Abstract
Li-Fraumeni Syndrome (LFS), caused by germline pathogenic variants in TP53, predisposes to a wide range of young-onset malignancies, particularly sarcoma, breast and brain cancer. More recently, an increased risk of gastric adenocarcinoma has been recognised, although uptake of surveillance upper endoscopy is unclear. Our retrospective review of 65 patients with LFS, of whom 53.8% had undergone endoscopy, identified four patients (6.2%) with gastro-esophageal junction (GEJ) adenocarcinomas. Two cases were found on asymptomatic screening and were early stage. No cases had family history of gastrointestinal malignancy. Reviewing genomic data from The Cancer Genome Atlas Program, 76.4% of sporadic esophageal adenocarcinomas harboured somatic TP53 pathogenic variants, compared with 39.9% of non-cardia gastric cancers. This similar pattern observed in germline and sporadic cases warrants further investigation. We propose that upper endoscopy be recommended to all patients with LFS, with a focus on appropriate surveillance of the GEJ.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Li-Fraumeni Syndrome (LFS) is defined by germline pathogenic variants in the tumour suppressor gene TP53 and is associated with increased rates of many different malignancies, some with up to 100% lifetime risk without appropriate intervention [1]. While the strongest associations have been demonstrated with sarcoma, breast cancer, brain tumours and adrenocortical carcinomas, the increased risk of colorectal and, more recently, gastric adenocarcinomas have also been acknowledged [2]. The relative risk of esophageal cancer is less well characterised with conflicting reports [1, 3]. An analysis of the International Agency for Research on Cancer database identified 0.5% of individuals with esophageal cancer (15/3043) and 3.3% (101/3043) with gastric cancer [3, 4]. Gastro-esophageal junction (GEJ) tumours arise at the histological transition between esophagus and stomach, and the commonly used Siewert classification divides GEJ adenocarcinomas into three categories for which oncological management differs: Type 1 arising from distal esophagus (1 to 5 cm proximal to GEJ), Type 2 located at the true junction (between 1 cm proximal to 2 cm distal of the GEJ) and Type 3 located 2 to 5 cm distal to the GEJ [5]. In contrast, the American Joint Committee on Cancer stages all GEJ tumors with epicentre ≤ 2 cm into the proximal stomach as esophageal cancers and those > 2 cm as gastric cancers [5]. Consequently, GEJ tumors have been variably categorised in the literature as esophageal, gastric or a separate entity.
In this context, expert consensus guidelines differ in their recommendations for upper endoscopy, or esophagogastroduodenoscopy, screening in LFS: North American guidelines support upper endoscopy every 2–5 years from the age of 25 alongside colonoscopy, while European guidelines do not routinely recommend upper endoscopy screening [2, 6]. The uptake of these recommendation is also unclear.
There is a biological basis to suspect pathogenic variants in TP53 as a driver of upper gastrointestinal cancers. The Cancer Genome Atlas Program classified 559 sporadic esophageal and gastric cancers into distinct molecular phenotypes. When analysing adenocarcinoma, four subclasses were identified, but only one, a chromosomal instability (CIN) subclass, accounted for the vast majority of lower esophagus and GEJ tumours [7]. CIN molecular subgroup accounted for 49% of adenocarcinomas from esophagus and stomach and was characterised by ERBB2 amplification, VEGFA amplification and, importantly, pathogenic variants in TP53.
The familial cancer centre at our institution offers a dedicated gastrointestinal risk management clinic for patients with high-risk genetic predisposition to gastrointestinal malignancy, and we routinely offer upper endoscopy at the same time as colonoscopy to patients with LFS. We sought to assess the rates of upper gastrointestinal cancers and their characteristics in our cohort, with comparison to results from the TCGA cohort.
Methods
A retrospective chart review was performed of adult patients with clinical class 4 or 5 germline pathogenic variants in TP53 managed by our centre between January 2000 to May 2023. Data regarding demographics, personal and family history of malignancy, duration of follow-up, and endoscopic/histologic results of upper and lower endoscopies were collected. Further clinical details were collated for cases of GEJ tumours. Ethical approval was obtained from the institutional human ethics committee (Project number: PMC97246, 29 May 2023).
Separately, data of each subject analysed in the TCGA study to characterise the molecular characteristics of sporadic esophageal and gastric cancers was obtained from the supplementary table in the initial publication. Corresponding details of pathogenic variants in TP53 were then obtained from the openly available dataset on cBioportal (accessed 27 June 2023) [7, 8]. From 559 subjects, 90 esophageal squamous cell cancers, 13 gastric adenocarcinomas of unclear location and 2 undifferentiated esophageal tumours were excluded. The remaining 454 cases of adenocarcinoma were then subdivided based on subtype (esophageal/probable esophageal GEJ adenocarcinoma, indeterminate GEJ adenocarcinoma, gastric/probable gastric GEJ adenocarcinoma) and proportion of cases with somatic pathogenic variants in TP53 calculated.
Results
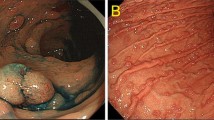
Sixty-five with LFS (57% female, mean at diagnosis 34.5, median follow-up 51 months) seen via the high-risk cancer genetics clinic over the study period, 35 (53.8%) of whom had at least one upper endoscopy. Four patients (6.2%) were diagnosed with cancer at the GEJ (Table 1), with no cancers elsewhere in the upper digestive tract and one separate case of colorectal cancer on colonoscopy. Two cases, a White man and North-East Asian woman in their 30s, were asymptomatic and undergoing screening, at their second and first procedures respectively. Both cancers were at early stage allowing for resection with curative intent. The lesions were subtle and best appreciated on close inspection of the GEJ in forward-view (Fig. 1). In the former case, index upper endoscopy revealed moderate reflux esophagitis (Los Angeles Classification grade B) with no dysplasia on biopsy, but a decision was made for early repeat surveillance at 18 months revealing subsequent carcinoma in situ. No cases of cancer were associated with prior family history of digestive tract malignancy, or abdominal radiotherapy. Furthermore, five patients undergoing screening (14.3% of those who had upper endoscopy) were diagnosed with a premalignant lesion (Barrett’s esophagus, non-Helicobacter associated gastric intestinal metaplasia and a proximal fundic gland polyp with low grade dysplasia).
On review of the data for these patients from the TCGA paper via cBioportal, we found that amongst esophageal or probable esophageal adenocarcinomas, 76.4% (55/72) had somatic pathogenic variants in TP53, with decreasing prevalence in the indeterminate GEJ (22/36, 61.1%), gastric/probable gastric GEJ (29/63, 46.0%) and gastric non-cardia (113/283, 39.9%) adenocarcinoma groups (Fig. 2).
Panel of endoscopic, surgical and histologic findings in one case of GEJ adenocarcinoma on screening: Top, left: a subtle GEJ nodule at endoscopy (blue arrow); Top, right: endoscopic biopsy (H&E stain, x40) demonstrating adenocarcinoma adjacent to squamous mucosa; Bottom, left: surgical resection specimen with irregularity at GEJ (yellow arrow); Bottom, right: resection histology (H&E stain, x40) demonstrating adenocarcinoma confined to the mucosa, at the GEJ
Discussion
It is still not globally recognised that upper endoscopy should be a routine component of surveillance in LFS. Moreover, in most guidelines recommending upper endoscopy, the focus has been on gastric adenocarcinoma, perhaps because some reports have presumably grouped GEJ cancers with gastric cancers [9], even though some cases are better characterised as esophageal or a distinct entity [5, 7]. The distinction is important as inspection of the gastric mucosa is performed within the stomach and on retroflexion during upper endoscopy, while the GEJ is best assessed in forward-view.
Our findings suggest that the GEJ and lower esophagus may be a particularly vulnerable region in LFS, in keeping with the molecular phenotyping seen in sporadic GEJ tumours. While it has been suggested that Asian carriers may be at higher risk than non-Asian carriers, and that risk of both gastric and colon cancers may run in families [9, 10], the risk in our cohort was not isolated to these specific groups. Both cases seen on surveillance were early stage, facilitating likely curative resection. This data suggests a harmonisation of germline and somatic observations that are highly suggestive of a biological effect of p53 involving the GEJ. There are limitations to the data given this is a retrospective analysis of a cohort of LFS, but the high prevalence of GEJ cancer in this group is compelling and warrants a prospective evaluation in patients who carry germline pathogenic variants of TP53.
Additionally, premalignant lesions in the upper gastrointestinal tract appeared relatively frequently in our cohort. Given the presence of gastric intestinal metaplasia in four of the five individuals with premalignant lesions, we would also endorse routine biopsy of the gastric antrum and body and eradication of Helicobacter pylori if present, similar to recommendations for Lynch Syndrome [11].
The optimal interval for upper endoscopy also requires further study. The progression from reflux esophagitis to carcinoma in situ occurred in 18 months in one case, and a longer surveillance interval may have led to a later stage diagnosis. Notably, this finding would not have been routinely followed-up in a patient with no germline pathogenic variant in TP53. It may therefore be reasonable for any endoscopic finding at index procedure to be followed-up at two years, and subsequent follow-up guided by the progression or resolution of the changes. Our practice, in the absence of data, is for patients with other risk factors such as family history of gastrointestinal malignancy or abdominal radiotherapy to undergo two to three yearly surveillance, generally coinciding with colonoscopy. Patients with normal index upper endoscopy and no other risk factors undergo surveillance every five years.
We believe our novel clinical observations provide practice-changing evidence for all clinicians caring for patients with germline pathogenic variants of TP53 and we would advocate for routine endoscopic surveillance in all cases, with particular focus on the GEJ.
Data availability
The retrospective clinical dataset generated during and/or analysed during the current study is not publicly available due privacy requirements but relevant data is available from the corresponding author on reasonable request.
References
McBride KA, Ballinger ML, Killick E, Kirk J, Tattersall MHN, Eeles RA et al (2014) Li-Fraumeni syndrome: cancer risk assessment and clinical management. Nat Rev Clin Oncol 11(5):260–271. https://doi.org/10.1038/nrclinonc.2014.41
Kratz CP, Achatz MI, Brugières L, Frebourg T, Garber JE, Greer MC et al (2017) Cancer Screening recommendations for individuals with Li-Fraumeni Syndrome. Clin Cancer Res 23(11):e38–e45. https://doi.org/10.1158/1078-0432.Ccr-17-0408
Katona BW, Powers J, McKenna DB, Long JM, Le AN, Hausler R et al (2020) Upper gastrointestinal cancer risk and surveillance outcomes in Li-Fraumeni Syndrome. Am J Gastroenterol 115(12):2095–2097. https://doi.org/10.14309/ajg.0000000000000935
Bouaoun L, Sonkin D, Ardin M, Hollstein M, Byrnes G, Zavadil J et al (2016) TP53 variations in human cancers: new lessons from the IARC TP53 database and Genomics Data. Hum Mutat 37(9):865–876. https://doi.org/10.1002/humu.23035
Chevallay M, Bollschweiler E, Chandramohan SM, Schmidt T, Koch O, Demanzoni G et al (2018) Cancer of the gastroesophageal junction: a diagnosis, classification, and management review. Ann N Y Acad Sci 1434(1):132–138. https://doi.org/10.1111/nyas.13954
Frebourg T, Bajalica Lagercrantz S, Oliveira C, Magenheim R, Evans DG (2020) Guidelines for the Li-Fraumeni and heritable TP53-related cancer syndromes. Eur J Hum Genet 28(10):1379–1386. https://doi.org/10.1038/s41431-020-0638-4
Kim J, Bowlby R, Mungall AJ, Robertson AG, Odze RD, Cherniack AD et al (2017) Integrated genomic characterization of oesophageal carcinoma. Nature 541(7636):169–175. https://doi.org/10.1038/nature20805
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2(5):401–404. https://doi.org/10.1158/2159-8290.Cd-12-0095
Masciari S, Dewanwala A, Stoffel EM, Lauwers GY, Zheng H, Achatz MI et al (2011) Gastric cancer in individuals with Li-Fraumeni syndrome. Genet Med 13(7):651–657. https://doi.org/10.1097/GIM.0b013e31821628b6
Ariffin H, Chan ASL, Oh L, Abd-Ghafar S, Ong GB, Mohamed M et al (2015) Frequent occurrence of gastric cancer in Asian kindreds with Li–Fraumeni syndrome. Clin Genet 88(5):450–455. https://doi.org/10.1111/cge.12525
Weiss JM, Gupta S, Burke CA, Axell L, Chen LM, Chung DC et al (2021) NCCN Guidelines® insights: Genetic/Familial High-Risk Assessment: Colorectal, Version 1.2021. J Natl Compr Canc Netw 19(10):1122–1132. https://doi.org/10.1164/jnccn.2021.0048
Acknowledgements
Dr Alan Pham provided the resection specimen photograph and images from histology provided in Figure 1. Chris Michael assisted with database searching and curation.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was supported by NHMRC Postgraduate Scholarship (Grant Number: GNT2022274).
Author information
Authors and Affiliations
Contributions
Conceptualisation, methodology, analysis and investigation: DT and AB. Writing—original draft preparation: DT. Writing—review and editing: AB.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of the Peter MacCallum Cancer Centre (Project number: PMC97246, 29 May 2023).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tjandra, D., Boussioutas, A. Li Fraumeni Syndrome predisposes to gastro-esophageal junction tumours. Familial Cancer 23, 29–33 (2024). https://doi.org/10.1007/s10689-023-00353-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-023-00353-0