollution 114(3): 415–430. https://doi.org/10.1016/S0269-7491(00)00230-X.
Park, J. C., D. H. Kim, M. C. Lee, J. Han, H. J. Kim, A. Hagiwara, U. K. Hwang, H. G. Park & J. S. Lee, 2018. Genome-wide identification of the entire 90 glutathione S-transferase (GST) subfamily genes in four rotifer Brachionus species and transcriptional modulation in response to endocrine disrupting chemicals. Comparative Biochemistry and Physiology Part D: Genomics and Proteomics 28: 183–195. https://doi.org/10.1016/j.cbd.2018.09.003.
Preston, B. L., T. W. Snell, T. L. Robertson & B. J. Dingmann, 2000. Use of freshwater rotifer Brachionus calyciflorus in screening assay for potential endocrine disruptors. Environmental Toxicology and Chemistry 19(12): 2923–2928. https://doi.org/10.1002/ETC.5620191212.
Radix, P., G. Severin, K. W. Schramm & A. Kettrup, 2002. Reproduction disturbances of Brachionus calyciflorus (rotifer) for the screening of environmental endocrine disrupters. Chemosphere 47: 1097–1101. https://doi.org/10.1016/S0045-6535(01)00335-6.
Ricci, C. & M. Balsamo, 2000. The biology and ecology of lotic rotifers and gastrotrichs. Freshwater Biology. 44: 15–28. https://doi.org/10.1046/j.1365-2427.2000.00584.x.
Segers, H., 2007. Annotated checklist of the rotifers (Phylum Rotifera), with notes on nomenclature, taxonomy and distribution. Zootaxa 1564: 1–104. https://doi.org/10.11646/zootaxa.1564.1.1.
Segers, H., 2008. Global diversity of rotifers (Rotifera) in freshwater. Hydrobiologia 595(1): 49–59. https://doi.org/10.1007/s10750-007-9003-7.
Serra, M. & T. W. Snell, 2009. Sex loss in monogonont rotifers. In Schön, I., K. Martens & P. Dijk (eds), Lost Sex. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2770-2_14.
Snell, T., 2011. A review of the molecular mechanisms of monogonont rotifer reproduction. Hydrobiologia 662: 89–97. https://doi.org/10.1007/s10750-010-0483-5.
Snell, T. W. & M. J. Carmona, 1994. Surface glycoproteins in copepods: potential signals for mate recognition. Hydrobiologia 292–293(1): 255–264. https://doi.org/10.1007/BF00229949/METRICS.
Snell, T. W., B. D. Moffat, C. Janssen & G. Persoone, 1991. Acute toxicity tests using rotifers: IV. Effects of cyst age, temperature and salinity on the sensitivity of Brachionus calyciflorus. Ecotoxicology and Environmental Safety 21(3): 308–317. https://doi.org/10.1016/0147-6513(91)90070-6.
Sprengel, J., P. A. Behnisch, H. Besselink, A. Brouwer & W. Vetter, 2021. In vitro human cell-based TTR-TRβ CALUX assay indicates thyroid hormone transport disruption of short-chain, medium-chain and long-chain chlorinated paraffins. Archives of Toxicology 95(4): 1391–1396. https://doi.org/10.1007/s00204-021-02994-5.
Sun, R., X. Luo, B. Tang, L. Chen, Y. Liu & B. Mai, 2017. Bioaccumulation of short chain chlorinated paraffins in a typical freshwater food web contaminated by e-waste in South China: Bioaccumulation factors, tissue distribution, and trophic transfer. Environmental Pollution 222: 165–174. https://doi.org/10.1016/j.envpol.2016.12.060.
Thompson, R. & J. Madeley, 1983. The acute and chronic toxicity of a chlorinated paraffin to the mysid shrimp (Mysidopsis bahia), Imperial Chemical Industries PLC, Devon:
Wallace, R. L., T. W. Snell & H. A. Smith, 2015. Phylum rotifera, Thorp and Covich’s Freshwater Invertebrates: Ecology and General Biology 4th ed. Elsevier, Amsterdam: 225–271. https://doi.org/10.1016/B978-0-12-385026-3.00013-9.
Warnasuriya, G. D., B. M. Elcombe, J. R. Foster & C. R. Elcombe, 2010. A Mechanism for the induction of renal tumours in male Fischer 344 rats by short-chain chlorinated paraffins. Archives of Toxicology 84(3): 233–243. https://doi.org/10.1007/s00204-009-0489-9.
Won, E. J., J. Han, D. H. Kim, H. U. Dahms, J. S. Lee, E. J. Won, J. Han, D. H. Kim, J. S. Lee & H. U. Dahms, 2017. Rotifers in ecotoxicology. Rotifers. https://doi.org/10.1007/978-981-10-5635-2_10.
Yang, J. & T. W. Snell, 2010. Effects of progesterone, testosterone, and estrogen on sexual reproduction of the rotifer Brachionus calyciflorus. International Review of Hydrobiology 95(6): 441–449. https://doi.org/10.1002/IROH.201011267.
Zhang, L., J. Niu, Y. Li, Y. Wang & D. Sun, 2013. Evaluating the sub-lethal toxicity of PFOS and PFOA using rotifer Brachionus calyciflorus. Environmental Pollution 180: 34–40. https://doi.org/10.1016/j.envpol.2013.04.031.
Zhang, L., J. Niu, Y. Wang, J. Shi & Q. Huang, 2014. Chronic effects of PFOA and PFOS on sexual reproduction of freshwater rotifer Brachionus calyciflorus. Chemosphere 114: 114–120. https://doi.org/10.1016/j.chemosphere.2014.03.099.
Funding
This work was supported by the Belgian national funds for scientific research (Fonds National de la Recherche Scientifique—FNRS) under Grant no FC 36575.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Guest editors: Maria Špoljar, Diego Fontaneto, Elizabeth J. Walsh & Natalia Kuczyńska-Kippen / Diverse Rotifers in Diverse Ecosystems
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jaegers, J., Michaux, C., Gismondi, É. et al. Effects of short-chain chlorinated paraffins on the sexual reproduction of Brachionus calyciflorus: a populational and biomolecular study. Hydrobiologia 851, 3213–3224 (2024). https://doi.org/10.1007/s10750-023-05348-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05348-7