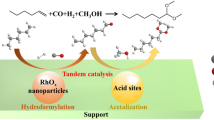
Abstract
The acetal formation mechanism under acid-free Rh-catalyzed hydroformylation–acetalization condition has been studied using different rhodium catalyst precursors in MeOH. In the absence of added acidic co-catalyst, the acetalization is catalyzed by the H+ formed in situ under hydroformylation condition, and Rh active site on Rh-phosphine catalyst did not exhibit catalytic activity for acetalization. Whether H+ can be generated in situ is related with the structure of rhodium catalyst precursor. Under hydroformylation condition, added Brønsted acids as co-catalysts can improve acetalization efficiency, but the H+ concentration in the system should not be excessively high to avoid the acid-induced inhibition for hydroformylation.
Graphical Abstract



Similar content being viewed by others
References
Patai S (1966) The Chemistry of the Carbonyl Group. Wiley-Interscience, New York, p 1970
Eilbracht P, Bärfacker L, Buss C, Hollmann C, Kitsos-Rzychon BE, Kranemann CL, Rische T, Roggenbuck R, Schmidt A (1999) Chem Rev 99:3329
Cabrera A, Mortreux A, Petit F (1988) J Mol Catal 47:11
Parrinello G, Stille JK (1987) J Am Chem Soc 109:7122
Stille JK, Su H, Brechot P, Parrinello G, Hegedus LS (1991) Organometallics 10:1183
Balue J, Bayon JC (1999) J Mol Catal A 137:193
Diwakar MM, Deshpande RM, Chaudhari RV (2005) J Mol Catal A 232:179
Fernández E, Castillón S (1994) Tetrahedron Lett 35:2361
Soulantica K, Sirol S, Koïnis S, Pneumatikakis G, Kalck Ph (1995) J Organomet Chem 498:C10
Fernández E, Ruiz A, Claver C, Castillón S, Pólo A (1998) Chem Commun 1803
El Ali B, Tijani J, Fettouhi M (2005) J Mol Catal A 230:9
El Ali B, Tijani J, Fettouhi M (2006) Appl Catal A 303:213
Vieira CG, da Silva JG, Penna CAA, dos Santos EN, Gusevskaya EV (2010) Appl Catal A 380:125
Diebolt O, Cruzeuil C, Müller C, Vogt D (2012) Adv Synth Catal 354:670
Schenck TG, Downes JM, Milne CRC, Mackenzie PB, Boucher H (1985) Inorg Chem 24:2334
Brauer DJ, Schenk S, Roßenbach S, Tepper M, Stelzer O, Hausler T, Sheldrick WSJ (2000) Organomet Chem 598:116
Krompieca S, Penkalaa M, Szczubiałkab K, Kowalska E (2012) Coord Chem Rev 256:2057
Jin X, Zhao K, Cui F, Kong F, Liu Q (2013) Green Chem. doi:10.1039/C3GC41231H
Evans D, Osborn JA, Wilkinson G (1964) J Chem Soc 3133
Acknowledgments
We gratefully thank the financial support from National Natural Science Foundation of China (No. 20606019, 20976086), the Foundation of Key Laboratory of Oil & Gas Fine Chemicals, Ministry of Education, China (No.XJDX0908-2010-01) and the Natural Science Foundation of Qingdao, China (No. 12-1-4-3-(6)-jch).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, X., Zhao, K., Kong, F. et al. The Mechanism of Acetal Formation in Acid-Free Rh-Catalyzed Tandem Hydroformylation–Acetalization of Olefins in MeOH. Catal Lett 144, 192–196 (2014). https://doi.org/10.1007/s10562-013-1109-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-013-1109-0