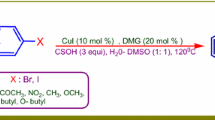
Abstract
We describe a copper(I) mediated halogenation via direct C–H functionalization of arenes. The combination of Cu(I)X and (diacetoxyiodo)benzene provides a robust system for halogenation of 2-arylpyridines. High regio- and mono-selectivity was achieved for chlorination, bromination, and iodination of arene C–H bonds.
Graphical Abstract







Similar content being viewed by others
References
Yamaguchi J, Yamaguchi AD, Itami K (2012) Angew Chem Int Ed 51:8960
Chen DYK, Youn SW (2012) Chem Eur J 18:9452
McGrath N, Brichacek M, Njardarson JT (2010) J Chem 8:1348
Cernak T, Dykstra KD, Tyagarajan S, Vachal P, Krska SW (2016) Chem Soc Rev 45:546
Dela Mare PBD (1976) Electrophilic halogenation. Cambridge University Press, New York
Snieckus V (1990) Chem Rev 90:879
Kondo Y, Shilai M, Uchiyama M, Sakamoto T (1999) J Am Chem Soc 121:3539
Giri R, Shi BF, Engle KM, Maugel N, Yu JQ (2009) Chem Soc Rev 38:3242
Lyons TW, Sanford MS (2010) Chem Rev 110:1147
Colby DA, Bergman RG, Ellman JA (2010) Chem Rev 110:624
Zhang M, Zhang Y, Jie X, Zhao H, Li G, Su W (2014) Org Chem Front 1:843
Liao G, Shi BF (2015) Acta Chim Sin 73:1283
Chen Z, Wang B, Zhang J, Yu W, Liu Z, Zhang Y (2015) Org Chem Front 2:1107
Petrone DA, Ye J, Lautens M (2016) Chem Rev 116:8003
Gang L, Bingfeng S (2015) Acta Chim Sin 73:1283
Dick AR, Hull KL, Sanford MS (2004) J Am Chem Soc 126:2300
Giri R, Chen X, Yu JQ (2005) Angew Chem Int Ed 44:2112
Wan XB, Ma ZX, Li BJ, Zhang KY, Cao SK, Zhang SW, Shi ZJ (2006) J Am Chem Soc 128:7416
Mei TS, Giri R, Maugel N, Yu JQ (2008) Angew Chem Int Ed 47:5215
Zhao X, Dimitrijevic E, Dong VM (2009) J Am Chem Soc 131:3466
Kakiuchi F, Kochi T, Mutsutani H, Kobayashi N, Urano S, Sato M, Nishiyama S, Tanabe T (2009) J Am Chem Soc 131:11310
Song B, Zheng X, Moand J, Xu B (2010) Adv Synth Catal 352:329
Bedford RB, Haddow MF, Mitchell CJ, Webster RL (2011) Angew Chem Int Ed 50:5524
Wang XC, Hu Y, Bonacorsi S, Hong Y, Burrell R, Yu JQ (2013) J Am Chem Soc 135:10326
Sarkar D, Melkonyan FS, Gulevich AV, Gevorgyan V (2013) Angew Chem Int Ed 52:10800
Chu L, Xiao KJ, Yu JQ (2014) Science 346:451
Zhang Q, Yang F, Wu Y (2014) Org Chem Front 1:694
Moghaddam FM, Tavakoli G, Saeednia B, Langer P, Jafari B (2016) J Org Chem 81:3868
Wang LH, Ackermann L (2014) Chem Commun 50:1083
Teskey CJ, Lui AYW, Greaney MF (2015) Angew Chem Int Ed 54:11677
Schroder N, Wencel-Delord J, Glorius F (2012) J Am Chem Soc 134:8298
Kuhl N, Schroder N, Glorius F (2013) Org Lett 15:3860
Hwang H, Kim J, Jeongand J, Chang S (2014) J Am Chem Soc 136:10770
Qian G, Hong X, Liu B, Mao H, Xu B (2014) Org Lett 16:5294
Schroder N, Lied F, Glorius F (2015) J Am Chem Soc 137:1448
Lied F, Lerchen A, Knecht T, Lichtenfeld CM, Glorius F (2016) ACS Catal 6:7839
Mo FY, Yan JM, Qiu D, Li F, Zhang Y, Wang JB (2010) Angew Chem Int Ed 49:2028
Truong T, Klimovica K, Daugulis O (2013) J Am Chem Soc 135:9342
Yao B, Wang ZL, Zhang H, Wang DX, Zhao L, Wang MX (2012) J Org Chem 77:3336
Urones B, Martinez AM, Rodriguez N, Arrayas RG, Carretero JC (2013) Chem Commun 49:11044
Li B, Liu B (2015) Chem Commun 51:5093
Subramanian MA, Manzer LE (2002) Science 297:1665
Rao WH, Shi BF (2016) Org Chem Front 3:1028
Shang M, Z.Sun S, Wang HL, Wang MM, Dai HX (2016) Synthesis 48:4381
Phipps RJ, Gaunt MJ (2009) Science 323:1593
Duong HA, Gilligan RE, Cooke ML, Phipps RJ, Gaunt MJ (2010) Angew Chem Int Ed 50:463
Yu DG, Gensch T, Azambuja F, Va´squez-Ce spedes, Glorius S F (2014) J Am Chem Soc 136:17722
Zhan BB, Liu YH, Fang H, Shi BF (2016) Chem Commun 52:4934
Chen X, Hao XS, Goodhue CE, Yu JQ (2006) J Am Chem Soc 128:6790
Wang WH, Pan CD, Chen F, Cheng J (2011) Chem Commun 47:3978
Mo S, Zhu YM, Shen ZM (2013) Org Biomol Chem 11:2756
Du ZJ, Gao LX, Lin YJ, Han FS (2014) Chem Cat Chem 6:123
Fan Z, Ni J, Zhang A (2016) J Am Chem Soc 138:8470
Zhdankin VV, Stang PJ (2002) Chem Rev 102:2523
Zhdankin VV, Stang PJ (2008) Chem Rev 108:5299
Yoshimura A, Zhdankin VV (2016) Chem Rev 116:3328
Arnold AM, Ulmer A, Gulder T (2016) Chem Eur J 22:8728
Xu J, Zhu X, Zhou G, Ying B, Ye P, Su L, Shen C, Zhang P (2016) Org Biomol Chem 14:3016
Liu XX, Wu ZY, Luo XL, He YQ, Zhou XQ, Fan YX, Huang GS (2016) RSC Adv 6:71485
Hao W, Liu Y (2015) Beilstein J Org Chem 11:2132
Wan JP, Lin Y, Cao X, Liu Y, Wei L (2016) Chem Commun 52:1270
Wan JP, Hu D, Bai F, Wei L, Liu Y (2016) RSC Adv 6:73132
Deprez NR, Sanford MS (2007) Inorg Chem 46:1924
Whitefield BW, Maimone TJ, Lin DW, Castroviejo MP, Baran PS (2007) J Am Chem Soc 129:12857
Phipps RJ, Grimster NP, Gaunt MJ (2008) J Am Chem Soc 130:8172
Hickman AJ, Sanford MS (2012) Nature 484:177
Acknowledgements
The authors are grateful to to the financial assistance provided by CSIR-Senior Research Associateship (Scientists Pool Scheme), New Delhi and UGC, Govt. of India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Perumgani, P.C., Parvathaneni, S.P., Surendra Babu, G.V. et al. Copper(I) Halide for Regioselective Ortho-Halogenation of Directed Arenes. Catal Lett 148, 1067–1072 (2018). https://doi.org/10.1007/s10562-018-2324-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-018-2324-5