Abstract
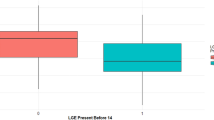
Cardiac MR (CMR) is increasingly used to assess for cardiac involvement in patients with Duchenne muscular dystrophy (DMD). The frequent use of gadolinium based contrast agents (GBCAs) has been called into question with reports of intracranial gadolinium deposition in patients receiving multiple administrations. We adopted a conservative GBCA administration policy, limiting the frequency of GBCA exposure in patients with previously documented late gadolinium enhancement. The aim of our study was to evaluate the clinical effects of this policy change. Data were retrospectively reviewed on 405 consecutive patients with DMD who underwent CMR evaluation. Patients were grouped into conservative GBCA administration or historical control. CMR reports were evaluated and clinical reports were reviewed to determine actionable changes. Ohio Medicaid reimbursements were used to estimate costs. A total of 187 patients comprised the conservative GBCA group and 218 patients the historical cohort. The conservative GBCA group had lower contrast administration rates (84% vs. 99%, p < 0.0001), shorter scan times (35.2 vs. 39.0 min, p < 0.0001), and lower estimated medical costs ($339 vs. $351/study). There was no change regarding the initial presence of first-time late gadolinium enhancement, and no difference in actionable change. Contrast administration substantially decreased 7 months post-policy change (65%) compared to the initial 7 months (96%, p < 0.0001). In the current era with unclear concern for intracranial gadolinium deposition, thoughtful GBCA administration is warranted in patients anticipated to undergo multiple CMRs. Our updated approach has resulted in fewer patients receiving contrast, shorter scan times, and less medical costs, without appreciable changes to patient management.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author [SML]. The data are not publicly available due to records which may contain information that could compromise patient confidentiality.
Abbreviations
- CMR:
-
Cardiac magnetic resonance
- DMD:
-
Duchenne muscular dystrophy
- GBCA:
-
Gadolinium based contrast agent
- LGE:
-
Late gadolinium enhancement
- SSFP:
-
Steady-state free precession
References
Muntoni F, Torelli S, Ferlini A (2003) Dystrophin and mutations: one gene, several proteins, multiple phenotypes. Lancet Neurol 2(12):731–740
Centers for Disease Control and Prevention (2009) Prevalence of Duchenne/Becker muscular dystrophy among males aged 5–24 years—four states, 2007. MMWR Morb Mortal Wkly Rep 58(40):1119–1122
Eagle M, Baudouin SV, Chandler C, Giddings DR, Bullock R, Bushby K (2002) Survival in Duchenne muscular dystrophy: improvements in life expectancy since 1967 and the impact of home nocturnal ventilation. Neuromuscul Disord 12(10):926–929
Connuck DM, Sleeper LA, Colan SD, Cox GF, Towbin JA, Lowe AM et al (2008) Characteristics and outcomes of cardiomyopathy in children with Duchenne or Becker muscular dystrophy: a comparative study from the Pediatric Cardiomyopathy Registry. Am Heart J 155(6):998–1005
Academy American, of Pediatrics Section on Cardiology and Cardiac Surgery (2005) Cardiovascular health supervision for individuals affected by Duchenne or Becker muscular dystrophy. Pediatrics 116(6):1569–1573
Bushby K, Muntoni F, Bourke JP (2003) 107th ENMC international workshop: the management of cardiac involvement in muscular dystrophy and myotonic dystrophy. 7th–9th June 2002, Naarden, the Netherlands. Neuromuscul Disord 13(2):166–172
Birnkrant DJ, Bushby K, Bann CM, Alman BA, Apkon SD, Blackwell A et al (2018) Diagnosis and management of Duchenne muscular dystrophy, part 2: respiratory, cardiac, bone health, and orthopaedic management. Lancet Neurol 17(4):347–361
Ong KS, Kinnett K, Soelaeman R, Webb L, Bain JS, Martin AS et al (2018) Evaluating implementation of the updated care considerations for duchenne muscular dystrophy. Pediatrics 142(Suppl 2):S118–S128
Feingold B, Mahle WT, Auerbach S, Clemens P, Domenighetti AA, Jefferies JL et al (2017) Management of cardiac involvement associated with neuromuscular diseases: a scientific statement from the american heart association. Circulation 136(13):e200–e231
Power A, Poonja S, Disler D, Myers K, Patton DJ, Mah JK et al (2017) Echocardiographic image quality deteriorates with age in children and young adults with duchenne muscular dystrophy. Front Cardiovasc Med 4:82
Tandon A, Villa CR, Hor KN, Jefferies JL, Gao Z, Towbin JA et al (2015) Myocardial fibrosis burden predicts left ventricular ejection fraction and is associated with age and steroid treatment duration in duchenne muscular dystrophy. J Am Heart Assoc 4(4):e001338
Hor KN, Wansapura J, Markham LW, Mazur W, Cripe LH, Fleck R et al (2009) Circumferential strain analysis identifies strata of cardiomyopathy in Duchenne muscular dystrophy: a cardiac magnetic resonance tagging study. J Am Coll Cardiol 53(14):1204–1210
Kanda T, Fukusato T, Matsuda M, Toyoda K, Oba H, Kotoku J et al (2015) Gadolinium-based contrast agent accumulates in the brain even in subjects without severe renal dysfunction: evaluation of autopsy brain specimens with inductively coupled plasma mass spectroscopy. Radiology 276(1):228–232
Kanda T, Ishii K, Kawaguchi H, Kitajima K, Takenaka D (2014) High signal intensity in the dentate nucleus and globus pallidus on unenhanced T1-weighted MR images: relationship with increasing cumulative dose of a gadolinium-based contrast material. Radiology 270(3):834–841
Kanda T, Matsuda M, Oba H, Toyoda K, Furui S (2015) Gadolinium deposition after contrast-enhanced MR imaging. Radiology 277(3):924–925
Kanda T, Oba H, Toyoda K, Kitajima K, Furui S (2016) Brain gadolinium deposition after administration of gadolinium-based contrast agents. Jpn J Radiol 34(1):3–9
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK et al (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105(4):539–542
Weinmann HJ, Brasch RC, Press WR, Wesbey GE (1984) Characteristics of gadolinium-DTPA complex: a potential NMR contrast agent. AJR Am J Roentgenol 142(3):619–624
Hao D, Ai T, Goerner F, Hu X, Runge VM, Tweedle M (2012) MRI contrast agents: basic chemistry and safety. J Magn Reson Imaging 36(5):1060–1071
Bae S, Lee HJ, Han K, Park YW, Choi YS, Ahn SS et al (2017) Gadolinium deposition in the brain: association with various GBCAs using a generalized additive model. Eur Radiol 27(8):3353–3361
Ersoy H, Rybicki FJ (2007) Biochemical safety profiles of gadolinium-based extracellular contrast agents and nephrogenic systemic fibrosis. J Magn Reson Imaging 26(5):1190–1197
McDonald RJ, McDonald JS, Kallmes DF, Jentoft ME, Murray DL, Thielen KR et al (2015) Intracranial gadolinium deposition after contrast-enhanced MR imaging. Radiology 275(3):772–782
Cao Y, Huang DQ, Shih G, Prince MR (2016) Signal change in the dentate nucleus on T1-weighted MR images after multiple administrations of gadopentetate Dimeglumine versus gadobutrol. AJR Am J Roentgenol 206(2):414–419
Radbruch A, Weberling LD, Kieslich PJ, Eidel O, Burth S, Kickingereder P et al (2015) Gadolinium retention in the dentate nucleus and globus pallidus is dependent on the class of contrast agent. Radiology 275(3):783–791
Stojanov DA, Aracki-Trenkic A, Vojinovic S, Benedeto-Stojanov D, Ljubisavljevic S (2016) Increasing signal intensity within the dentate nucleus and globus pallidus on unenhanced T1W magnetic resonance images in patients with relapsing-remitting multiple sclerosis: correlation with cumulative dose of a macrocyclic gadolinium-based contrast agent, gadobutrol. Eur Radiol 26(3):807–815
Silva MC, Magalhaes TA, Meira ZM, Rassi CH, Andrade AC, Gutierrez PS et al (2017) Myocardial fibrosis progression in Duchenne and becker muscular dystrophy: a randomized clinical trial. JAMA Cardiol 2(2):190–199
McNally EM, Kaltman JR, Benson DW, Canter CE, Cripe LH, Duan D et al (2015) Contemporary cardiac issues in Duchenne muscular dystrophy Working group of the National Heart, Lung, and blood institute in collaboration with parent project muscular dystrophy. Circulation 131(18):1590–1598
Chan RH, Maron BJ, Olivotto I, Pencina MJ, Assenza GE, Haas T et al (2014) Prognostic value of quantitative contrast-enhanced cardiovascular magnetic resonance for the evaluation of sudden death risk in patients with hypertrophic cardiomyopathy. Circulation 130(6):484–495
House AV, Muthurangu V, Spanel AJ, Danford DA, Mir B, Schuster A, Hsu H, Kutty S (2019) Can abbreviated cardiac magnetic resonance imaging adequately support clinical decision making after repair of tetralogy of Fallot? Pediatr Cardiol 40(3):616–622
Johnson TV, Rose GA, Fenner DJ, Rozario NL (2014) Improving appropriate use of echocardiography and single-photon emission computed tomographic myocardial perfusion imaging: a continuous quality improvement initiative. J Am Soc Echocardiogr 27(7):749–757
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests or funds to disclose.
Ethical approval
This study was approved by the Institutional Review Board at Cincinnati Children’s Hospital Medical Center.
Informed consent
The requirement for informed consent was waived due to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lang, S.M., Alsaied, T., Moore, R.A. et al. Conservative gadolinium administration to patients with Duchenne muscular dystrophy: decreasing exposure, cost, and time, without change in medical management. Int J Cardiovasc Imaging 35, 2213–2219 (2019). https://doi.org/10.1007/s10554-019-01670-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-019-01670-1