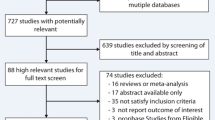
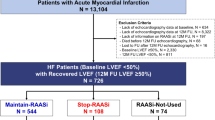
Abstract
Background and Aims
Aldosterone is one of the major factors to cause organ damage during an acute phase of heart failure (HF), and many reports have demonstrated that patients with acute decompensated HF (ADHF) have high blood aldosterone concentrations, and the high aldosterone concentrations predict poor prognosis in patients with HF. These findings suggest that eplerenone, an antagonist of aldosterone receptors may provide a new concept and strategy for the treatment of ADHF, protecting the heart and other organs during chronic phases, depending on the restoration of hemodynamic abnormalities.
Methods
EARLIER is an event-driven clinical trial with an estimated enrolment of 300 patients hospitalized with ADHF with reduced left ventricular ejection fraction. ADHF includes ischemic or non-ischemic HF, and patients can be enrolled within 72 h after the visit to the hospital. We randomize the patients taking standard therapies for ADHF to the eplerenone and placebo groups. Eplerenone, either 25 or 50 mg, is administered for 6 months in the eplerenone group, and the corresponding placebo is administered in the placebo group on top of the standard care. We set the primary endpoint as the incidence of the composite endpoint (cardiac death or first re-hospitalization due to cardiac disease) 6 months after the enrollment, and also check the quality of life, i.e., exercise capacity and safety features of eplerenone.
Conclusion and Perspectives
EARLIER is a clinical trial of eplerenone targeting ADHF and also the first multicenter investigator-initiated phase III trial in the cardiovascular field in Japan, funded by the Japanese government.

Similar content being viewed by others
References
Chang PP, Chambless LE, Shahar E, et al. Incidence and survival of hospitalized acute decompensated heart failure in four US communities (from the atherosclerosis risk in communities study). Am J Cardiol. 2014;113:504–10.
Guidelines for treatment of acute heart failure (JCS 2011). Cir J: Off J Jpn Circ Soc. 2013;77:2157–201.
McMurray JJ, Adamopoulos S, Anker SD, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European society of cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–69.
Mentz RJ, Bakris GL, Waeber B, et al. The past, present and future of renin-angiotensin aldosterone system inhibition. Int J Cardiol. 2013;167:1677–87.
O’Connor CM, Fiuzat M, Swedberg K, et al. Influence of global region on outcomes in heart failure beta-blocker trials. J Am Coll Cardiol. 2011;58:915–22.
Fonarow GC, Corday E. Overview of acutely decompensated congestive heart failure (ADHF): a report from the ADHERE registry. Heart Fail Rev. 2004;9:179–85.
Sato N, Kajimoto K, Keida T, et al. Clinical features and outcome in hospitalized heart failure in Japan (from the ATTEND registry). Circ J: Off J Jpn Circ Soc. 2013;77:944–51.
Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized aldactone evaluation study investigators. N Engl J Med. 1999;341:709–17.
Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348:1309–21.
Girerd N, Pang PS, Swedberg K, et al. Serum aldosterone is associated with mortality and re-hospitalization in patients with reduced ejection fraction hospitalized for acute heart failure: analysis from the EVEREST trial. Eur J Heart Fail. 2013;15:1228–35.
Gheorghiade M, Pang PS, Ambrosy AP, et al. A comprehensive, longitudinal description of the in-hospital and post-discharge clinical, laboratory, and neurohormonal course of patients with heart failure who die or are re-hospitalized within 90 days: analysis from the EVEREST trial. Heart Fail Rev. 2012;17:485–509.
Zannad F, McMurray JJ, Krum H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364:11–21.
McMurray JJ. CONSENSUS to EMPHASIS: the overwhelming evidence which makes blockade of the renin-angiotensin-aldosterone system the cornerstone of therapy for systolic heart failure. Eur J Heart Fail. 2011;13:929–36.
Cleland JG, Coletta AP, Buga L, et al. Clinical trials update from the American heart association meeting 2010: EMPHASIS-HF, RAFT, TIM-HF, Tele-HF, ASCEND-HF, ROCKET-AF, and PROTECT. Eur J Heart Fail. 2011;13:460–5.
Dzau V. The cardiovascular continuum and renin-angiotensin-aldosterone system blockade. J Hyperten Suppl: Off J Int Soc Hypertens. 2005;23:S9–17.
Maisel A, Mueller C, Nowak R, et al. Mid-region pro-hormone markers for diagnosis and prognosis in acute dyspnea: results from the BACH (biomarkers in acute heart failure) trial. J Am Coll Cardiol. 2010;55:2062–76.
Solomon SD, Dobson J, Pocock S, et al. Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure. Circ. 2007;116:1482–7.
O’Connor CM, Gattis WA, Uretsky BF, et al. Continuous intravenous dobutamine is associated with an increased risk of death in patients with advanced heart failure: insights from the Flolan International Randomized Survival Trial (FIRST). Am Heart J. 1999;138:78–86.
Silver MA, Horton DP, Ghali JK, Elkayam U. Effect of nesiritide versus dobutamine on short-term outcomes in the treatment of patients with acutely decompensated heart failure. J Am Coll Cardiol. 2002;39:798–803.
Chen HH, Anstrom KJ, Givertz MM, et al. Low-dose dopamine or low-dose nesiritide in acute heart failure with renal dysfunction: the ROSE acute heart failure randomized trial. Jama. 2013;310:2533–43.
Hernandez AF, O’Connor CM, Starling RC, et al. Rationale and design of the Acute Study of Clinical Effectiveness of Nesiritide in Decompensated Heart Failure Trial (ASCEND-HF). Am Heart J. 2009;157:271–7.
Nagai R, Kinugawa K, Inoue H, et al. Urgent management of rapid heart rate in patients with atrial fibrillation/flutter and left ventricular dysfunction: comparison of the ultra-short-acting beta1-selective blocker landiolol with digoxin (J-Land Study). Circ J: Off J Jpn Circ Soc. 2013;77:908–16.
Montalescot G, Pitt B, de Sa Lopez E, et al. Early eplerenone treatment in patients with acute ST-elevation myocardial infarction without heart failure: the randomized double-blind reminder study. Eur Heart J. 2014;35:2295–302.
Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2014;370:1383–92.
Kitakaze M, Asakura M, Kim J, et al. Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction (J-WIND): two randomised trials. Lancet. 2007;370:1483–93.
Acknowledgments
We acknowledge the great help from Ms Y.Miyawaki, M.Ono and C.Takayama, and Mr. T.Uraki for the secretarial works. Mr. M Yamamoto, Mr. S Fukunaga and Ms K Terasawa at the Center for Clinical Trial, the Japan Medical Association (JMACCT) advises us how to appropriately form the EARLIER study organization for study implementation.
Funding
This work was supported by a clinical trial promotion project of Japan Medical Association, which is funded by a Health Labour Sciences Research Grant from the Ministry of Health, Labour and Welfare.
Conflict of interest
MK, KA, KH, KK, YK, YS and AY received honorarium form Pfizer Japan. MK 、AH and YS received the research funding from Pfizer Japan. MK and TY is advisory board members of clinical trial on eplerenone in chronic heart failure (A6141114).
The drugs are generously provided by Pfizer USA based on the policy of J-GCP.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Asakura, M., Yamamoto, H., Asai, K. et al. Rationale and Design of the Double-Blind, Randomized, Placebo-Controlled Multicenter Trial on Efficacy of Early Initiation of Eplerenone Treatment in Patients with Acute Heart Failure (EARLIER). Cardiovasc Drugs Ther 29, 179–185 (2015). https://doi.org/10.1007/s10557-014-6565-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-014-6565-2