Abstract
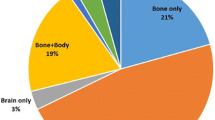
Guidelines do not support utilization of high technology radiologic imaging (HTRI) for surveillance after curative treatment for early stage breast cancer. Surveillance, Epidemiology, and End Results (SEER)-Medicare linked data were used to identify 25,555 women diagnosed with stage I–II breast cancer between 1998 and 2003 who survived ≥48 months from diagnosis without evidence of second primary or recurrent cancer in this interval. HTRI utilization (computerized tomography scanning (CT), bone scan (BS), breast magnetic resonance imaging, and positron emission tomography scans) was measured in months 13–48 post-diagnosis. Cases were individually matched to 75,669 female Medicare enrollees without cancer. Factors associated with HTRI utilization were evaluated. Forty percent of women with stage I–II breast cancer and 25% of controls had ≥1 HTRI during the surveillance interval (P < 0.001). High utilization rates were observed for CT (30%) and BSs (19%). The proportion of women who had a CT during the surveillance period increased in both cancer survivors and controls. Among breast cancer cases age <80, higher comorbidity index, stage II disease, and more recent diagnosis were independently associated with receipt of HTRI. Paralleling patterns observed in controls, HTRI utilization for surveillance following diagnosis of early stage breast cancer has steadily increased among Medicare beneficiaries. Strategies to foster judicious utilization of HTRI should be a priority.

Similar content being viewed by others
References
Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, Mandelblatt JS, Yakovlev AY, Habbema JD, Feuer EJ (2005) Cancer intervention and surveillance modeling network (CISNET) collaborators. Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med 353:1784–1792
Clarke M, Collins R, Darby S, Davies C, Elphinstone P, Evans E, Godwin J, Gray R, Hicks C, James S, MacKinon E, McGale P, McHugh T, Peto R, Taylor C, Wang Y, Early Breast Cancer Collaborative Group (EBCTCG) (2005) Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomized trials. Lancet 366:2087–2106
Ries LAG, Eisner MP, Kosary CL, Hankey BF, Miller BA, Clegg L, Mariotto A, Feuer EJ, Edwards BK (eds) (2011) SEER Cancer Statistics Review, 1975–2002. National Cancer Institute, Bethesda. http://seer.cancer.gov/csr/1975_2002/. Accessed 27 Aug 2011
Rojas M, Telaro E, Russo A, Moschetti I, Coe L, Fossati R, Palli D, del Roselli TM, Liberti A (2005) Follow-up strategies for women treated for early breast cancer. Cochrane Library, Oxford, CD001768, Review
Grunfeld E (2009) Optimizing follow-up after breast cancer treatment. Curr Opin Obstet Gynecol 21:92–96
Rosselli Del Turco M, Palli D, Cariddi A, Ciatto S, Pacini P, Distante V (1994) Intensive diagnostic follow-up after treatment of primary breast cancer: a randomized trial—National Research Council Project on Breast Cancer Follow-up. JAMA 271:1593–1597
Palli D, Russo A, Saieva C, Ciatto S, Rosselli Del Turco M, Distante V, Pacini P (1999) Intensive vs clinical follow-up after treatment of primary breast cancer: 10-year update of a randomized trial. National Research Council Project on Breast Cancer Follow-up. JAMA 281:1586
The GIVIO Investigators (1994) Impact of follow-up testing on survival and health-related quality of life in breast cancer patients: a multicenter randomized controlled trial. JAMA 271:1587–1592
US Preventive Services Task Force (2009) Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med 151:716–726, W-236
National Comprehensive Cancer Network (2000) NCCN Clinical Practice Guidelines in Oncology. Breast Cancer. http://www.nccn.org/professionals/physician_gls/PDF/breast.pdf. Accessed 11 Jun 2010
Khatcheressian JL, Wolff AC, Smith TJ, Grunfeld E, Muss HB, Vogel VG, Halberg F, Somerfield MR, Davidson NE (2006) American Society of Clinical Oncology 2006 update of the breast cancer follow-up and management guidelines in the adjuvant setting. J Clin Oncol 24:5091–5097
Saslow MO, Lehman CD, Morris E, Pisano E, Schnall M, Sener S, Smith RA, Warner E, Yaffe M, Boetes C, Burke W, Harms S, Leach Andrews KS, Russell CA, American Cancer Society Breast Cancer Advisory Group (2007) American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin 57:75–89
Loprinzi CL (1994) It is now the age to define the appropriate follow-up of primary breast cancer patients. J Clin Oncol 12:881–883
Loprinzi CL, Hayes D, Smith T (2000) Doc, shouldn’t we be getting some tests? J Clin Oncol 18:2345–2348
Keating NL, Landrum MB, Guadagnoli E, Winer EP, Ayanian JZ (2007) Surveillance testing among survivors of early-stage breast cancer. J Clin Oncol 25:1074–1081
Report to the Congress: Medicare payment policy. Medicare Payment Advisory Commission. http://www.medpac.gov/documents/Mar06_EntireReport.pdf. Accessed 25 Aug 2011
Dinan MA, Curtis LH, Hammill BG, Patz EF, Abernethy AP, Shea AM, Schulman KA (2010) Changes in the use and costs of diagnostic imaging among Medicare beneficiaries with cancer, 1999–2006. JAMA 303:1625–1631
Brenner DJ, Hall EJ (2007) Computed tomography—an increasing source of radiation exposure. N Engl J Med 357:2277–2284
Scientists say FDA ignored radiation warnings. NY Times 28 Mar 2010
National Cancer Institute. SEER-Medicare: medicare enrollment and claims data. http://www.healthservices.cancer.gov/seermedicare/medicare. Accessed 27 Aug 2010
Warren JL, Klabunde CN, Schrag D, Bach PB, Riley GF (2002) Overview of the SEER-Medicare data: content, research applications, and generalizability to the United States elderly population. Med Care 40 Supplement, pp IV-1–IV-3-18
Potosky AL, Riley GF, Lubitz JD, Mentnech RM, Kessler LG (1993) Potential for cancer related health services research using a linked Medicare-tumor registry database. Med Care 31:732–748
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Romano PS, Roos LL, Jollis JG (1993) Adapting a clinical comorbidity index for use with ICD-9-CM administrative data: differing perspectives. J Clin Epidemiol 46:1075–1079
International Classification of Disease. http://www.icd9data.com/. Accessed 27 Mar 2010
CPT standard edition: current procedural terminology (2007). American Medical Association Press, Chicago
Brennan S, Liberman L, Dershaw DD, Morris E (2010) Breast MRI screening in women with a personal history of breast cancer. Am J Roentgenol 195:510–516
Hillman BJ, Goldsmith JC (2010) The uncritical use of high-tech medical imaging. N Engl J Med 363:4–6
Acknowledgments
The authors have no conflicts of interest to disclose. We acknowledge the efforts of several groups responsible for creation and dissemination of the SEER-Medicare linked database (including the Applied Research Branch, Division of Cancer Control and Population Sciences, National Cancer Institute; the Office of Strategic Planning and the Office of Information Services, Center for Medicare and Medicaid Services; Information Management Series; and the Surveillance, Epidemiology and End Results program tumor registries).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panageas, K.S., Sima, C.S., Liberman, L. et al. Use of high technology imaging for surveillance of early stage breast cancer. Breast Cancer Res Treat 131, 663–670 (2012). https://doi.org/10.1007/s10549-011-1773-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-011-1773-y