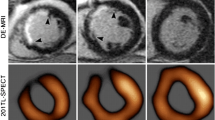
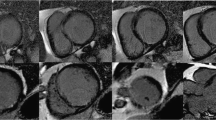
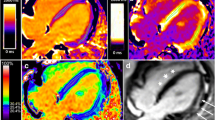
Abstract
Myocardial perfusion studies using dynamic contrast-enhanced cardiac magnetic resonance imaging (CMRI) could provide valuable, quantitative information regarding heart physiology in diseases such as Duchenne muscular dystrophy (DMD), that lead to diffuse myocardial damage. The goal of this effort was to develop an intuitive but physiologically meaningful method for quantifying myocardial perfusion by CMRI and to test its ability to detect global myocardial differences in a dog model of DMD. A discrete-time model was developed that parameterizes contrast agent kinetics in terms of an uptake coefficient that describes the forward flux of contrast agent into the tissue, and a retention coefficient that describes the rate of decay in tissue concentration due to contrast agent efflux. This model was tested in 5 dogs with DMD and 6 healthy controls which were imaged using a perfusion sequence on a 3T clinical scanner. CINE and delayed-enhancement CMRI acquisitions were also used to assess cardiac function and the presence of myocardial scar. Among functional parameters measured by CMRI, no significant differences were observed. No myocardial scar was observed. Increased perfusion in DMD was observed with an uptake coefficient of 6.76 ± 2.41 % compared to 2.98 ± 1.46 % in controls (p = 0.03). Additionally, the retention coefficient appeared lower at 82.2 ± 5.8 % in dogs with DMD compared to 90.5 ± 6.6 % in controls (p = 0.12). A discrete-time kinetic model of uptake and retention of contrast agent in perfusion CMRI shows potential for the study of DMD.




Similar content being viewed by others
References
Cury RC, Cattani CA, Gabure LA, Racy DJ, de Gois JM, Siebert U, Lima SS, Brady TJ (2006) Diagnostic performance of stress perfusion and delayed-enhancement MR imaging in patients with coronary artery disease. Radiology 240:39–45
Bingham SE, Hachamovitch R (2011) Incremental prognostic significance of combined cardiac magnetic resonance imaging, adenosine stress perfusion, delayed enhancement, and left ventricular function over preimaging information for the prediction of adverse events. Circulation 123:1509–1518
Korosoglou G, Elhmidi Y, Steen H, Schellberg D, Riedle N, Ahrens J, Lehrke S, Merten C, Lossnitzer D, Radeleff J, Zugck C, Giannitsis E, Katus HA (2010) Prognostic value of high-dose dobutamine stress magnetic resonance imaging in 1,493 consecutive patients: assessment of myocardial wall motion and perfusion. J Am Coll Cardiol 56:1225–1234
Doesch C, Seeger A, Doering J, Herdeg C, Burgstahler C, Claussen CD, Gawaz M, Miller S, May AE (2009) Risk stratification by adenosine stress cardiac magnetic resonance in patients with coronary artery stenoses of intermediate angiographic severity. JACC Cardiovasc Imaging 2:424–433
Tsukiji M, Nguyen P, Narayan G, Hellinger J, Chan F, Herfkens R, Pauly JM, McConnell MV, Yang PC (2006) Peri-infarct ischemia determined by cardiovascular magnetic resonance evaluation of myocardial viability and stress perfusion predicts future cardiovascular events in patients with severe ischemic cardiomyopathy. J Cardiovasc Magn Reson 8:773–779
Selvanayagam JB, Jerosch-Herold M, Porto I, Sheridan D, Cheng AS, Petersen SE, Searle N, Channon KM, Banning AP, Neubauer S (2005) Resting myocardial blood flow is impaired in hibernating myocardium: a magnetic resonance study of quantitative perfusion assessment. Circulation 112:3289–3296
Gerber BL, Raman SV, Nayak K, Epstein FH, Ferreira P, Axel L, Kraitchman DL (2008) Myocardial first-pass perfusion cardiovascular magnetic resonance: history, theory, and current state of the art (review). J Cardiovasc Magn Reson 10:18
Jerosch-Herold M (2010) Quantification of myocardial perfusion by cardiovascular magnetic resonance (review). J Cardiovasc Magn Reson 12:57
Tyler KL (2003) Origins and early descriptions of “Duchenne muscular dystrophy”. Muscle Nerve 28:402–422
Muntoni F, Torelli S, Ferlini A (2003) Dystrophin and mutations: one gene, several proteins, multiple phenotypes (review). Lancet Neurol 2:731–740
Finsterer J, Stollberger C (2003) The heart in human dystrophinopathies (review). Cardiology 99:1–19
Duan D (2006) Challenges and opportunities in dystrophin-deficient cardiomyopathy gene therapy. Hum Mol Genet 15(Spec. No. 2):R253–R261
Hoogerwaard EM, van der Wouw PA, Wilde AA, Bakker E, Ippel PF, Oosterwijk JC, Majoor-Krakauer DF, van Essen AJ, Leschot NJ, de Visser M (1999) Cardiac involvement in carriers of Duchenne and Becker muscular dystrophy. Neuromuscul Disord 9:347–351
Silva MC, Meira ZM, Gurgel GJ, da Silva MM, Campos AF, Barbosa MM, Starling Filho GM, Ferreira RA, Zatz M, Rochitte CE (2007) Myocardial delayed enhancement by magnetic resonance imaging in patients with muscular dystrophy. J Am Coll Cardiol 49:1874–1879
Puchalski MD, Williams RV, Askovich B, Sower CT, Hor KH, Su JT, Pack N, Dibella E, Gottliebson WM (2009) Late gadolinium enhancement: precursor to cardiomyopathy in Duchenne muscular dystrophy? Int J Cardiovasc Imaging 25:57–63
Bilchick KC, Salerno M, Plitt D, Dori Y, Crawford TO, Drachman D, Thompson WR (2011) Prevalence and distribution of regional scar in dysfunctional myocardial segments in Duchenne muscular dystrophy. J Cardiovasc Magn Reson 13:20
Perloff JK, Henze E, Schelbert HR (1984) Alterations in regional myocardial metabolism, perfusion, and wall motion in Duchenne muscular dystrophy studied by radionuclide imaging. Circulation 69:33–42
Nishimura T, Yanagisawa A, Sakata H, Sakata K, Shimoyama K, Ishihara T, Yoshino H, Ishikawa K (2001) Thallium-201 single photon emission computed tomography (SPECT) in patients with Duchenne’s progressive muscular dystrophy: a histopathologic correlation study. Jpn Circ J 65:99–105
Kelle S, Graf K, Dreysse S, Schnackenburg B, Fleck E, Klein C (2010) Evaluation of contrast wash-in and peak enhancement in adenosine first pass perfusion CMR in patients post bypass surgery. J Cardiovasc Magn Reson 12:28
Nagel E, Klein C, Paetsch I, Hettwer S, Schnackenburg B, Wegscheider K, Fleck E (2003) Magnetic resonance perfusion measurements for the noninvasive detection of coronary artery disease. Circulation 108:432–437
Diesbourg LD, Prato FS, Wisenberg G, Drost DJ, Marshall TP, Carroll SE, O’Neill B (1992) Quantification of myocardial blood flow and extracellular volumes using a bolus injection of Gd-DTPA: kinetic modeling in canine ischemic disease. Magn Reson Med 23:239–253
Pack NA, Dibella EV, Wilson BD, McGann CJ (2008) Quantitative myocardial distribution volume from dynamic contrast-enhanced MRI. Magn Reson Imaging 26:532–542
Li X, Springer CS Jr, Jerosch-Herold M (2009) First-pass dynamic contrast-enhanced MRI with extravasating contrast reagent: evidence for human myocardial capillary recruitment in adenosine-induced hyperemia. NMR Biomed 22:148–157
Patel PP, Koppenhafer SL, Scholz TD (1995) Measurement of kinetic perfusion parameters of gadoteridol in intact myocardium: effects of ischemia/reperfusion and coronary vasodilation. Magn Reson Imaging 13:799–806
Valentine BA, Cooper BJ, de Lahunta A, O’Quinn R, Blue JT (1988) Canine X-linked muscular dystrophy. An animal model of Duchenne muscular dystrophy: clinical studies. J Neurol Sci 88:69–81
Kerwin WS, Cai J, Yuan C (2002) Noise and motion correction in dynamic contrast-enhanced MRI for analysis of atherosclerotic lesions. Magn Reson Med 47:1211–1217
Kerwin WS, Oikawa M, Yuan C, Jarvik GP, Hatsukami TS (2008) MR imaging of adventitial vasa vasorum in carotid atherosclerosis. Magn Reson Med 59:507–514
Ahmad N, Welch I, Grange R, Hadway J, Dhanvantari S, Hill D, Lee TY, Hoffman LM (2011) Use of imaging biomarkers to assess perfusion and glucose metabolism in the skeletal muscle of dystrophic mice. BMC Muscul Disord 12:127
Melacini P, Vianello A, Villanova C, Fanin M, Miorin M, Angelini C, Dalla VS (1996) Cardiac and respiratory involvement in advanced stage Duchenne muscular dystrophy. Neuromuscul Disord 6:367–376
Lanza GA, Dello RA, Giglio V, De Luca L, Messano L, Santini C, Ricci E, Damiani A, Fumagalli G, De Martino G, Mangiola F, Bellocci F (2001) Impairment of cardiac autonomic function in patients with Duchenne muscular dystrophy: relationship to myocardial and respiratory function. Am Heart J 141:808–812
van Bockel EA, Lind JS, Zijlstra JG, Wijkstra PJ, Meijer PM, van den Berg MP, Slart RH, Aarts LP, Tulleken JE (2009) Cardiac assessment of patients with late stage Duchenne muscular dystrophy. Netherlands Heart J 17:232–237
Acknowledgments
We thank Baocheng Chu and Jennifer Totman for technical assistance with MRI; A. Joslyn, E. Zellmer, Jennifer Duncan, DVM, M. Spector, DVM and their team for their care of the dogs. We further thank S. Carbonneau, H. Crawford, B. Larson, K. Carbonneau, J. Vermeulen, and D. Gayle for administrative assistance and manuscript preparation. This work was supported by NIH U54-HD47175; NIH R01-AR056949; and by a Career Development Grant (to Z. Wang) from the Muscular Dystrophy Association (MDA 114979).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kerwin, W.S., Naumova, A., Storb, R. et al. Mapping contrast agent uptake and retention in MRI studies of myocardial perfusion: case control study of dogs with Duchenne muscular dystrophy. Int J Cardiovasc Imaging 29, 819–826 (2013). https://doi.org/10.1007/s10554-012-0137-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-012-0137-y