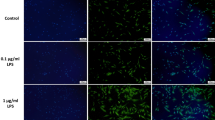
Abstract
Lipopolysaccharides (LPS), which are components of the cell wall of Gram-negative bacteria, are among the important factors that induce inflammation, including pulpitis. Autophagy in human dental pulp cells (hDPCs) acts as a protective mechanism that promotes cell survival under adverse conditions through different signaling pathways. In this study, we examined whether LPS increases autophagy in hDPCs and investigated the role of mitogen-activated protein kinases signaling and nuclear factor κB (NF-κB) in this process. We found that stimulation of hDPCs with 0.1 µg/mL LPS increased the protein and mRNA levels of autophagy markers, beclin1 and microtubule associated protein light chain 3II (LC3II). In addition, acridine orange staining and transmission electron microscopy demonstrated the induction of autophagy upon the treatment of LPS. Furthermore, LPS affected phosphorylation of p38, extracellular signal-regulated kinase (ERK), and c-Jun N-terminal kinase (JNK), and the nuclear translocation of NF-κB. While p38 inhibitor suppressed the LPS-induced increase in protein levels of beclin1 and LC3-II. Our results suggest that LPS induced autophagy in hDPCs and affected the phosphorylation of p38, ERK, and JNK, as well as the nuclear translocation of NF-κB. Phosphorylation of p38 may be involved in LPS-induced autophagy in hDPCs.




Similar content being viewed by others
References
Chen M, Liu J, Yang W, Ling W (2017) Lipopolysaccharide mediates hepatic stellate cell activation by regulating autophagy and retinoic acid signaling. Autophagy 13:1813–1827
Eskelinen EL, Reggiori F, Baba M, Kovacs AL, Seglen PO (2011) Seeing is believing: the impact of electron microscopy on autophagy research. Autophagy 7:935–956
Gao Y, You X, Liu Y, Gao F, Zhang Y, Yang J, Yang C (2020) Induction of autophagy protects human dental pulp cells from lipopolysaccharide-induced pyroptotic cell death. Exp Ther Med 19:2202–2210
Gong QM, Jiang HP, Wei XP, Ling JP, Wang JP (2010) Expression of erythropoietin and erythropoietin receptor in human dental pulp. J Endod 36:1972–1977
Han C, Ding Z, Shi H, Qian W, Hou X, Lin R (2016) The role of probiotics in lipopolysaccharide-induced Autophagy in intestinal epithelial cells. Cell Physiol Biochem 38:2464–2478
He Y, She H, Zhang T, Xu H, Cheng L, Yepes M, Zhao Y, Mao Z (2018) p38 MAPK inhibits autophagy and promotes microglial inflammatory responses by phosphorylating ULK1. J Cell Biol 217:315–328
Huang Y, Jiang H, Gong Q, Li X, Ling J (2015a) Lipopolysaccharide stimulation improves the odontoblastic differentiation of human dental pulp cells. Mol Med Rep 11:3547–3552
Jung CH, Ro S, Cao J, Otto NM, Kim D (2010) mTOR regulation of autophagy. Febs Lett 584:1287–1295
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T, Kominami E, Ohsumi Y, Yoshimori T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO JOURNAL 19:5720–5728
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132:27–42
Li L, Zhu YQ, Jiang L, Peng W (2012) Increased autophagic activity in senescent human dental pulp cells. Int Endod J 45:1074–1079
Li Y, Wang H, Pei F, Chen Z, Zhang L (2018) FoxO3a regulates inflammation-induced Autophagy in odontoblasts. J Endod 44:786–791
Liu J, Wang X, Zheng M, Luan Q (2018) Lipopolysaccharide from Porphyromonas gingivalis promotes autophagy of human gingival fibroblasts through the PI3K/Akt/mTOR signaling pathway. Life Sci 211:133–139
Marino G, Lopez-Otin C (2004) Autophagy: molecular mechanisms, physiological functions and relevance in human pathology. Cell Mol Life Sci 61:1439–1454
Meng N, Zhao J, Su L, Zhao B, Zhang Y, Zhang S, Miao J (2012a) A butyrolactone derivative suppressed lipopolysaccharide-induced autophagic injury through inhibiting the autoregulatory loop of p8 and p53 in vascular endothelial cells. Int J Biochem Cell Biol 44:311–319
Meng N, Zhao J, Su L, Zhao B, Zhang Y, Zhang S, Miao J (2012b) A butyrolactone derivative suppressed lipopolysaccharide-induced autophagic injury through inhibiting the autoregulatory loop of p8 and p53 in vascular endothelial cells. Int J Biochem Cell Biol 44:311–319
Mizushima N (2005) The pleiotropic role of autophagy: from protein metabolism to bactericide. Cell Death Differ 12(Suppl 2):1535–1541
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451:1069–1075
Park SY, Sun EG, Lee Y, Kim MS, Kim JH, Kim WJ, Jung JY (2018) Autophagy induction plays a protective role against hypoxic stress in human dental pulp cells. J Cell Biochem 119:1992–2002
Sul OJ, Park HJ, Son HJ, Choi HS (2017) Lipopolysaccharide (LPS)-induced Autophagy is responsible for enhanced osteoclastogenesis. Mol Cells 40:880–887
Takanche JS, Kim J, Kim J, Han S, Yi H (2018) Schisandrin C enhances odontoblastic differentiation through autophagy and mitochondrial biogenesis in human dental pulp cells. Arch Oral Biol 88:60–66
Trocoli A, Djavaheri-Mergny M (2011) The complex interplay between autophagy and NF-κB signaling pathways in cancer cells. Am J Cancer Res 1:629
Wang J, Wei X, Ling J, Huang Y, Gong Q (2010) Side population increase after simulated transient ischemia in human dental pulp cell. J Endod 36:453–458
Wang J, Feng X, Zeng Y, Fan J, Wu J, Li Z, Liu X, Huang R, Huang F, Yu X, Yang X (2013) Lipopolysaccharide (LPS)-induced autophagy is involved in the restriction of Escherichia coli in peritoneal mesothelial cells. BMC Microbiol 13:255
Wang X, Wu TT, Jiang L, Rong D, Zhu YQ (2017) Deferoxamine-induced migration and odontoblast differentiation via ROS-dependent Autophagy in dental pulp stem cells. Cell Physiol Biochem 43:2535–2547
Yang JW, Zhang YF, Wan CY, Sun ZY, Nie S, Jian SJ, Zhang L, Song GT, Chen Z (2015) Autophagy in SDF-1alpha-mediated DPSC migration and pulp regeneration. Biomaterials 44:11–23
Yoshioka A, Miyata H, Doki Y, Yamasaki M, Sohma I, Gotoh K, Takiguchi S, Fujiwara Y, Uchiyama Y, Monden M (2008) LC3, an autophagosome marker, is highly expressed in gastrointestinal cancers. Int J Oncol 33:461
Yue Z, Jin S, Yang C, Levine AJ, Heintz N (2003) Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci U S A 100:15077–15082
Zhou QD, Liu HD, Sun QP, Zhang LP, Lin HP, Yuan GPD, Zhang LDP, Chen ZPD (2013) Adenosine monophosphate–activated protein kinase/mammalian target of rapamycin–dependent Autophagy protects human dental pulp cells against hypoxia. J Endod 39:768–773
Acknowledgements
This study was financially supported by the National Nature Science Foundation of China (No. 81700957, No. 81870750), the Natural Science Foundation of Guangdong province (No. 2016A030310197). The funding bodies had no role in the design, collection, analysis, interpretation of data, or writing of manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10735_2021_10004_MOESM1_ESM.tif
Supplementary material 1 Effectsof LPS and p38 inhibitor on autophagic vacuoles in hDPCs. (A) Controlgroup shows fewer autolysosomes (×30000). (B) hDPCs stimulated with 0.1 μg/mLLPS for 12 hours contain more autolysosomes in the cytoplasm (×30000). (C)hDPCs pretreated with SB203580 before stimulation with 0.1 μg/mL LPS for 12hours contain fewer autolysosomes (×30000). Autophagic vacuoles aremarked by red arrows (tif 6099.9 kb)
Rights and permissions
About this article
Cite this article
Huang, Y., Li, X., Liu, Y. et al. LPS-induced autophagy in human dental pulp cells is associated with p38. J Mol Histol 52, 919–928 (2021). https://doi.org/10.1007/s10735-021-10004-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-021-10004-2