Abstract
Purpose
Approximately 20% of breast cancer survivors develop breast cancer-related lymphedema (BCRL), and current therapies are limited. We compared acupuncture (AC) to usual care wait-list control (WL) for treatment of persistent BCRL.
Methods
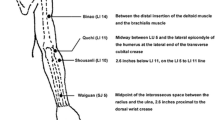
Women with moderate BCRL lasting greater than six months were randomized to AC or WL. AC included twice weekly manual acupuncture over six weeks. We evaluated the difference in circumference and bioimpedance between affected and unaffected arms. Responders were defined as having a decrease in arm circumference difference greater than 30% from baseline. We used analysis of covariance for circumference and bioimpedance measurements and Fisher’s exact to determine the proportion of responders.
Results
Among 82 patients, 73 (89%) were evaluable for the primary endpoint (36 in AC, 37 in WL). 79 (96%) patients received lymphedema treatment before enrolling in our study; 67 (82%) underwent ongoing treatment during the trial. We found no significant difference between groups for arm circumference difference (0.38 cm greater reduction in AC vs. WL, 95% CI − 0.12 to 0.89, p = 0.14) or bioimpedance difference (1.06 greater reduction in AC vs. WL, 95% CI − 5.72 to 7.85, p = 0.8). There was also no difference in the proportion of responders: 17% AC versus 11% WL (6% difference, 95% CI – 10 to 22%, p = 0.5). No severe adverse events were reported.
Conclusions
Our acupuncture protocol appeared to be safe and well tolerated. However, it did not significantly reduce BCRL in pretreated patients receiving concurrent lymphedema treatment. This regimen does not improve upon conventional lymphedema treatment for breast cancer survivors with persistent BCRL.

Similar content being viewed by others
References
Ridner SH (2013) Pathophysiology of lymphedema. Semin Oncol Nurs 29(1):4–11
Chachaj A et al (2010) Physical and psychological impairments of women with upper limb lymphedema following breast cancer treatment. Psychooncology 19(3):299–305
Passik SD, McDonald MV (1998) Psychosocial aspects of upper extremity lymphedema in women treated for breast carcinoma. Cancer 83((12 Suppl American)):2817–2820
Ganz PA (1999) The quality of life after breast cancer—solving the problem of lymphedema. N Engl J Med 340(5):383–385
Ahmed RL et al (2008) Lymphedema and quality of life in breast cancer survivors: the Iowa Women’s Health Study. J Clin Oncol 26(35):5689–5696
McLaughlin SA et al (2008) Prevalence of lymphedema in women with breast cancer 5 years after sentinel lymph node biopsy or axillary dissection: patient perceptions and precautionary behaviors. J Clin Oncol 26(32):5220–5226
DiSipio T et al (2013) Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol 14(6):500–515
McLaughlin SA et al (2008) Prevalence of lymphedema in women with breast cancer 5 years after sentinel lymph node biopsy or axillary dissection: objective measurements. J Clin Oncol 26(32):5213–5219
Paskett ED et al (2012) Cancer-related lymphedema risk factors, diagnosis, treatment, and impact: a review. J Clin Oncol 30(30):3726–3733
Ahmed RL et al (2011) Risk factors for lymphedema in breast cancer survivors, the Iowa Women’s Health Study. Breast Cancer Res Treat 130(3):981–991
Kwan ML et al (2010) Risk factors for lymphedema in a prospective breast cancer survivorship study: the Pathways Study. Arch Surg 145(11):1055–1063
Goldberg JI et al (2010) Morbidity of sentinel node biopsy in breast cancer: the relationship between the number of excised lymph nodes and lymphedema. Ann Surg Oncol 17(12):3278–3286
Hopkins JO et al (2017) Lymphedema, musculoskeletal events and arm function in older patients receiving adjuvant chemotherapy for breast cancer (Alliance A171302). Breast Cancer Res Treat 166(3):793–808
Nguyen TT et al (2017) Breast cancer-related lymphedema risk is related to multidisciplinary treatment and not surgery alone: results from a Large Cohort Study. Ann Surg Oncol 24(10):2972–2980
Shaitelman SF et al (2015) Recent progress in the treatment and prevention of cancer-related lymphedema. CA Cancer J Clin 65(1):55–81
Runowicz CD et al (2016) American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. J Clin Oncol 34(6):611–635
Ezzo J et al (2015) Manual lymphatic drainage for lymphedema following breast cancer treatment. Cochrane Database Syst Rev 5:CD003475
Huang TW et al (2013) Effects of manual lymphatic drainage on breast cancer-related lymphedema: a systematic review and meta-analysis of randomized controlled trials. World J Surg Oncol 11:15
McNeely ML et al (2011) Conservative and dietary interventions for cancer-related lymphedema: a systematic review and meta-analysis. Cancer 117(6):1136–1148
Moseley AL, Carati CJ, Piller NB (2007) A systematic review of common conservative therapies for arm lymphoedema secondary to breast cancer treatment. Ann Oncol 18(4):639–646
Devoogdt N et al (2010) Different physical treatment modalities for lymphoedema developing after axillary lymph node dissection for breast cancer: a review. Eur J Obstet Gynecol Reprod Biol 149(1):3–9
Johansson K et al (1999) Effects of compression bandaging with or without manual lymph drainage treatment in patients with postoperative arm lymphedema. Lymphology 32(3):103–110
McNeely ML et al (2004) The addition of manual lymph drainage to compression therapy for breast cancer related lymphedema: a randomized controlled trial. Breast Cancer Res Treat 86(2):95–106
Javid SH, Anderson BO (2013) Mounting evidence against complex decongestive therapy as a first-line treatment for early lymphedema. J Clin Oncol 31(30):3737–3738
Greenlee H et al (2017) Clinical practice guidelines on the evidence-based use of integrative therapies during and after breast cancer treatment. CA Cancer J Clin 67(3):194–232
Javdan B, Cassileth B (2015) Acupuncture Research at Memorial Sloan Kettering Cancer Center. J Acupunct Meridian Stud 8(3):115–121
Zia FZ et al (2017) The National Cancer Institute’s conference on acupuncture for symptom management in oncology: state of the science, evidence, and research gaps. J Natl Cancer Inst Monogr. https://doi.org/10.1093/jncimonographs/lgx005
Alem M, Gurgel MS (2008) Acupuncture in the rehabilitation of women after breast cancer surgery—a case series. Acupunct Med 26(2):87–93
Kanakura Y et al (2002) Effectiveness of acupuncture and moxibustion treatment for lymphedema following intrapelvic lymph node dissection: a preliminary report. Am J Chin Med 30(1):37–43
Cassileth BR et al (2011) A safety and efficacy pilot study of acupuncture for the treatment of chronic lymphoedema. Acupunct Med 29(3):170–172
Cassileth BR et al (2013) Acupuncture in the treatment of upper-limb lymphedema: results of a pilot study. Cancer 119(13):2455–2461
Cornish BH et al (2001) Early diagnosis of lymphedema using multiple frequency bioimpedance. Lymphology 34(1):2–11
Smith CA, Pirotta M, Kilbreath S (2014) A feasibility study to examine the role of acupuncture to reduce symptoms of lymphoedema after breast cancer: a randomised controlled trial. Acupunct Med 32(5):387–393
Jeong YJ et al (2015) Treatment of lymphedema with saam acupuncture in patients with breast cancer: a pilot study. Med Acupunct 27(3):206–215
Yao C et al (2016) Effects of warm acupuncture on breast cancer-related chronic lymphedema: a randomized controlled trial. Curr Oncol 23(1):e27–e34
Acknowledgements
This study was funded in part by a National Cancer Institute R21CA173263 (Bao) and R01CA158243 (Mao), a Memorial Sloan Kettering Cancer Center P30 Grant (P30-CA008748), and the Byrne Fund and the Translational Research and Integrative Medicine Fund, both at Memorial Sloan Kettering Cancer Center. The funding sources had no involvement in the study design; collection, analysis and interpretation of data; writing of the report; or decision to submit the article for publication.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bao, T., Iris Zhi, W., Vertosick, E.A. et al. Acupuncture for breast cancer-related lymphedema: a randomized controlled trial. Breast Cancer Res Treat 170, 77–87 (2018). https://doi.org/10.1007/s10549-018-4743-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-4743-9