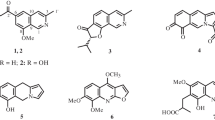
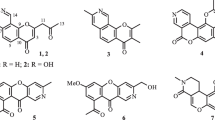
With the aim of screening more antiviral activity metabolites, in this study, two new (1 and 2), together with five known (3–7), isoquinolines were isolated from the cigar tobacco-derived endophytic fungi Aspergillus fumigatus. Their structures were determined by means of HR-ESI-MS and extensive 1D and 2D NMR spectroscopic studies. Interestingly, the anti-TMV activities test revealed that compounds 1 and 2 showed potential anti-TMV activities with inhibition rates of 31.2 and 28.5%, respectively.

Similar content being viewed by others

References
A. Elnahal, M. T. El-Saadony, A. M. Saad, E. M. Desoky, A. M. El-Ta han, M. M. Rady, S. F. Abu Qamar, and K. A. El-Tarabily, Eur. J. Plant. Pathol., 162 (4), 759 (2022).
K. H. Chen and N. Jessica, J. Exp. Bot., 73 (13), 4496 (2022).
L. O. Omoarelojie and J. V. Staden, S. Afr. J. Bot., 134, 280 (2020).
A. Hagag, M. F. Abdelwahab, A. El-Kader, and M. A. Fouad, J. Appl. Microbiol., 132 (6), 4150 (2022).
K. L. Yang, J. Tian, and N. P. Keller, Environ. Microbiol., 24 (7), 2857 (2022).
G. Y. Yang, J. M. Dai, Q. L. Mi, Z. J. Li, X. M. Li, J. D. Zhang, J. Wang, Y. K. Li, W. G. Wang, M. Zhou, and Q. F. Hu, Phytochemistry, 198, 113137 (2022).
J. M. Dai, Q. L. Mi. X. M. Li, D. Gang, G. Y. Yang, J. D. Zhang, J. Wang, Y. K. Li, H. Y. Yang, M. Dong, Z. J. Li, and Q. F. Hu, Phytochemistry, 205, 113485 (2023).
Y. N. Zhu, M. X. Liu, B. B. Cai, Y. Li, M. F. Li, H. S. Wang, M. Zhou, G. Y. Yang, Q. F. Hu, and Y. K. Li, Chem. Nat. Compd., 58, 712 (2022).
M. F. Li, D. Xiao, L. C. Zhu, L. Liu, J. N. Zheng, X. J. Gu, Y. N. Zhu, J. Xie, X. Wang, J. M. Dai, Q. L. Mi, Y. K. Yang, Q. F. Hu, Y. K. Li, and J. Q. Shi, Chem. Nat. Compd., 58, 1093 (2022).
W. G. Wang, L. Q. Du, S. L. Sheng, A. Li, Y. P. Li, G. G. Cheng, G. P. Li, G. L. Sun, Q. F. Hu, and Y. Matsuda, Org. Chem. Front., 6, 571 (2019).
L. Yuan, W. Z. Huang, K. Zhou, Y. D. Wang, W. Dong, G. Du, X. M. Gao, Y. H. Ma, and Q. F. Hu, Nat. Prod. Res., 29, 1914 (2015).
M. Zhou, G. Du, H. Y. Yang, C. F. Xia, J. X. Yang, Y. Q. Ye, X. M. Gao, X. N. Li, and Q. F. Hu, Planta Med., 81, 235 (2015).
M. Zhou, K. Zhou, P. He, K. M. Wang, R. Z. Zhu, Y. D. Wang, W. Dong, G. P. Li, H. Y. Yang, Y. Q. Ye, G. Du, X. M. Li, and Q. F. Hu, Planta Med., 82, 414 (2016).
M. Zhou, J. Lou, Y. K. Li, Y. D. Wang, K. Zhou, B. K. Ji, W. Dong, X. M. Gao, G. Du, and Q. F. Hu, Arch. Pharm. Res., 40, 32 (2017).
J. M. Dai, L. C. Zhu, D. Xiao, J. Xie, X. Wang, Q. L. Mi, J. Q. Shi, G. Y. Yin, Y. K. Yang, G. Y. Yang, Q. F. Hu, and W. Kai, Chem. Nat. Compd., 58, 1005 (2022).
B. Debnath, W. S. Singh, M. Das, S. Goswami, M. K. Singh, D. Maiti, and K. Manna, Mater. Today Chem., 9, 56 (2018).
M. Mohammad-Taghi, K. Ali Karimi, and L. Zahra, Toxin. Rev., 37 (1), 11 (2018).
Q. Li, C. M. Chen, Y. He, M. S. Wei, L. Cheng, X. Kang, J. P. Wang, X. C. Hao, H. C. Zhu, and Y. H Zhang, Phytochemistry, 169, 112177 (2020).
L. P. Chi, X. M. Li, Y. P. Wan, Y. H. Li, X. Li, and B. G. Wang, Chem. Biodiv., 18, e2100512 (2021).
C. M. Liu, F. H. Yao, X. H. Lu, X. X. Zhang, L. X. Luo, X. Liang, and S. H. Qi, Mar. Drugs., 20, 78 (2022).
Y. G. Sun, H. B. Liu, H. J. Gao, B. S. Lin, X. H. Xiang, H. K. Lu, Y. H. Wu, and X. H. Ma, Chin. Tob. Sci., 40 (3), 91 (2019).
C. Nord, J. J. Levenfors, J. Bjerketorp, C. Sahlberg, B. Guss, B. Ober, and A. Broberg, Molecules, 24, 4616 (2019).
J. Kohno, H. Hiramatsu, M. Nishio, M. Sakurai, T. Okuda, and S. Komatsubara, Tetrahedron, 55 (37), 11247 (1999).
F. Zhu, G. Y. Chen, J. S. Wu, and J. H. Pan, Nat. Prod. Res., 27 (21), 1960 (2013).
M. El-Aasr, D. Eliwa, M. Albadry, A. S. Ibrahim, A. Kabbash, K. M. Meepagala, I. A. Khan, S. I. Khan, and S. A. Ross, Phytochemistry, 189, 112828 (2021).
D. N. Quang, J. Schmidt, A. Porzel, L. Wessjohann, M. Haid, and N. Arnold, Nat. Prod. Commun., 5 (6), 869 (2010).
Q. F. Hu, L. M. Liao, H. T. Huang, Y. Xu, J. Wang, W. S. Kong, Q. L. Mi, M. Zhou, G. Y. Yang, and C. M. Song, Chem. Nat. Compd., 56, 500 (2020).
D. Luo, N. Lv, L. J. Zhu, L. M. Liao, Y. Xu, J. Wang, W. S. Kong, H. T. Huang, M. Zhou, G. Y. Yang, Q. F. Hu, and X. X. Si, Chem. Nat. Compd., 56, 504 (2020).
Z. X. Qing, P. Yang, Q. Tang, P. Cheng, X. B. Liu, Y. J. Zheng, Y. S. Liu, and J. G. Zeng, Curr. Org. Chem., 21 (18), 1920 (2017).
G. Y. Yang, J. M. Dai, Z. J. Li, J. Wang, F. X. Yang, X. Liu, J. Li, Q. Gao, X. M. Li, Y. K. Li, W. G. Wang, M. Zhou, and Q. F. Hu, Arch. Pharm. Res., 45, 572 (2022).
Q. F. Hu, Y. Y. Ma, H. Y. Liu, J. M. Dai, F. X. Yang, J. D. Zhang, J. Wang, X. M. Li, X. Liu, J. Li, Y. K. Li, W. G. Wang, M. Zhou, and G. Y. Yang, Chem. Biol. Technol. Agric., 9, 88 (2022).
Acknowledgment
This project was supported by the Foundation of the China Tobacco Monopoly Bureau Grants and Yunnan Provincial Tobacco Monopoly Bureau Grants (110202103018, 2022530000241002), the National Natural Science Foundation of China (No. 32260111), and the Foundation of Yunnan Innovative Research Team (2019HC020).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2023, pp. 739–742.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pu, G., Cha, XJ., Ma, YY. et al. Antiviral Isoquinolines from the Cigar Tobacco Derived Endophytic Fungi Aspergillus fumigatus. Chem Nat Compd 59, 876–880 (2023). https://doi.org/10.1007/s10600-023-04139-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04139-9