Abstract
Purpose
While statins and antiplatelet therapies are largely prescribed together worldwide, limited information is available on the safety of their association regarding rhabdomyolysis occurrence. We aimed to assess the reporting of rhabdomyolysis in patients treated with a combination of statin and antiplatelet therapy, compared to statin alone.
Methods
We used the World Health Organization pharmacovigilance database (VigiBase®) to compare the rhabdomyolysis reporting between statin (atorvastatin, fluvastatin, pravastatin, rosuvastatin, and simvastatin) plus antiplatelet therapy (acetylsalicylic acid, clopidogrel, prasugrel and ticagrelor) groups versus statin alone groups, for each statin and antiplatelet therapy. Study setting was restricted to patients aged 45 or older, including reports up until 1st September, 2021. We computed reporting Odds-Ratio (ROR) and their 95% confidence interval (CI) to quantify the disproportionality between groups, adjusted on age and sex.
Results
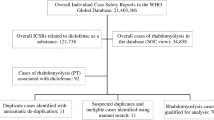
Among the 11,431,708 reports of adverse reactions, we extracted 9,489 cases of rhabdomyolysis in patients treated with statins, of whom 2,464 (26%) were also treated with antiplatelet therapy. The reporting of rhabdomyolysis was increased when ticagrelor was associated with atorvastatin (ROR 1.30 [1.02–1.65]) or rosuvastatin (ROR 1.90 [1.42–2.54]) compared to the respective statin alone but did not change when aspirin, clopidogrel or prasugrel were considered.
Conclusion
Rhabdomyolysis reporting was increased when ticagrelor -but not other antiplatelet agents- was notified with the most prescribed statins in practice. This finding needs to be considered by physicians especially in high-risk patients.

Similar content being viewed by others
Data Availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet. 2016;388(10059):2532–61.
Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients with Non-ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;64(24):e139–228.
Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289–367.
Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119–77.
Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670–81.
Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment Aetiology and Management. Eur Heart J. 2015;36(17):1012–22.
Kashani A, Phillips CO, Foody JM, et al. Risks associated with statin therapy: a systematic overview of randomized clinical trials. Circulation. 2006;114(25):2788–97.
Kariyanna PT, Haseeb S, Chowdhury YS, et al. Ticagrelor and Statin Interaction Induces Rhabdomyolysis and Acute Renal Failure: Case reports and Scoping Review. Am J Med Case Rep. 2019;7(12):337–41.
Mrotzek SM, Rassaf T, Totzeck M. Ticagrelor Leads to Statin-Induced Rhabdomyolysis: A Case Report. Am J Case Rep. 2017;18:1238–41.
Vrkić Kirhmajer M, Macolić Šarinić V, Šimičević L, et al. Rosuvastatin-Induced Rhabdomyolysis - Possible Role of Ticagrelor and Patients’ Pharmacogenetic Profile. Basic Clin Pharmacol Toxicol. 2018;123(4):509–18.
Tsao CW, Aday AW, Almarzooq ZI, et al. Heart Disease and Stroke Statistics-2023 Update: A Report From the American Heart Association. Circulation. 2023;147(8):e93–621.
Mozzicato P. Standardised MedDRA queries: their role in signal detection. Drug Saf. 2007;30(7):617–9.
Chrétien B, Lelong-Boulouard V, Chantepie S, et al. Haematologic malignancies associated with clozapine v. all other antipsychotic agents: a pharmacovigilance study in VigiBase(®). Psychol Med. 2021;51(9):1459–66.
Salem JE, Manouchehri A, Moey M, et al. Cardiovascular toxicities associated with immune checkpoint inhibitors: an observational, retrospective, pharmacovigilance study. Lancet Oncol. 2018;19(12):1579–89.
Puymirat E, Simon T, Cayla G, et al. Acute Myocardial Infarction: Changes in Patient Characteristics, Management, and 6-Month Outcomes Over a Period of 20 Years in the FAST-MI Program (French Registry of Acute ST-Elevation or Non-ST-Elevation Myocardial Infarction) 1995 to 2015. Circulation. 2017;136(20):1908–19.
Townsend N, Wilson L, Bhatnagar P, Wickramasinghe K, Rayner M, Nichols M. Cardiovascular disease in Europe: epidemiological update 2016. Eur Heart J. 2016;37(42):3232–45.
Hopewell JC, Offer A, Haynes R, et al. Independent risk factors for simvastatin-related myopathy and relevance to different types of muscle symptom. Eur Heart J. 2020;41(35):3336–42.
Wishart DS, Feunang YD, Guo AC, et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2018;46(D1):D1074-D82.
Sirtori CR. The pharmacology of statins. Pharmacol Res. 2014;88:3–11.
Zhou D, Andersson TB, Grimm SW. In vitro evaluation of potential drug-drug interactions with ticagrelor: cytochrome P450 reaction phenotyping, inhibition, induction, and differential kinetics. Drug Metab Dispos. 2011;39(4):703–10.
Teng R, Mitchell PD, Butler KA. Pharmacokinetic interaction studies of co-administration of ticagrelor and atorvastatin or simvastatin in healthy volunteers. Eur J Clin Pharmacol. 2013;69(3):477–87.
Holbrook A, Wright M, Sung M, Ribic C, Baker S. Statin-associated rhabdomyolysis: is there a dose-response relationship? Can J Cardiol. 2011;27(2):146–51.
Danielak D, Karaźniewicz-Łada M, Główka F. Assessment of the Risk of Rhabdomyolysis and Myopathy During Concomitant Treatment with Ticagrelor and Statins. Drugs. 2018;78(11):1105–12.
Calderon-Ospina CA, Hernández-Sómerson M, García AM, et al. A Pharmacogenomic Dissection of a Rosuvastatin-Induced Rhabdomyolysis Case Evokes the Polygenic Nature of Adverse Drug Reactions. Pharmgenomics Pers Med. 2020;13:59–70.
Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361(11):1045–57.
Turner RM, Pirmohamed M. Statin-Related Myotoxicity: A Comprehensive Review of Pharmacokinetic, Pharmacogenomic and Muscle Components. J Clin Med. 2019; 9(1)
Potential Signals of Serious Risks/New Safety Information Identified by the FDA Adverse Event Reporting System (FAERS). July - September 2018. https://www.fda.gov/drugs/questions-and-answers-fdas-adverse-event-reporting-system-faers/july-september-2018-potential-signals-serious-risksnew-safety-information-identified-fda-adverse.
Macolic Sarinic, V. Interaction between rosuvastatin and ticagrelor resulting in rhabdomyolysis. WHO Pharmaceutical Newsletter. 2018. https://apps.who.int/iris/bitstream/handle/10665/272966/WPN-2018-03-eng.pdf?sequence=1&isAllowed=y
Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients–the PRIMO study. Cardiovasc Drugs Ther. 2005;19(6):403–14.
Zhang H, Plutzky J, Skentzos S, et al. Discontinuation of statins in routine care settings: a cohort study. Ann Intern Med. 2013;158(7):526–34.
Alfirevic A, Neely D, Armitage J, et al. Phenotype standardization for statin-induced myotoxicity. Clin Pharmacol Ther. 2014;96(4):470–6.
Law M, Rudnicka AR. Statin safety: a systematic review. Am J Cardiol. 2006;97(8A):52C-60C.
Steg PG, Bhatt DL, Simon T, et al. Ticagrelor in Patients with Stable Coronary Disease and Diabetes. N Engl J Med. 2019;381(14):1309–20.
Nguyen KA, Li L, Lu D, et al. A comprehensive review and meta-analysis of risk factors for statin-induced myopathy. Eur J Clin Pharmacol. 2018;74(9):1099–109.
Kitzmiller JP, Mikulik EB, Dauki AM, Murkherjee C, Luzum JA. Pharmacogenomics of statins: understanding susceptibility to adverse effects. Pharmgenomics Pers Med. 2016;9:97–106.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
VR and CD: Formal analysis, Writing—Original Draft; CD: statistical analysis and methodology; AL, BC, MS and SF: Writing – Review, Validation; FB and JA: Writing—Review & Editing, Supervision.
Corresponding author
Ethics declarations
Competing Interests
Dr Roule has received research grants from Medtronic; speaker fees from Bristol-Myers Squibb, AstraZeneca.
Dr Beygui reports receiving consulting and lecture from Astrazeneca, Bristol-myers Squibb, medtronic, biosensors, Boston scientific Institutionnal research grants: Medtronic, Biosensors, Acist, Boston scientific.
Other authors have nothing to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Roule, V., Alexandre, J., Lemaitre, A. et al. Rhabdomyolysis with Co-Administration of Statins and Antiplatelet Therapies—Analysis of the WHO Pharmacovigilance Database. Cardiovasc Drugs Ther (2023). https://doi.org/10.1007/s10557-023-07459-8
Accepted:
Published:
DOI: https://doi.org/10.1007/s10557-023-07459-8