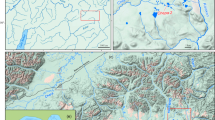
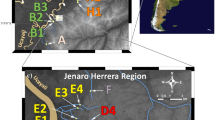
Abstract
Arctic charr Salvelinus alpinus (L.) complex has two or more sympatric forms in many lakes representing a subject for discussions on the mechanisms of their origin and taxonomic status. To determine the number of sympatric forms in three large lakes of the Norilo-Pyasinskaya water system (Taimyr) and their reproductive and phylogenetic relationships, 17 microsatellite loci, sequences of the mtDNA control region, and several morphological characteristics were analyzed. Our data indicate that eight reproductively isolated forms inhabit lakes Lama and Kapchuk, and at least three forms are distributed in Lake Sobach’e. The origin of the forms is associated with the secondary contact and hybridization of the European and Siberian phylogenetic lineages. The same names of the forms from different lakes of Taimyr and Chukotka, for example, Boganida charr and Dryagin’s charr, may reflect their certain morphological similarity, but rarely close relationships. Our data indirectly indicate that introgressive hybridization and biogeographical changes of the lakes within the region in the last glacial and post-glacial periods played a significant role in the formation of a high Arctic charr biodiversity. The problem associated with systematics, taxonomy, and conservation of the forms in Taimyr lakes is complex and should be solved gradually.





Similar content being viewed by others
Data availability
Sequence data are downloaded at GenBank; accession numbers MZ934408–MZ934411.
References
Abbott, R., D. S. Albach, J. W. Ansell, S. Arntzen, et al., 2013. Hybridization and speciation. Journal of Evolutionary Biology 26: 229–246.
Aguilée, R., D. Claessen & A. Lambert, 2013. Adaptive radiation driven by the interplay of eco-evolutionary and landscape dynamics: adaptive radiation in dynamic landscapes. Evolution 67: 1291–1306.
Alekseyev, S. S., R. Bajno, N. V. Gordeeva, J. D. Reist, M. Power, A. F. Kirillov, V. P. Samusenok & A. N. Matveev, 2009. Phylogeography and sympatric differentiation of the Arctic charr Salvelinus alpinus (L.) complex in Siberia as revealed by mtDNA sequence analysis. Journal of Fish Biology 75: 368–392.
Alekseyev, S. S., M. Yu. Pichugin, V. P. Samusenok, N. V. Gordeeva & A. L. Yur’ev, V. S. Khlystov & A. N. Matveev, 2019. Reproductive strategies of Arctic charr Salvelinus alpinus (L.) forms in Kiryalta lakes, Transbaikalia. Russia. Hydrobiologia 840: 113–136.
Allendorf, F. W. & G. H. Thorgaard, 1984. Tetraploidy and the evolution of salmonid fishes. In Turner, B. J. (ed), Evolutionary Genetics of Fishes Plenum Press, New York: 1–53.
Anderson, E. C. & K. K. Dunham, 2008. The influence of family groups on inferences made with the program Structure. Molecular Ecology Resources 8: 1219–1229.
Arnold, M. L., 2006. Evolution Through Genetic Exchange, Oxford University Press, Oxford:
Arkhipov, C. A., 1997. Chronology of geological events of the late Pleistocene in Western Siberia. Geologiya i Geofizika 38: 1863–1884 (In Russian).
Bandelt, H.-J., P. Forster & A. Rohl, 1999. Median-joining networks for inferring intraspecific phylogenies. Molecular Biology and Evolution 16: 37–48.
Barton, N. H., 2013. Does hybridization influence speciation? Journal of Evolutionary Biology 26: 267–269.
Batchelor, C. L., M. Margold, M. Krapp, C. R. Stokes, J. B. Murton & A. Manica, 2019. The configuration of Northern Hemisphere ice sheets through the Quaternary. Nature Communications 10: 3713.
Behnke, R. J., 1972. The systematics of salmonid fishes of recently glaciated lakes. Journal of the Fisheries Research Board of Canada 29: 639–671.
Behnke, R. J., 1980. A systematic review of the genus Salvelinus. In Balon, E. K. (ed), Charrs: Salmonid Fishes of the Genus Salvelinus Junk Publishers, The Hague, Dr. W: 441–481.
Berg, L. S., 1948. Freshwater Fishes of the USSR and Adjacent Countries, 4th ed. USSR Academy of Sciences, Moscow: (In Russian).
Bolnick, D. I. & B. M. Fitzpatrick, 2007. Sympatric speciation: models and empirical evidence. Annual Review of Ecology, Evolution, and Systematics 38: 459–487.
Bolshianov, DYu., O. M. Antonov, G. B. Fedorov & M. V. Pavlov, 2007. Glaciation of the Putorana Plateau during the last glacial maximum. Izvestiya Russkogo Geograficheskogo Obshchestva 139: 47–61 (In Russian).
Brunner, P. C., M. R. Douglas, A. Osinov, C. C. Wilson & L. Bernatchez, 2001. Holarctic phylogeography of Arctic charr (Salvelinus alpinus L.) inferred from mitochondrial DNA sequences. Evolution 55: 573–586.
Buerkle, C. A. & L. H. Rieseberg, 2008. The rate of genome stabilization in homoploid hybrid species. Evolution 62: 266–275.
Campana, S. E. & J. M. Casselman, 1993. Stock discrimination using otolith shape analysis. Canadian Journal of Fisheries and Aquatic Sciences 50: 1062–1083.
Chapman, M. A. & J. M. Burke, 2007. Genetic divergence and hybrid speciation. Evolution 61: 1773–1780.
Claessen, D., J. Andersson, L. Persson & A. M. Roos, 2008. The effect of population size and recombination on delayed evolution of polymorphism and speciation in sexual populations. The American Naturalist 172: E18–E34.
Cornuet, J.-M. & G. Luikart, 1996. Description and power analysis of two tests for detecting recent population bottlenecks from allele frequency data. Genetics 144: 2001–2014.
Coyne, J. A. & H. A. Orr, 2004. Speciation, Sinauer Associates, Sunderland, MA:
Czekanski-Moir, J. E. & R. J. Rundell, 2019. The ecology of nonecological speciation and nonadaptive radiations. Trends in Ecology and Evolution 34: 400–415.
Do, C., R. S. Waples, D. Peel, G. M. Macbeth, B. J. Tillett & J. R. Ovenden, 2014. NeEstimator V2: re-implementation of software for the estimation of contemporary effective population size (Ne) from genetic data. Molecular Ecology Resources 14: 209–214.
Doenz, C. J., A. K. Krähenbüh, J. Walker, O. Seehausen & J. Brodersen, 2019. Ecological opportunity shapes a large Arctic charr species radiation. Proceedings of the Royal Society London B 286: 20191992.
Esin, E. V., E. S. Bocharova, N. S. Mugue & G. N. Markevich, 2017. Occurrence of sympatric charr groups, Salvelinus, Salmonidae, in the lakes of Kamchatka: a legacy of the last glaciations. Journal of Fish Biology 91: 628–644.
Esin, E. V., E. S. Bocharova, E. A. Borisova & G. N. Markevich, 2020. Interaction among morphological, trophic and genetic groups in the rapidly radiating Salvelinus fishes from Lake Kronotskoe. Evolutionary Ecology 34: 611–632.
Estoup, A., I. J. Wilson, C. Sullivan, J.-M. Cornuet & C. Moritz, 2001. Inferring population history from microsatellite and enzyme data in serially introduced cane toads, Bufo marinus. Genetics 159: 1671–1687.
Evanno, G., S. Regnaut & J. Goudet, 2005. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Molecular Ecology 14: 2611–2620.
Excoffier, L., P. Smouse & J. Quattro, 1992. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitochondrial DNA restriction data. Genetics 131: 479–491.
Excoffier, L., G. Laval & S. Schneider, 2005. Arlequin ver. 3.0: An integrated software package for population genetics data analysis. Evolutionary Bioinformatics Online 1: 47–50.
Fitzpatrick, B. M., J. A. Fordyce & S. Gavrilets, 2008. What, if anything, is sympatric speciation? Journal of Evolutionary Biology 21: 1452–1459.
Foote, A. D., 2018. Sympatric speciation in the genomic era. Trends in Ecology and Evolution 33: 85–95.
Garza, J. C. & E. G. Williamson, 2001. Detection of reduction in population size using data from microsatellite loci. Molecular Ecology 10: 305–318.
Gavrilets, S., 2003. Models of speciation: what have we learned in 40 years? Evolution 57: 2197–2215.
Gilbert, K. J., R. L. Andrew, D. G. Bock, et al., 2012. Recommendations for utilizing and reporting population genetic analyses: the reproducibility of genetic clustering using the program STRUCTURE. Molecular Ecology 21: 4925–4930.
Gillespie, R. G., G. M. Bennett, L. De Meester, et al., 2020. Comparing adaptive radiations across space, time, and taxa. Journal of Heredity 111: 1–20.
Givnish, T. J., 2015. Adaptive radiation versus “radiation” and “explosive diversification”: why conceptual distinctions are fundamental to understanding evolution. New Phytologist 207: 297–303.
Golberg, A. V., V. L. Gudina, V. D. Кryukov, V. V. Rogozshin, et al., 1971. The complex geological study of the quaternary sediments in the upper part of the Tokalanda valley (Norilsk Plateau). In Troitskiy, S. L. (ed), Marine Pleistocene of Siberian Lowlands Nauka, Moscow: 133–138.
Gordeeva, N. V., S. S. Alekseyev, A. F. Kirillov, A. I. Vokin & I. V. Samusenok, 2018. Distribution, composition and relationships of phylogenetic groups of arctic Charr Salvelinus alpinus (L.) (Salmoniformes, Salmonidae) in the European part of Russia and in Siberia as revealed by the analysis of nucleotide sequences of mitochondrial DNA. Journal of Ichthyology 58: 808–818.
Gordeeva, N. V., S. S. Alekseyev, M. Y. Pichugin, A. F. Kirillov & V. I. Romanov, 2021. New data about the distribution of three phylogenetic lineages of Arctic Charr Salvelinus alpinus (Salmonidae) in their contact zones in the north of East Siberia. Journal of Ichthyology 61: 701–708.
Gross, B. L. & L. H. Rieseberg, 2005. The ecological genetics of homoploid hybrid speciation. Journal of Heredity 96: 241–252.
Grosswald, M. G., 1999. Cataclysmic Megafloods in Eurasia and the Polar Ice Sheets, Scientific World, Moscow: (In Russian).
Hammer, Ø., D. A. T. Harper & P. D. Ryan, 2001. PAST: paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4: 1–9.
Hedrick, P. W., 2013. Adaptive introgression in animals: examples and comparison to new mutation and standing variation as sources of adaptive variation. Molecular Ecology 22: 4606–4618.
Hughes, A. L. C., R. Gyllencreutz, Ø. S. Lohne, J. Mangerud & J. I. Svendsen, 2016. The last Eurasian ice sheets–a chronological database and time-slice reconstruction, DATED-1. Boreas 45: 1–45.
Jacobs, A., M. Carruthers, A. Yurchenko, et al., 2020. Parallelism in eco-morphology and gene expression despite variable evolutionary and genomic backgrounds in a Holarctic fish. PLoS Genetics 16: e1008658.
Jakobsson, M. & N. A. Rosenberg, 2007. Clumpp: A cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23: 1801–1806.
Johnson, K. R., J. E. Wright & B. May, 1987. Linkage relationships reflecting ancestral tetraploidy in salmonid fish. Genetics 116: 579–591.
Jonsson, B. & N. Jonsson, 2001. Polymorphism and speciation in Arctic charr. Journal of Fish Biology 58: 605–638.
Kagawa, K. & G. Takimoto, 2018. Hybridization can promote adaptive radiation by means of transgressive segregation. Ecology Letters 21: 264–274.
Kalinowski, S. T., 2004. Counting alleles with rarefaction: private alleles and hierarchical sampling designs. Conservation Genetics 5: 539–543.
Kalinowski, S. T., 2005. HP-rare: a computer program for performing rarefaction on measures of allelic diversity. Molecular Ecology Notes 5: 187–189.
Kalinowski, S. T., 2011. The computer program STRUCTURE does not reliably identify the main genetic clusters within species: simulations and implications for human population structure. Heredity 106: 625–632.
Klemetsen, A., 2010. The charr problem revisited: Exceptional phenotypic plasticity promotes ecological speciation in postglacial lakes. Freshwater Reviews 3: 49–74.
Kopelman, N. M., J. Mayzel, M. Jakobsson, N. A. Rosenberg & I. Mayrose, 2015. Clumpak: a program for identifying clustering modes and packaging population structure inferences across K. Molecular Ecology Resources 15: 1179–1191.
Kryukov, V. D., 1975. On the origin and Quaternary history of deep Norilsk lakes in the western edge of the Putorana plateau. In History of lakes in the Pleistocene. Institut Ozerovedeniya, Leningrad 2: 146–150 (In Russian).
Kvasov, D. D., 1975. Late Quaternary History of Large Lakes and Inland Seas of Eastern Europe, Nauka, Leningrad: (In Russian).
Leigh, J. W. & D. Bryant, 2015. PopART: full-feature software for haplotype network construction. Methods in Ecology and Evolution 6: 1110–1116.
Libungan, L. A. & S. Pálsson, 2015. ShapeR: an R package to study otolith shape variation among fish populations. PLoS ONE 10: e0121102.
Lleonart, J., J. Salat & G. J. Torres, 2000. Removing allometric effects of body size in morphological analysis. Journal of Theoretical Biology 205: 85–93.
Logashev, M. V., 1940. Lake Melkoe and its fisheries exploitation. Trudy Nauchno-Issledovatel’skogo Instituta Polyarnogo Zemledeliya, Zhivotnovodstva i Promyshlennogo Khozyaistva 11: 7–71 (In Russian).
Losos, J. B., 2010. Adaptive radiation, ecological opportunity, and evolutionary determinism. The American Naturalist 175: 623–639.
Luikart, G. & J.-M. Cornuet, 1998. Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conservation Biology 12: 228–237.
Mallet, J., A. Meyer, P. Nosil & J. L. Feder, 2009. Space, sympatry and speciation. Journal of Evolutionary Biology 22: 2332–2341.
Maksimov, V. A., K. A. Savvaitova, B. M. Mednikov, et al., 1995. Mountain charr – a new form of Arctic charr (genus Salvelinus) from the water bodies of Taimyr. Voprosy Ikhtiologii 35: 296–301 (In Russian).
Makeyeva, A. P., 1992. Embryology of fishes, Moscow University Press, Moscow: (In Russian).
Meirmans, P. G., 2019. Subsampling reveals, that unbalanced sampling affects structure results in a multi-species dataset. Heredity 122: 276–287.
Moore, J.-S., R. Bajno, J. D. Reist & E. B. Taylor, 2015. Post-glacial recolonization of the North American Arctic by Arctic char (Salvelinus alpinus): Genetic evidence of multiple northern refugia and hybridization between glacial lineages. Journal of Biogeography 42: 2089–2100.
Morat, F., S. Betoulle, M. Robert, et al., 2008. What can otolith examination tell us about the level of perturbations of salmonid fish from the Kerguelen Islands? Ecology of Freshwater Fish 17: 617–627.
Nei, M. & L. Jin, 1989. Variances of the average numbers of nucleotide substitutions within and between populations. Molecular Biology and Evolution 6: 290–300.
Nei, M., F. Tajima & Y. Tateno, 1983. Accuracy of estimated phylogenetic trees from molecular data. Journal of Molecular Evolution 19: 153–170.
Osinov, A. G., 2002. Arctic charr Salvelinus alpinus from Transbaikalia and Taimyr: genetic differentiation and origin. Journal of Ichthyology 42: 141–151.
Osinov, A. G. & L. Bernatchez, 1996. Atlantic and Danubian phylogenetic groupings of brown trout Salmo trutta complex: genetic divergence, evolution, and conservation. Journal of Ichthyology 36: 723–746.
Osinov, A. G., S. D. Pavlov & V. A. Maksimov, 1996. Allozyme variation and genetic differentiation in populations of Arctic char Salvelinus alpinus (L). from the Baltic Sea-Taimyr part of the range. Russian Journal of Genetics 32: 474–485.
Osinov, A. G., A. L. Senchukova, N. S. Mugue, S. D. Pavlov & I. A. Chereshnev, 2015. Speciation and genetic divergence of three species of charrs from ancient Lake El’gygytgyn (Chukotka) and their phylogenetic relationships with other representatives of the genus Salvelinus. Biological Journal of the Linnean Society 116: 63–85.
Osinov, A. G., A. A. Volkov, S. S. Alekseyev, A. A. Sergeev, M. V. Oficerov & A. F. Kirillov, 2017. On the origin and phylogenetic position of Arctic charr (Salvelinus alpinus complex, Salmonidae) from Lake Cherechen’ (middle Kolyma River basin): controversial genetic data. Polar Biology 40: 777–786.
Osinov, A. G., D. A. Pavlov & A. A. Volkov, 2018. On the origin of the lacustrine charr Salvelinus alpinus complex from the Kolyma and Sea of Okhotsk basins. Journal of Ichthyology 58: 353–370.
Osinov, A. G., A. A. Volkov & N. S. Mugue, 2021. Charrs of the genus Salvelinus (Salmonidae): hybridization, phylogeny and evolution. Hydrobiologia 848: 705–726.
Østbye, K., M. H. Hassve, A.-M. Peris Tamayo & M. Hagenlund,| T. Vogler & K. Præbel, 2020. “And if you gaze long into an abyss, the abyss gazes also into thee”: four morphs of Arctic charr adapting to a depth gradient in Lake Tinnsjøen. Evolutionary Applications 13: 1240–1261.
Parisi-Baradad, V., A. Lombarte, E. García-Ladona, et al., 2005. Otolith shape contour analysis using affine transformation invariant wavelet transforms and curvature scale space representation. Marine and Freshwater Research. 56: 795–804.
Pavlov, D. S., K. A. Savvaitova, M. A. Gruzdeva, et al., 1999. Diversity of Fishes from Taimyr (Systematics, Ecology, Species Structure as the Base of Biodiversity in High Latitudes, and Present State in the Condition of Anthropogenic Pressure, Nauka, Moscow: (In Russian).
Pavlov, S. D., 1997. Sympatric forms of charrs (gen. Salvelinus) from Ayan Lake (Taimyr Peninsula). Journal of Ichthyology 37: 485–494.
Pavlov, S. D., K. A. Savvaitova & V. A. Maksimov, 1994. On the relationships of sympatric groups of Arctic charr in Lake Sobach’e (Norilo-Pyasinskaya water system). In Systematics, biology, and cultivation biotechnics of salmonid fishes. Gosudarstvennyi nauchno-issledovatel’skii institut ozernogo i rechnogo rybnogo khozyaistva, Sankt-Petersburg: 148–151 (In Russian).
Peery, M. Z., R. Kirby, B. N. Reid, et al., 2012. Reliability of genetic bottleneck tests for detecting recent population declines. Molecular Ecology 21: 3403–3418.
Pichugin, MYu. & Yu. V. Chebotareva, 2011. Patterns of development of the coldwater lacustrine-riverine form of the Drjagin charr (genus Salvelinus) from Lake Lama (the Taimyr Peninsula) during the larval period. Journal of Ichthyology 51: 248–262.
Piry, S., G. Luikart & J. M. Cornuet, 1999. BOTTLENECK: a computer program for detecting recent reductions in the effective population size using allele frequency data. Journal of Heredity 90: 502–503.
Popper, A. N. & S. Coombs, 1982. The morphology and evolution of the ear in Actinopterygian fishes. American Zoologist 22: 311–328.
Posada, D. & K. A. Crandall, 1998. Modeltest: testing the model of DNA substitution. Bioinformatics 14: 817–818.
Pritchard, J. K., M. Stephens & P. Donnelly, 2000. Inference of population structure from multilocus genotype data. Genetics 155: 945–959.
Pritchard, J. K., X. Wen & D. Falush, 2010. Documentation for Structure software: Version 2.3. http://pritch.bsd.uchicago.edu/structure.html.
Radchenko, O. A., 2005. Variability of Mitochondrial DNA in Chars of the Genus Salvelinus. NESC FEB RAS, Magadan: (In Russian).
Rice, W. R., 1989. Analyzing tables of statistical tests. Evolution 43: 223–225.
Richards, E. J., J. W. Poelstra & C. H. Martin, 2018. Don’t throw out the sympatric speciation with the crater lake water: fine-scale investigation of introgression provides equivocal support for causal role of secondary gene flow in one of the clearest examples of sympatric speciation. Evolution Letters 2–5: 524–540.
Robinson, B. W. & K. J. Parsons, 2002. Changing times, spaces, and faces: tests and implications of adaptive morphological plasticity in the fishes of northern postglacial lakes. Canadian Journal of Fisheries and Aquatic Sciences 59: 1819–1833.
Romanov, V. I., 1983. Ecological structure of charrs (genus Salvelinus) of Lake Khantaiskoe. Voprosy geografii Sibiri. Tomskii Gosudarstvennyi Universitet, Tomsk 14: 73–88 (In Russian).
Rosenberg, N. A., 2004. Distruct: a program for the graphical display of population structure. Molecular Ecology Notes 4: 137–138.
Rousset, F., 2008. Genepop’007: a complete reimplementation of the Genepop software for Windows and Linux. Molecular Ecology Resources 8: 103–106.
Salisbury, S. J., G. R. McCracken, D. Keefe, R. Perry & D. E. Ruzzante, 2019. Extensive secondary contact among three glacial lineages of Arctic Char (Salvelinus alpinus) in Labrador and Newfoundland. Ecology and Evolution 9: 2031–2045.
Sarana, V. A., 2017. Glaciation of the western part of the Putorana Plateau in the late Pleistocene and Holocene. Vestnik Moskovskogo universiteta. Seriya 5. Geografiya 1: 73–81 (In Russian).
Savvaitova, K. A., 1989. Arctic Charrs: Structure of Population Systems, Perspectives of Economic Use. Agropromizdat, Moscow: (In Russian).
Savvaitova, K. A., 1995. Patterns of diversity and processes of speciation in Arctic charr. Nordic Journal of Freshwater Research 71: 81–91.
Savvaitova, K. A., V. A. Maksimov & V. D. Nesterov, 1980. On the systematics and ecology of charrs of the genus Salvelinus (fam. Salmonidae) from water bodies of Taimyr Peninsula. Voprosy Ikhtiologii 20: 203–219 (In Russian).
Schluter, D., 2009. Evidence for Ecological Speciation and Its Alternative. Science 323: 737–741.
Schwarzhans, W., 2014. Head and otolith morphology of the genera Hymenocephalus, Hymenogadus and Spicomacrurus (Macrouridae), with the description of three new species. Zootaxa 3888: 1–73.
Secor, D. H., J. M. Dean & E. H. Laban, 1991. Manual for Otolith Removal and Preparation for Microstructural Examination, Electric Power Research Institute, Washington, DC:
Seehausen, O., 2004. Hybridization and adaptive radiation. Trends in Ecology and Evolution 19: 198–207.
Seehausen, O., 2013. Conditions when hybridization might predispose populations for adaptive radiation. Journal of Evolutionary Biology 26: 279–281.
Skulason, S., K. J. Parsons & R. Svanback, 2019. A way forward with eco evo devo: an extended theory of resource polymorphism with postglacial fishes as model systems. Biological Reviews 94: 1786–1808.
Smith, T. B. & S. Skulason, 1996. Evolutionary significance of resource polymorphisms in fishes, amphibians, and birds. Annual Review of Ecology and Systematics 27: 111–133.
Taylor, E. B., 2016. The Arctic char (Salvelinus alpinus) ‘“complex”’ in North America revisited. Hydrobiologia 783: 283–293.
Timireva, S. N., V. A. Sarana & Yu. M. Kononov, 2016. The glaciation of river Imandra valley (western part of Putorana plateau) in the late Pleistocene and Holocene. Geomorfologiya 1: 82–87.
Tuset, V. M., J. L. Otero-Ferrer, J. G. Gómez-Zurita, et al., 2016. Otolith shape lends support to the sensory drive hypothesis in rockfishes. Journal of Evolutionary Biology 29: 2083–2097.
Van Oosterhout, C., W. F. Hutchinson, D. P. Wills & P. Shipley, 2004. MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Molecular Ecology Notes 4: 535–538.
Vignon, M. & F. Morat, 2010. Environmental and genetic determinant of otolith shape revealed by a non-indigenous tropical fish. Marine Ecology Progress Series 411: 231–241.
Waples, R. S. & C. Do, 2008. LDNE: a program for estimating effective population size from data on linkage disequilibrium. Molecular Ecology Resources 8: 753–756.
Waples, R. S. & C. Do, 2010. Linkage disequilibrium estimates of contemporary Ne using highly variable genetic markers: a largely untapped resource for applied conservation and evolution. Evolutionary Applications 3: 244–262.
Williamson-Natesan, E. G., 2005. Comparison of methods for detecting bottlenecks from microsatellite loci. Conservation Genetics 6: 551–562.
Wilson, A. J., D. Gıslason, S. Skulason, et al., 2004. Population genetic structure of Arctic charr, Salvelinus alpinus from northwest Europe on large and spatial scales. Molecular Ecology 13: 1129–1142.
Zadelenov, V. A., L. A. Glushchenko, V. V. Matasov & E. N. Shadrin, 2015. Ichthyofauna of large Norilsk lakes (Kutaramakan, Lama, Sobach’e). Nauchnye trudy gosudarstvennogo budzhetnogo uchrezhdeniya “Ob’’edinennaya direktsiya zapovednikov Taimyra” 1: 116–130 (In Russian).
Zadelenov, V. A., E. N. Shadrin & V. V. Matasov, 2017. Charrs of Taimyr Peninsula (review). Rybovodstvo i Rybnoe Khozyaistvo 12: 19–28 (In Russian).
Acknowledgements
We thank F. S. Lobyrev, V. A. Maksimov, K. A. Savvaitova. M. Yu. Pichugin, V. A. Zadelenov, I. A. Chereshnev, S. S. Alekseyev and Clemens Ratschan for help in the collection of the material.
Funding
This study was supported by Russian Foundation for Basic Research (Project No. 17-04-00063).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Handling Editor: Michael Power
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is dedicated to the memory of our colleagues K. A. Savvaitova and V. A. Maksimov (Department of Ichthyology, Faculty of Biology, Moscow State University) who made an invaluable contribution to the investigation of Arctic charr in Taimyr and throughout Russia.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Osinov, A.G., Volkov, A.A. & Pavlov, D.A. Secondary contact, hybridization, and diversification in Arctic charr (Salvelinus alpinus (L.) species complex) from lakes of the Norilo-Pyasinskaya water system, Taimyr: how many forms exist there?. Hydrobiologia 849, 2521–2547 (2022). https://doi.org/10.1007/s10750-022-04869-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-04869-x