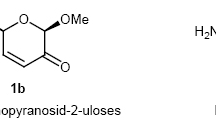
Chiral 2-trichloromethyloxazolines 4,5-cis-annulated to a carbohydrate fragment were synthesized on the basis of levoglucosenone. The synthesized compounds were converted by acid hydrolysis into 4-substituted 3-amino-1,6-anhydro-3-deoxy-β-D-mannopyranoses. Intramolecular amination of allyl trichloroacetimidates by electrophilic reagents was used to construct the oxazoline ring. It was established that trichloroacetimidates containing an epoxide ring in α,β-positions spontaneously undergo recyclization with the formation of an oxazoline ring.
Similar content being viewed by others
References
Zurabyan, S. E.; Antonenko, T. S.; Khorlin, A. Y. Carbohydr. Res. 1970, 15, 21.
(а) Fustero, S.; Salavert, E.; Pina, B.; Ramír ez de Arellano, C.; Asensio, A. Tetrahedron 2001, 57, 6475. a Pauls, H. W.; Fraser-Reid, B. J .Chem. Soc., Chem. Commun. 1983, 1031.
(a) Deng, T.; Cai, C. J. Fluorine Chem. 2013, 156, 183. (b) Zhao, Q. Y.; Shi, M. Tetrahedron 2011, 67, 3724.
Javadi, M. M.; Moghadam, M.; Mohammadpoor-Baltork, I.; Tangestaninejad, S.; Mirkhani, V.; Kargar, H.; Tahir, M. N. Polyhedron 2014, 72, 19.
(a) Plisson, F.; Prasad, P.; Xiao, X.; Piggott, A. M.; Huang, X.; Khalil, Z.; Capon, R. J. Org. Biomol. Chem. 2014, 12, 1579 (b) Sakuda, S.; Isogai, A.; Matsumoto, S.; Suzuki, A. J. Antibiot. 1987, 40, 296.
Pirrung, M. C.; Tumey, L. N.; McClerren, A. L.; Raetz, C. R. H. J. Am. Chem. Soc. 2003, 125, 1575.
Padmavathi, V.; Mahesh, K.; Venkata Subbaiah, D. R. C.; Deepti, D.; Sudhakar Reddy, G. ARKIVOC 2009, (x), 195.
Moraski, G. C.; Markley, L. D.; Chang, M.; Cho. S.; Franzblau, S. G.; Hwang, C. H.; Boshoff, H.; Miller, M. J. Bioorg. Med. Chem. 2012, 20, 2214.
Gordey, E. E.; Yadav, P. N.; Merrin, M. P.; Davies, J.; Ward, S. A.; Woodman, G. M. J.; Sadowy, A. L.; Smith, T. G.; Gossage, R. A. Bioorg. Med. Chem. Lett. 2011, 21, 4512.
Li, Q.; Woods, K. W.; Claiborne, A.; Gwaltney, S. L.; Barr, K. J.; Liu, G.; Gehrke, L.; Credo, R. B.; Hui, Y. H.; Lee, J.; Warner, R. B.; Kovar, P.; Nukkala, M. A.; Zielinski, N. A.; Tahir, S. K.; Fitzgerald, M.; Kim, K. H.; Marsh, K.; Frost, D.; Ng, S.-C.; Rosenberg, S.; Sham, H. L. Bioorg. Med. Chem. Lett. 2002, 12, 465.
Diana, G. D.; Oglesby, R. C.; Akullian, V.; Carabateas, P. M.; Cutcliffe, D.; Mallamo, J. P.; Otto, M. J.; McKinlay, M. A.; Maliski, E. G.; Michalec, S. J. J. Med. Chem. 1987, 30, 383.
(a) Irmak, M.; Groschner, A.; Boysen, M. M. K. Chem. Commun. 2007, 177. (b) Kraft, J.; Golkowski, M.; Ziegler, T. Beilstein J. Org. Chem. 2016, 12, 166. (c) Mata, Y.; Diéguez, M.; Pàmies, O.; Claver, C. Adv. Synth. Catal. 2005, 347, 1943.
Davydova, A. N.; Sharipov, B. T.; Valeev, F. A. Russ. J. Org. Chem. 2015, 51, 1408.
Das, B.; Venkateswarlu, K.; Damodar, K.; Suneel, K. J. Mol. Catal. A: Chem. 2007, 269, 17.
(a) Bongini, A.; Cardillo, G.; Orena, M.; Sandri, S.; Tomasini, C. Tetrahedron 1983, 39, 3801. (b) Shabany, H.; Spilling, C. D. Tetrahedron Lett. 1998, 39, 1465.
Shafizadeh, F.; Furneaux, R. H.; Stevenson, T. T. Carbohydr. Res. 1979, 71,169.
Ma, X.; Liu, X.; Yates, P.; Raverty, W.; Banwell, M. G.; Ma, C.; Willis, A. C.; Carr, P. D. Tetrahedron 2018, 74, 5000.
(a) Giuliano, R. M.; Deisenroth, T. W.; Frank, W. C. J. Org. Chem. 1986, 51, 2304. (b) Smith, G. R.; Giuliano, R. M. Carbohydr. Res. 1999, 323, 208.
Russell, G. A.; Shi, B. Z. Synlett 1993, 701.
(а) Comba, M. B.; Mangione, M. I.; Suárez, A. G.; Sarotti, A. M.; Spanevello, R. A. Eur. J. Org. Chem. 2018, 6848. a Matsumoto, K.; Ebata, T.; Koseki, K.; Okano, K.; Kawakami, H.; Matsushita, H. Heterocycles 1992, 34, 1935. b Ma, X.; Anderson, N.; White, L. V.; Bae, S.; Raverty, W.; Willis, A. C.; Banwell, M. G. Aust. J. Chem. 2015, 68, 593.
Jacobsen, S. Acta Chem. Scand., Ser. B 1988, B42, 605.
The authors are grateful to the Collective Use Center “Khimiya”, Ufa Institute of Chemistry, Ufa Federal Research Center of the Russian Academy of Sciences, the Regional Center for Collective Use “Agidel” of the Ufa Federal Research Center of the Russian Academy of Sciences for analytical and spectral studies, and Circa Group (Melbourne, Australia) for the generous gift of levoglucosenone.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2022, 58(4/5), 199–205
Supplementary Information
ESM 1
(PDF 1576 kb)
Rights and permissions
About this article
Cite this article
Sharipov, B.T., Davidova, A.N. & Valeev, F.А. cis-Annulation of an oxazoline fragment to levoglucosenone and the synthesis of 4-substituted 3-amino-1,6-anhydro-3-deoxy-β-D-mannopyranose. Chem Heterocycl Comp 58, 199–205 (2022). https://doi.org/10.1007/s10593-022-03073-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-022-03073-y