Abstract
Purpose
The cytochrome P450 (CYP) genes are oxygenases involved in estrogen biosynthesis and metabolism, generation of DNA damaging procarcinogens, and response to anti-estrogen therapies. Since lifetime estrogen exposure is an established risk factor for breast cancer, determining the role of CYP genes in breast cancer etiology may provide critical information for understanding tumorigenesis and response to treatment.
Methods
This review summarizes literature available in PubMed published between 1993 and 2013 that focuses on studies evaluating the effects of DNA variants in CYP genes on estrogen synthesis, metabolism, and generation of procarcinogens in addition to response to anti-estrogen therapies.
Results
Evaluation of DNA variants in estrogen metabolism genes was largely inconclusive. Meta-analyses of data from CYP19A1 support an association between the number of (TTTA) n repeats in intron 4 and breast cancer risk, but the biological mechanism for this relationship is unknown. Associations between single nucleotide polymorphism in CYP1B1 and DNA damage caused by procarcinogenic estrogen metabolites were ambiguous. Variants in CYP2D6 are associated with altered metabolism tamoxifen; however, current data do not support widespread clinical testing. The effect of variants in CYP19A1 in response to aromatase inhibitors is also questionable.
Conclusion
Evaluation of DNA variants in CYP genes involved with estrogen metabolism or treatment response has been inconclusive, reflecting small samples sizes, tumor heterogeneity, and differences between populations. Better-powered studies that account for genetic backgrounds and tumor phenotypes are thus necessary.



Similar content being viewed by others
References
Nebert DW, Russell DW (2002) Clinical importance of the cytochromes P450. Lancet 360(9340):1155–1162
Daly AK (2004) Pharmacogenetics of the cytochromes P450. Curr Top Med Chem 4(16):1733–1744
Aithal GP, Day CP, Kesteven PJ, Daly AK (1999) Association of polymorphisms in the cytochrome P450 CYP2C9 with warfarin dose requirement and risk of bleeding complications. Lancet 353(9154):717–719
Marichalar-Mendia X, Rodrigurez-Tojo MJ, Acha-Sagredo A, Rey-Barja N, Aguirre-Urizar JM (2010) Oral cancer and polymorphisms of ethanol metabolising genes. Oral Oncol 46:9–13
Kristiansen W, Haugen TB, Witczak O, Andersen JM, Fossa SD, Aschim EL (2011) CYP1A1, CYP3A5 and CYP3A7 polymorphisms and testicular cancer susceptibility. Int J Androl 34(1):77–83
Pastina I, Giovannetti E, Chioni A et al (2010) Cytochrome 450 1B1 (CYP1B1) polymorphisms associated with response to docetaxel in castration–resistant prostate cancer (CRPC) patients. BMC Cancer 10:511
Kaaks R, Berrino F, Key T et al (2005) Serum sex steroids in premenopausal women and breast cancer risk within the European Prospective Investigation into Cancer and Nutrition (EPIC). J Natl Cancer Inst 97(10):755–765
Kaaks R, Rinaldi S, Key TJ et al (2005) Postmenopausal serum androgens, oestrogens and breast cancer risk: the European prospective investigation into cancer and nutrition. Endocr Relat Cancer 12(4):1071–1082
Key T, Appleby P, Barnes I, Reeves G, Endogenous Hormones and Breast Cancer Collaborative Group (2002) Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J Natl Cancer Inst 94(8):606–616
Hayashi SI, Eguchi H, Tanimoto K et al (2003) The expression and function of estrogen receptor alpha and beta in human breast cancer and its clinical application. Endocr Relat Cancer 10(2):193–202
Witt BR, Thorneycroft IH (1990) Reproductive steroid hormones: generation, degradation, reception and action. Clin Obstet Gynecol 33(3):563–575
Tsuchiya Y, Nakajima M, Yokoi T (2005) Cytochrome P450-mediated metabolism of estrogens and its regulation in human. Cancer Lett 227(2):115–124
Le Marchand L, Donlon T, Kolonel LN, Henderson BE, Wilkens LR (2005) Estrogen metabolism-related genes and breast cancer risk: the multiethnic cohort study. Cancer Epidemiol Biomark Prev 14(8):1998–2003
Long JR, Egan KM, Dunning L et al (2006) Population-based case–control study of AhR (aryl hydrocarbon receptor) and CYP1A2 polymorphisms and breast cancer risk. Pharmacogenet Genomics 16(4):237–243
Qiu LX, Yao L, Mao C et al (2010) Lack of association of CYP1A2-164 A/C polymorphism with breast cancer susceptibility: a meta-analysis involving 17,600 subjects. Breast Cancer Res Treat 122(2):521–525
Sangrajrang S, Sato Y, Sakamoto H et al (2009) Genetic polymorphisms of estrogen metabolizing enzyme and breast cancer risk in Thai women. Int J Cancer 125(4):837–843
Cribb AE, Joy Knight M, Guemsey J et al (2011) CYP17, catechol-o-methyltransferase, and glutathione transferase M1 genetic polymorphisms, lifestyle factors, and breast cancer risk in women on Prince Edward Island. Breast J 17(1):24–31
Verla-Tebit E, Wang-Gohrke S, Chang-Claude J (2005) CYP17 5′-UTR MspA1 polymorphism and the risk of premenopausal breast cancer in a German population-based case–control study. Breast Cancer Res 7(4):R455–R464
Bergman-Jungestrom M, Gentile M, Lundin AC, Wingren S (1999) Association between CYP17 gene polymorphism and risk of breast cancer in young women. Int J Cancer 84(4):350–353
Feigelson HS, Coetzee GA, Kolonel LN, Ross RK, Henderson BE (1997) A polymorphism in the CYP17 gene increases the risk of breast cancer. Cancer Res 57(6):1063–1065
Justenhoven C, Hamann U, Schubert F et al (2008) Breast cancer: a candidate gene approach across the estrogen metabolic pathway. Breast Cancer Res Treat 108(1):137–149
Travis RC, Churchman M, Edwards SA et al (2012) No association of polymorphisms in CYP17, CYP19, and HSD17-B1 with plasma estradiol concentrations in 1,090 British women. Cancer Epidemiol Biomark Prev 13(12):2282–2284
Chang JH, Gertig DM, Chen X et al (2005) CYP17 genetic polymorphism, breast cancer, and breast cancer risk factors: Australian Breast Cancer Family Study. Breast Cancer Res 7(4):R513–R521
Einarsdottir K, Rylander-Rudqvist T, Humphreys K et al (2005) CYP17 gene polymorphism in relation to breast cancer risk: a case–control study. Breast Cancer Res 7(6):R890–R896
Sliva SN, Cabral MN, Bezerra de Castro G et al (1998) Breast cancer risk and polymorphisms in genes involved in metabolism of estrogens (CYP17, HSD17beta1, COMT and MnSOD): possible protective role of MnSOD gene polymorphism Val/Ala and Ala/Ala in women that never breast fed. J Oncol Rep 16(4):781–788
Helzlsouer KJ, Huang H, Strickland PT et al (1998) Association between CYP17 polymorphisms and development of breast cancer. Cancer Epidemiol Biomark Prev 7:945–949
Agarwal VR, Bulun SE, Leitch M, Rohrich R, Simpson ER (1996) Use of alternative promoters to express the aromatase cytochrome P450 (CYP19) gene in breast adipose tissues of cancer-free and breast cancer patients. J Clin Endocrinol Metab 81(11):3843–3849
Utsumi T, Harada N, Maruta M, Takagi Y (1996) Presence of alternatively spliced transcripts of aromatase gene in human breast cancer. J Clin Endocrinol Metab 81(6):2344–2349
Singh S, Chakravarti D, Edney JA et al (2005) Relative imbalances in the expression of estrogen-metabolizing enzymes in the breast tissue of women with breast carcinoma. Oncol Rep 14(4):1091–1096
Salhab M, Reed MJ, Al Sarakbi W, Jiang WG, Mokbel K (2006) The role of aromatase and 17-beta-hydroxysteroid dehydrogenase type 1 mRNA expression in predicting the clinical outcome of human breast cancer. Breast Cancer Res Treat 99(2):155–162
Okobia MN, Bunker CH, Zmuda JM et al (2006) Simple tandem repeat (TTTA)n polymorphism in CYP19 (aromatase) gene and breast cancer risk in Nigerian women. J Carcinog 5:12
Ribeiro FS, de Amorim LM, de Almeida Simao T, Mendonca GA, de Moura Gallo CV, Pinto LF (2006) CYP19 (TTTA)n polymorphism and breast cancer risk in Brazilian women. Toxicol Lett 164(1):90–95
Han DF, Zhou X, Hu MB et al (2005) Polymorphisms of estrogen-metabolizing genes and breast cancer risk: a multigenic study. Chin Med J 118(18):1507–1516
Baxter SW, Choong DY, Eccles DM, Campbell IG (2001) Polymorphic variation in CYP19 and the risk of breast cancer. Carcinogenesis 22(2):347–349
Miyoshi Y, Iwao K, Ikeda N, Egawa C, Noguchi S (2000) Breast cancer risk associated with polymorphism in CYP19 in Japanese women. Int J Cancer 89(4):325–328
Haiman CA, Hankinson SE, Spiegelman D et al (2000) A tetranucleotide repeat polymorphism in CYP19 and breast cancer risk. Int J Cancer 87(2):204–210
Healey CS, Dunning AM, Durocher F et al (2000) Polymorphisms in the human aromatase cytochrome P450 gene (CYP19) and breast cancer risk. Carcinogenesis 21(2):189–193
Probst-Hensch NM, Ingles SA, Diep AT et al (1999) Aromatase and breast cancer susceptibility. Endocr Relat Cancer 6(2):165–173
Cai Q, Kataoka N, Li C et al (2008) Haplotype analyses of CYP19A1 gene variants and breast cancer risk: results from the Shanghai Breast Cancer Study. Cancer Epidemiol Biomark Prev 17(1):27–32
Kim JY, Lee CS, Kim HO et al (2009) The association between genetic polymorphisms in CYP19 and breast cancer risk in Korean women. Oncol Rep 22(3):487–492
Ma X, Qi X, Chen C et al (2010) Association between CYP19 polymorphisms and breast cancer risk: results from 10,592 cases and 11,720 controls. Breast Cancer Res Treat 122:2–495
Zhang B, Beeghly-Fadiel A, Long J, Zheng W (2011) Genetic variants associated with breast-cancer risk: comprehensive research synopsis, meta-analysis, and epidemiological evidence. Lancet Oncol 12(5):477–488
Olson JE, Ingle JN, Ma CX et al (2007) A comprehensive examination of CYP19 variation and risk of breast cancer using two haplotype tagging approaches. Breast Cancer Res Treat 102(2):237–247
Kristensen VN, Harada N, Yoshimura N et al (2000) Genetic variants of CYP19 (aromatase) and breast cancer risk. Oncogene 1919:1329–1333
Zhang L, Gu L, Qian B et al (2009) Association of genetic polymorphisms of ER-alpha and the estradiol-synthesizing enzyme genes CYP17 and CYP19 with breast cancer risk in Chinese women. Breast Cancer Res Treat 114(2):327–338
Zins K, Mogg M, Schneeberger C, Abraham D, Schreiber M (2014) Analysis of the rs10046 polymorphism of aromatase (CYP19) in premenopausal onset of human breast cancer. Int J Mol Sci 15(1):712–724
Chen C, Sakoda LC, Doherty JA et al (2008) Genetic variation in CYP19A1 and risk of breast cancer and fibrocystic breast conditions among women in Shanghai, China. Cancer Epidemiol Biomark Prev 17(12):3457–3466
Haiman CA, Hankinson SE, Spiegelman D, Brown M, Hunter DJ (2002) No association between a single nucleotide polymorphism in CYP19 and breast cancer risk. Cancer Epidemiol Biomark Prev 11:215–216
Dunning AM, Dowsett M, Healey CS et al (2004) Polymorphisms associated with circulating sex hormone levels in postmenopausal women. J Natl Cancer Inst 96(12):936–945
Haiman CA, Dossus L, Setiawan VW et al (2007) Genetic variation at the CYP19A1 locus predicts circulating estrogen levels but not breast cancer risk in postmenopausal women. Cancer Res 67(5):1893–1897
Pineda B, Garcia-Perez MA, Cano A, Lluch A, Eroles P (2013) Associations between aromatase CYP19 rs10046 polymorphism and breast cancer risk: from a case–control to a meta-analysis of 20,098 subjects. PLoS One 8(1):e53902
Ma CX, Adjei AA, Salavaggione OE et al (2005) Human aromatase: gene resequencing and functional genomics. Cancer Res 65(23):11071–11082
Hirose K, Matsuo K, Toyama T, Iwata H, Hamajima N, Tajima K (2004) The CYP19 gene codon 19 Trp/Arg polymorphism increases breast cancer risk in subsets of premenopausal Japanese. Cancer Epidemiol Biomark Prev 13(8):1407–1411
Lee K-M, Abel J, Ko Y et al (2003) Genetic polymorphisms of cytochrome P450 19 and 1B1, alcohol use, and breast cancer risk in Korean women. Br J Cancer 88:675–678
Cavalieri E, Frenkel K, Liehr JG, Rogan E, Roy D (2000) Chapter 4: estrogens as endogenous genotoxic agents—DNA adducts and mutations. J Natl Cancer Inst Monogr 27:75–93
Liehr JG (2000) Is estradiol a genotoxic mutagenic carcinogen? Endocr Rev 21(1):40–54
Kong LY, Szaniszlo P, Albrecht T, Liehr JG (2012) Frequency and molecular analysis of hprt mutations induced by estradiol in Chinese hamster V79 cells. Int J Oncol 17(6):1141–1149
Fernandez SV, Russo IH, Russo J (2006) Estradiol and its metabolites 4-hydroxyestradiol and 2-hydroxyestradiol induce mutations in human breast epithelial cells. Int J Cancer 118(8):1862–1868
Hontz AE, Li SA, Lingle WL et al (2007) Aurora A and B overexpression and centrosome amplification in early estrogen-induced tumor foci in the Syrian hamster kidney: implications for chromosomal instability, aneuploidy, and neoplasia. Cancer Res 67(7):2957–2963
Li JJ, Weroha J, Lingle WL, Papa D, Salisbury JL, Li SA (2004) Estrogen mediates Aurora-A overexpression, centrosome amplification, chromosomal instability, and breast cancer in female ACI rats. Proc Natl Acad Sci USA 191(52):18123–18128
Stopper H, Schmitt E, Gregor C, Mueller SO, Fischer WH (2003) Increased cell proliferation is associated with genomic instability: elevated micronuclei frequencies in estradiol-treated human ovarian cancer cells. Mutagenesis 18(3):243–247
Cavalieri EL, Stack DE, Devanesan PD et al (1997) Molecular origin of cancer: catechol estrogen-3,4-quinones as endogenous tumor initiators. Proc Natl Acad Sci USA 94(20):10937–10942
Chakravarti D, Mailander PC, Li K-M et al (2001) Evidence that a burst of DNA depurination in SENCAR mouse skin induces error-prone repair and forms mutations in the H-ras gene. Oncogene 20:7945–7953
Demple B, Harrison L (1994) Repair of oxidative damage to DNA: enzymology and biology. Annu Rev Biochem 63:915–948
Djuric Z, Heibrun LK, Simon MS et al (1996) Levels of 5-hydroxymethyl-2′-deoxyuridine in DNA from blood as a marker of breast cancer. Cancer 77(4):691–696
Frenkel K, Karkoszka J, Glassman T et al (1998) Serum autoantibodies recognizing 5-hydroxymethyl-2′-deoxyuridine, an oxidized DNA base, as biomarkers of cancer risk in women. Cancer Epidemiol Biomark Prev 7(1):49–57
Poulsen HE (2005) Oxidative DNA modifications. Exp Toxicol Pathol 57(Suppl 1):161–169
Malins DC, Polissar NL, Gunselman SJ (1996) Progression of human breast cancers to the metastatic state is linked to hydroxyl radical-induced DNA damage. Proc Natl Acad Sci USA 93(6):2557–2563
Yoshie Y, Ohshima H (1998) Synergistic induction of DNA strand breakage by catechol-estrogen and nitric oxide: implications for hormonal carcinogenesis. Free Radic Biol Med 24(2):341–348
Liehr JG, Fang WF, Sirbasku DA, Ari-Ulubelen A (1986) Carcinogenicity of catechol estrogens in Syrian hamsters. J Steroid Biochem 24(1):353–356
Hanna IH, Dawling S, Roodi N, Guengerich FP, Parl FF (2000) Cytochrome P450 1B1 (CYP1B1) pharmacogenetics: association of polymorphisms with functional differences in estrogen hydroxylation activity. Cancer Res 60(13):3440–3444
Bandiera S, Weidlich S, Harth V, Broede P, Ko Y, Friedberg T (2005) Proteasomal degradation of human CYP1B1: effect of the Asn453Ser polymorphism on the post-translational regulation of CYP1B1 expression. Mol Pharmacol 67(2):435–443
Landi MT, Bergen AW, Baccarelli A et al (2005) CYP1A1 and CYP1B1 genotypes, haplotypes, and TCDD-induced gene expression in subjects from Seveso, Italy. Toxicology 207(2):191–202
Matyjasik J, Cybulski C, Masojc B et al (2007) CYP1B1 and predisposition to breast cancer in Poland. Breast Cancer Res Treat 106(3):383–388
Gaudet MM, Chanock S, Lissowska J et al (2006) Genetic variation of Cytochrome P450 1B1 (CYP1B1) and risk of breast cancer among Polish women. Pharmacogenet Genomics 16(8):547–553
Delort L, Satih S, Kwiatkowski F, Bignon YJ, Bernard-Gallon DJ (2010) Evaluation of breast cancer risk in a multigenic model including low penetrance genes involved in xenobiotic and estrogen metabolisms. Nutr Cancer 62(2):243–251
Zheng W, Xie DW, Jin F et al (2000) Genetic polymorphism of cytochrome P450-1B1 and risk of breast cancer. Cancer Epidemiol Biomark Prev 9(2):147–150
Bailey LR, Roodi N, Dupont WD, Parl FF (1998) Association of cytochrome P450 1B1 (CYP1B1) polymorphism with steroid receptor status in breast cancer. Cancer Res 58(22):5038–5041
Paracchini V, Raimondi S, Gram IT et al (2007) Meta- and pooled analyses of the cytochrome P-450 1B1 Val432Leu polymorphism and breast cancer: a HuGE-GSEC review. Am J Epidemiol 165(2):115–125
Wen W, Cai Q, Shu XO et al (2005) Cytochrome P450 1B1 and catechol-O-methyltransferase genetic polymorphisms and breast cancer risk in Chinese women: results from the shanghai breast cancer study and a meta-analysis. Cancer Epidemiol Biomark Prev 14(2):329–335
Yao L, Fang F, Wu Q, Zhong Y, Yu L (2010) No association between CYP1B1 Val432Leu polymorphism and breast cancer risk: a meta-analysis involving 40,303 subjects. Breast Cancer Res Treat 122(1):237–242
Keen JC, Davidson NE (2003) The biology of breast carcinoma. Cancer 97(3 Suppl):825–833
Elledge RM, Fuqua SA (2000) Estrogen and progesterone receptors. In: Harris JR (ed) Diseases of the breast, 2nd edn. Lippencott Williams and Wilkins, Philadelphia, pp 471–488
Kennecke H, Yerushalmi R, Woods R et al (2010) Metastatic behavior of breast cancer subtypes. J Clin Oncol 28(20):3271–3277
Fisher B, Costantino JP, Wickerham DL et al (1998) Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst 90(18):1371–1388
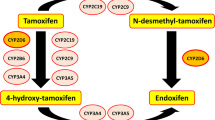
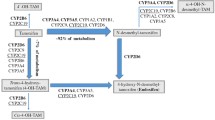
Higgins MJ, Stearns V (2010) CYP2D6 polymorphisms and tamoxifen metabolism: clinical relevance. Curr Oncol Rep 12(1):7–15
Ramon y Cajal T, Altes A, Pare L et al (2010) Impact of CYP2D6 polymorphisms in tamoxifen adjuvant breast cancer treatment. Breast Cancer Res Treat 119(1):33–38
Schroth W, Antoniadou L, Fritz P et al (2007) Breast cancer treatment outcome with adjuvant tamoxifen relative to patient CYP2D6 and CYP2C19 genotypes. J Clin Oncol 25(33):5187–5193
Bonanni B, Macis D, Maisonneuve P et al (2006) Polymorphism in the CYP2D6 tamoxifen-metabolizing gene influences clinical effect but not hot flashes: data from the Italian tamoxifen trial. J Clin Oncol 24(22):3708–3709
Wegman P, Vainikka L, Stal O et al (2005) Genotype of metabolic enzymes and the benefit of tamoxifen in postmenopausal breast cancer patients. Breast Cancer Res 7(3):R284–R290
Goetz MP, Rae JM, Suman VJ et al (2005) Pharmacogenetics of tamoxifen biotransformation is associated with clinical outcomes of efficacy and hot flashes. J Clin Oncol 23(36):9312–9318
Lim HS, Ju Lee H, Seok Lee K, Sook Lee E, Jang IJ, Ro J (2007) Clinical implications of CYP2D6 genotypes predictive of tamoxifen pharmacokinetics in metastatic breast cancer. J Clin Oncol 25(25):3837–3845
Kiyotani K, Mushiroda T, Sasa M et al (2008) Impact of CYP2D6*10 on recurrence-free survival in breast cancer patients receiving adjuvant tamoxifen therapy. Cancer Sci 99(5):995–999
Hertz DL, McLeod HL, Irwin WJ (2012) Tamoxifen and CYP2D6: a contradiction of data. Oncologist 17(5):620–630
Markkula A, Hjertberg M, Rose C, Ingvar C, Jernström H (2014) No association found between CYP2D6 genotype and early breast cancer events in tamoxifen-treated patients. Acta Oncol 53(2):195–200
Nowell SA, Ahn J, Rae JM et al (2005) Association of genetic variation in tamoxifen-metabolizing enzymes with overall survival and recurrence of disease in breast cancer patients. Breast Cancer Res Treat 91(3):249–258
Okishiro M, Taguchi T, Jin Kim S, Shimazu K, Tamaki Y, Noguchi S (2009) Genetic polymorphisms of CYP2D6 10 and CYP2C19 2, 3 are not associated with prognosis, endometrial thickness, or bone mineral density in Japanese breast cancer patients treated with adjuvant tamoxifen. Cancer 115(5):952–961
Seruga B, Amir E (2010) Cytochrome P450 2D6 and outcomes of adjuvant tamoxifen therapy: results of a meta-analysis. Breast Cancer Res Treat 122(3):609–617
Zeng Z, Liu Y, Liu Z et al (2013) CYP2D6 polymorphisms influence tamoxifen treatment outcomes in breast cancer patients: a meta-analysis. Cancer Chemother Pharmacol 72(2):287–303
Province MA, Goetz MP, Brauch H et al (2014) CYP2D6 genotype and adjuvant tamoxifen: meta-analysis of heterogeneous study populations. Clin Pharmacol Ther 95(2):216–227
Jung JA, Lim HS (2014) Association between CYP2D6 genotypes and the clinical outcomes of adjuvant tamoxifen for breast cancer: a meta-analysis. Pharmacogenomics 15(1):49–60
Flockhart DA (2007) Drug interactions: cytochrome P450 drug interaction table
Steams V, Johnson MD, Rae JM, Morocho A, Novielli A, Bhargava P, Hayes DF, Desta Z, Flockhart DA (2003) Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J Natl Cancer Inst 95(23):1758–1764
Jin Y, Desta Z, Steams V et al (2005) CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst 97(1):30–39
Kelly CM, Jurrlink DN, Gomes T et al (2010) Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population based cohort study. BMJ 340:c693
Lash TL, Cronin-Fenton D, Ahern TP et al (2010) Breast cancer recurrence risk related to concurrent use of SSRI antidepressants and tamoxifen. Acta Oncol 49(3):305–312
Holtzman D (2009) Tamoxifen, antidepressants, and CYP2D6: the conundrum continues. J Natl Cancer Inst 101(20):1370–1371
Binkhorst L, Mathijssen RH, van Herk-Sukel MP et al (2013) Unjustified prescribing of CYP2D6 inhibiting SSRIs in women treated with tamoxifen. Breast Cancer Res Treat 139(3):923–929
Jain KK (2005) Applications of AmpliChip CYP450. Mol Diagn 9(3):119–127
Hartman AR, Helft P (2007) The ethics of CYP2D6 testing for patients considering tamoxifen. Breast Cancer Res 9(2):103
Hayes DF, Steams V, Rae J, Flockhart D, Consortium on Breast Cancer Pharmacogenomics (2008) A model citizen? Is tamoxifen more effective than aromatase inhibitors if we pick the right patients? J Natl Cancer Inst 100(9):610–613
Fleeman N, Martin Saborido C, Payne K et al (2011) The clinical effectiveness and cost-effectiveness of genotyping for CYP2D6 for the management of women with breast cancer treated with tamoxifen: a systematic review. Health Technol Assess 15(33):1–102
Regan MM, Leyland-Jones B, Bouzyk M et al (2012) CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the breast international group 1–98 trial. J Natl Cancer Inst 104(6):441–451
Lum DW, Perel P, Hingorani AD, Holmes MV (2013) CYP2D6 genotype and tamoxifen response for breast cancer: a systematic review and meta-analysis. PLoS One 8(10):e76648
Dowsett M (1999) Drug and hormone interactions of aromatase inhibitors. Endocr Relat Cancer 6(2):181–185
Baum M, Budzar AU, Cuzick J et al (2002) Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial. Lancet 359(9324):2131–2139
Breast International Group (BIG) 1–98 Collaborative Group, Thurlimann B, Keshaviah A et al (2005) A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med 353(26):2747–2757
Coombes RC, Hall E, Gibson LJ et al (2004) A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 350(11):1081–1092
Amir E, Seruga B, Niraula S, Carlsson L, Ocana A (2011) Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst 103(17):1299–1309
Colomer R, Monzo M, Tusquets I et al (2008) A single-nucleotide polymorphism in the aromatase gene is associated with the efficacy of the aromatase inhibitor letrozole in advanced breast carcinoma. Clin Cancer Res 14(3):811–816
Garcia-Casado Z, Guerrero-Zotano A, Llombart-Cussac A et al (2010) A polymorphism at the 3′-UTR region of the aromatase gene defines a subgroup of postmenopausal breast cancer patients with poor response to neoadjuvant letrozole. BMC Cancer 10:36
Wang L, Ellsworth KA, Moon I et al (2010) Functional genetic polymorphisms in the aromatase gene CYP19 vary the response of breast cancer patients to neoadjuvant therapy with aromatase inhibitors. Cancer Res 70(1):319–328
Park IH, Lee YS, Lee KS et al (2011) Single nucleotide polymorphisms of CYP19A1 predict clinical outcomes and adverse events associated with letrozole in patients with metastatic breast cancer. Cancer Chemother Pharmacol 68(5):1263–1271
Ferraldeschi R, Amedos M, Hadfield KD et al (2012) Polymorphisms of CYP19A1 and response to aromatase inhibitors in metastatic breast cancer patients. Breast Cancer Res Treat 133(3):1191–1198
Bell DW, Brannigan BW, Matsuo K et al (2008) Increased prevalence of EGFR-mutant lung cancer in women and in East Asian populations: analysis of estrogen-related polymorphisms. Clin Cancer Res 14(13):4079–4084
Weyandt J, Ellsworth RE, Hooke JA, Shriver CD, Ellsworth DL (2008) Environmental chemicals and breast cancer risk—a structural chemistry perspective. Curr Med Chem 15(26):2680–2701
Ahmed A, Shahabuddin S, Sheikh S et al (2010) Endoxifen, a new cornerstone of breast cancer therapy: demonstration of safety, tolerability, and systemic bioavailability in healthy human subjects. Clin Pharmacol Ther 88(6):814–817
Acknowledgments
This research was supported by a grant from the United States Department of Defense (Military Molecular Medicine Initiative MDA W81XWH-05-2-0075, Protocol 01-20006). The opinion and assertions contained herein are the private views of the authors and are not to be construed as official or as representing the views of the Department of the Army or the Department of Defense.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blackburn, H.L., Ellsworth, D.L., Shriver, C.D. et al. Role of cytochrome P450 genes in breast cancer etiology and treatment: effects on estrogen biosynthesis, metabolism, and response to endocrine therapy. Cancer Causes Control 26, 319–332 (2015). https://doi.org/10.1007/s10552-014-0519-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-014-0519-7