Abstract
Background
Colitis-associated cancer (CAC) is the serious complication of ulcerative colitis (UC), and molecular markers to evaluate the individual risk are required. MicroRNA-124a (miR - 124a) is known to have tumor-suppressive function and be methylation-silenced during exposure to chronic inflammation.
Aim
We analyzed whether higher methylation levels of miR-124a genes correlated with the higher epidemiologic risk of CAC development in UC patients.
Methods
Forty UC patients without CAC, four patients with CAC or dysplasia, eight sporadic colorectal cancer (S-CRC) patients, and 12 healthy volunteers (HV) were studied. Methylation status of miR-124a genes (miR-124a-1, -2, and -3) was analyzed by methylation-specific polymerase chain reaction (MSP), and methylation levels were quantified by real-time MSP. Expression of cyclin-dependent kinase 6 (CDK6), a target of miR-124a, was analyzed by immunohistochemistry.
Results
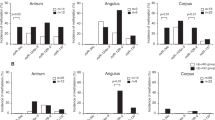
Three miR-124a genes were methylated in all neoplastic tissues (CAC, dysplasia, and S-CRC), and CDK6 was highly expressed in those tissues. Regarding disease extent, mean methylation levels of miR-124a-3 in HV, non-pancolitis, and pancolitis were 2.0, 5.3, and 12.3 %, respectively, and were significantly higher in pancolitis than in HV (p < 0.01). Regarding disease duration, mean methylation levels in short-term and long-standing UC patients were 2.5 and 13.2 %, respectively. Long-standing UC patients had significantly higher methylation levels than HV (p < 0.01). Moreover, UC patients with both pancolitis and long-standing had 7.4-fold higher methylation levels than those without these risk factors.
Conclusions
MiR-124a genes are methylated during carcinogenesis in UC patients. The methylation level of miR-124a-3 is a promising marker for estimating individual risk for CAC.



Similar content being viewed by others
Abbreviations
- UC:
-
Ulcerative colitis
- CAC:
-
Colitis-associated cancer
- ER:
-
Estrogen receptor
- miRNA:
-
MicroRNAs
- CDK6:
-
Cyclin-dependent kinase 6
- S-CRC:
-
Sporadic colorectal cancer
- MSP:
-
Methylation-specific polymerase chain reaction
- PCR:
-
Polymerase chain reaction
References
Ekbom A, Helmick C, Zack M, Adami HO. Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med. 1990;323:1228–1233.
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–535.
Itzkowitz SH, Present DH. Consensus conference: colorectal cancer screening and surveillance in inflammatory bowel disease. Inflamm Bowel Dis. 2005;11:314–321.
Winawer S, Fletcher R, Rex D, Bond J, et al. Colorectal cancer screening and surveillance: clinical guidelines and rationale-update based on new evidence. Gastroenterology. 2003;124:544–560.
Efthymiou M, Taylor AC, Kamm MA. Cancer surveillance strategies in ulcerative colitis: the need for modernization. Inflamm Bowel Dis. 2011;17:1800–1813.
Farraye FA, Odze RD, Eaden J, Itzkowitz SH. AGA technical review on the diagnosis and management of colorectal neoplasia in inflammatory bowel disease. Gastroenterology. 2010;138:746–774, 774 e1–e4; quiz e12–e3.
Jones PA, Baylin SB. The epigenomics of cancer. Cell. 2007;128:683–692.
Chiba T, Marusawa H, Ushijima T. Inflammation-associated cancer development in digestive organs: mechanisms and roles for genetic and epigenetic modulation. Gastroenterology. 2012;143:550–563.
Azarschab P, Porschen R, Gregor M, Blin N, Holzmann K. Epigenetic control of the E-cadherin gene (CDH1) by CpG methylation in colectomy samples of patients with ulcerative colitis. Genes Chromosom Cancer. 2002;35:121–126.
Fleisher AS, Esteller M, Harpaz N, Leytin A, et al. Microsatellite instability in inflammatory bowel disease-associated neoplastic lesions is associated with hypermethylation and diminished expression of the DNA mismatch repair gene, hMLH1. Cancer Res. 2000;60:4864–4868.
Garrity-Park MM, Loftus EV Jr, Sandborn WJ, Bryant SC, Smyrk TC. Methylation status of genes in non-neoplastic mucosa from patients with ulcerative colitis-associated colorectal cancer. Am J Gastroenterol. 2010;105:1610–1619.
Goel A, Richard Boland C. Epigenetics of colorectal cancer. Gastroenterology. 2012;143:1442–1460.
Moriyama T, Matsumoto T, Nakamura S, Jo Y, et al. Hypermethylation of p14 (ARF) may be predictive of colitic cancer in patients with ulcerative colitis. Dis Colon Rectum. 2007;50:1384–1392.
Sato F, Harpaz N, Shibata D, Xu Y, et al. Hypermethylation of the p14(ARF) gene in ulcerative colitis-associated colorectal carcinogenesis. Cancer Res. 2002;62:1148–1151.
Wheeler JM, Kim HC, Efstathiou JA, Ilyas M, Mortensen NJ, Bodmer WF. Hypermethylation of the promoter region of the E-cadherin gene (CDH1) in sporadic and ulcerative colitis associated colorectal cancer. Gut. 2001;48:367–371.
Arai E, Ushijima S, Fujimoto H, Hosoda F, et al. Genome-wide DNA methylation profiles in both precancerous conditions and clear cell renal cell carcinomas are correlated with malignant potential and patient outcome. Carcinogenesis. 2009;30:214–221.
He L, Hannon GJ. MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5:522–531.
Lujambio A, Ropero S, Ballestar E, Fraga MF, et al. Genetic unmasking of an epigenetically silenced microRNA in human cancer cells. Cancer Res. 2007;67:1424–1429.
Furuta M, Kozaki KI, Tanaka S, Arii S, Imoto I, Inazawa J. miR-124 and miR-203 are epigenetically silenced tumor-suppressive microRNAs in hepatocellular carcinoma. Carcinogenesis. 2010;31:766–776.
Agirre X, Vilas-Zornoza A, Jimenez-Velasco A, Martin-Subero JI, et al. Epigenetic silencing of the tumor suppressor microRNA Hsa-miR-124a regulates CDK6 expression and confers a poor prognosis in acute lymphoblastic leukemia. Cancer Res. 2009;69:4443–4453.
Ando T, Yoshida T, Enomoto S, Asada K, et al. DNA methylation of microRNA genes in gastric mucosae of gastric cancer patients: its possible involvement in the formation of epigenetic field defect. Int J Cancer. 2009;124:2367–2374.
Kaneda A, Kaminishi M, Sugimura T, Ushijima T. Decreased expression of the seven ARP2/3 complex genes in human gastric cancers. Cancer Lett. 2004;212:203–210.
Enomoto S, Maekita T, Tsukamoto T, Nakajima T, et al. Lack of association between CpG island methylator phenotype in human gastric cancers and methylation in their background non-cancerous gastric mucosae. Cancer Sci. 2007;98:1853–1861.
Maekita T, Nakazawa K, Mihara M, Nakajima T, et al. High levels of aberrant DNA methylation in Helicobacter pylori-infected gastric mucosae and its possible association with gastric cancer risk. Clin Cancer Res. 2006;12:989–995.
Nakajima T, Maekita T, Oda I, Gotoda T, et al. Higher methylation levels in gastric mucosae significantly correlate with higher risk of gastric cancers. Cancer Epidemiol Biomark Prev. 2006;15:2317–2321.
Nagashio R, Arai E, Ojima H, Kosuge T, Kondo Y, Kanai Y. Carcinogenetic risk estimation based on quantification of DNA methylation levels in liver tissue at the precancerous stage. Int J Cancer. 2011;129:1170–1179.
Fowler A, Thomson D, Giles K, Maleki S, et al. miR-124a is frequently down-regulated in glioblastoma and is involved in migration and invasion. Eur J Cancer. 2011;47:953–963.
Hackanson B, Bennett KL, Brena RM, Jiang J, et al. Epigenetic modification of CCAAT/enhancer binding protein alpha expression in acute myeloid leukemia. Cancer Res. 2008;68:3142–3151.
Pierson J, Hostager B, Fan R, Vibhakar R. Regulation of cyclin dependent kinase 6 by microRNA 124 in medulloblastoma. J Neurooncol. 2008;90:1–7.
Wilting SM, van Boerdonk RA, Henken FE, Meijer CJ, et al. Methylation-mediated silencing and tumour suppressive function of hsa-miR-124 in cervical cancer. Mol Cancer. 2010;9:167.
Ushijima T, Okochi-Takada E. Aberrant methylations in cancer cells: where do they come from? Cancer Sci. 2005;96:206–211.
Katsurano M, Niwa T, Yasui Y, Shigematsu Y, et al. Early-stage formation of an epigenetic field defect in a mouse colitis model, and non-essential roles of T- and B-cells in DNA methylation induction. Oncogene. 2012;31:342–351.
Foran E, Garrity-Park MM, Mureau C, Newell J, et al. Upregulation of DNA methyltransferase-mediated gene silencing, anchorage-independent growth, and migration of colon cancer cells by interleukin-6. Mol Cancer Res. 2010;8:471–481.
Koukos G, Polytarchou C, Kaplan JL, Morley-Fletcher A, et al. MicroRNA-124 regulates STAT3 expression and is down-regulated in colon tissues of pediatric patients with ulcerative colitis. Gastroenterology. 2013;145:842–852.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ueda, Y., Ando, T., Nanjo, S. et al. DNA Methylation of MicroRNA-124a Is a Potential Risk Marker of Colitis-Associated Cancer in Patients with Ulcerative Colitis. Dig Dis Sci 59, 2444–2451 (2014). https://doi.org/10.1007/s10620-014-3193-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3193-4