Abstract
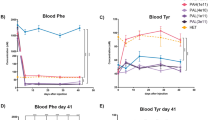
Hyperphenylalaninemia (HPA) caused by hepatic phenylalanine hydroxylase (PAH) deficiency has severe consequences on brain monoamine neurotransmitter metabolism. We have studied monoamine neurotransmitter status and the effect of tetrahydrobiopterin (BH4) treatment in Pahenu1/enu2 (ENU1/2) mice, a model of partial PAH deficiency. These mice exhibit elevated blood L-phenylalanine (L-Phe) concentrations similar to that of mild hyperphenylalaninemia (HPA), but brain levels of L-Phe are still ~5-fold elevated compared to wild-type. We found that brain L-tyrosine, L-tryptophan, BH4 cofactor and catecholamine concentrations, and brain tyrosine hydroxylase (TH) activity were normal in these mice but that brain serotonin, 5-hydroxyindolacetic acid (5HIAA) and 3-methoxy-4-hydroxyphenylglycol (MHPG) content, and brain TH protein, as well as tryptophan hydroxylase type 2 (TPH2) protein levels and activity were reduced in comparison to wild-type mice. Parenteral L-Phe loading conditions did not lead to significant changes in brain neurometabolite concentrations. Remarkably, enteral BH4 treatment, which normalized brain L-Phe levels in ENU1/2 mice, lead to only partial recovery of brain serotonin and 5HIAA concentrations. Furthermore, indirect evidence indicated that the GTP cyclohydrolase I (GTPCH) feedback regulatory protein (GFRP) complex may be a sensor for brain L-Phe elevation to ameliorate the toxic effects of HPA. We conclude that BH4 treatment of HPA toward systemic L-Phe lowering reverses elevated brain L-Phe content but the recovery of TPH2 protein and activity as well as serotonin levels is suboptimal, indicating that patients with mild HPA and mood problems (depression or anxiety) treated with the current diet may benefit from supplementation with BH4 and 5-OH-tryptophan.





Similar content being viewed by others
Abbreviations
- PAH:
-
phenylalanine hydroxylase
- LNAA:
-
large neutral amino acid
- BH4 :
-
6R–L-erythro-5,6,7,8-tetrahydrobiopterin
- GTPCH:
-
GTP cyclohydrolase I
- GFRP:
-
GTPCH feedback regulatory protein (gene Gchfr)
- HPA:
-
hyperphenylalaninemia
- PKU:
-
phenylketonuria
- L-Phe:
-
L-phenylalanine
- TH:
-
tyrosine hydroxylase
- TPH:
-
tryptophan hydroxylase
- L-Dopa:
-
L-3,4-dihydroxyphenylalanine
- 3OMD:
-
3-o-methyldopa
- HVA:
-
homovanillic acid
- 5HTP:
-
5-hydroxytryptophan
- 5HIAA:
-
5-hydroxyindolacetic acid
- MHPG:
-
3-methoxy-4-hydroxyphenylglycol
References
Adler-Abramovich L, Vaks L, Carny O et al (2012) Phenylalanine assembly into toxic fibrils suggests amyloid etiology in phenylketonuria. Nat Chem Biol 8:701–706
Blau N, Thöny B (2008) Pterins and related enzymes. In: Blau N, Duran M, Gibson KM (eds) Laboratory guide to the methods in biochemical genetics. Springer, Berlin, p 665–701
Blau N, van Spronsen FJ, Levy HL (2010) Phenylketonuria. Lancet 376:1417–1427
Calvo AC, Scherer T, Pey AL et al (2010) Effect of pharmacological chaperones on brain tyrosine hydroxylase and tryptophan hydroxylase 2. J Neurochem 114:853–863
Cansev M, Wurtman RJ (2007) Aromatic amino acids in the brain. In: Lajtha A (ed) Handbook of neurochemistry and molecular neurobiology. Springer, Berlin, pp 60–97
Curtius HC, Niederwieser A, Viscontini M et al (1981) Serotonin and dopamine synthesis in phenylketonuria. Adv Exp Med Biol 133:277–291
de Groot MJ, Hoeksma M, Blau N, Reijngoud DJ, van Spronsen FJ (2010) Pathogenesis of cognitive dysfunction in phenylketonuria: review of hypotheses. Mol Genet Metab 99 Suppl 1:S86–S89
Donlon J, Sarkissian CN, Levy HL, Scriver CR (2015) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. Chapter 77. In: Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Stylianos E, Antonarakis MD, Ballabio A, Gibson M, Mitchell G (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York http://ommbid.mhmedical.com/content.aspx?bookid=971§ionid=62673211
Elzaouk L, Leimbacher W, Turri M et al (2003) Dwarfism and low insulin-like growth factor-1 due to dopamine depletion in pts−/− mice rescued by feeding neurotransmitter precursors and H4-biopterin. J Biol Chem 278:28303–28311
Erlandsen H, Pey AL, Gamez A et al (2004) Correction of kinetic and stability defects by tetrahydrobiopterin in phenylketonuria patients with certain phenylalanine hydroxylase mutations. Proc Natl Acad Sci U S A 101:16903–16908
Gersting SW, Kemter KF, Staudigl M et al (2008) Loss of function in phenylketonuria is caused by impaired molecular motions and conformational instability. Am J Hum Genet 83:5–17
Guttler F, Lou H (1986) Dietary problems of phenylketonuria: effect on CNS transmitters and their possible role in behaviour and neuropsychological function. J Inherit Metab Dis 9 Suppl 2:169–177
Harada T, Kagamiyama H, Hatakeyama K (1993) Feedback regulation mechanism for the control of GTP cyclohydrolase I activity. Science 260:1507–1510
Heintz C, Cotton RG, Blau N (2013b) Tetrahydrobiopterin, its mode of action on phenylalanine hydroxylase, and importance of genotypes for pharmacological therapy of phenylketonuria. Hum Mutat 34:927-36
Hoeksma M, Reijngoud DJ, Pruim J, de Valk HW, Paans AM, van Spronsen FJ (2009) Phenylketonuria: high plasma phenylalanine decreases cerebral protein synthesis. Mol Genet Metab 96:177–182
Joseph B, Dyer CA (2003) Relationship between myelin production and dopamine synthesis in the PKU mouse brain. J Neurochem 86:615–626
Kapatos G, Hirayama K, Shimoji M, Milstien S (1999) GTP cyclohydrolase I feedback regulatory protein is expressed in serotonin neurons and regulates tetrahydrobiopterin biosynthesis. J Neurochem 72:669–675
Kolinsky MA, Gross SS (2004) The mechanism of potent GTP cyclohydrolase I inhibition by 2,4-diamino-6-hydroxypyrimidine: requirement of the GTP cyclohydrolase I feedback regulatory protein. J Biol Chem 279:40677–40682
Kure S, Hou DC, Ohura T et al (1999) Tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. J Pediatr 135:375–378
Lagler FB, Gersting SW, Zsifkovits C et al (2010) New insights into tetrahydrobiopterin pharmacodynamics from Pah enu1/2, a mouse model for compound heterozygous tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. Biochem Pharmacol 80:1563–1571
Lichter-Konecki U, Hipke CM, Konecki DS (1999) Human phenylalanine hydroxylase gene expression in kidney and other nonhepatic tissues. Mol Genet Metab 67:308–316
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Lykkelund C, Nielsen JB, Lou HC et al (1988) Increased neurotransmitter biosynthesis in phenylketonuria induced by phenylalanine restriction or by supplementation of unrestricted diet with large amounts of tyrosine. Eur J Pediatr 148:238–245
McKean CM (1972) The effects of high phenylalanine concentrations on serotonin and catecholamine metabolism in the human brain. Brain Res 47:469–476
Moller HE, Ullrich K, Weglage J (2000) In vivo proton magnetic resonance spectroscopy in phenylketonuria. Eur J Pediatr 159 Suppl 2:S121–S125
Ogawa S, Ichinose H (2006) Effect of metals and phenylalanine on the activity of human tryptophan hydroxylase-2: comparison with that on tyrosine hydroxylase activity. Neurosci Lett 401:261–265
Oh HJ, Lee H, Park JW et al (2005) Reversal of gene expression profile in the phenylketonuria mouse model after adeno-associated virus vector-mediated gene therapy. Mol Genet Metab 86 Suppl 1:S124–S132
Park JW, Park ES, Choi EN, Park HY, Jung SC (2009) Altered brain gene expression profiles associated with the pathogenesis of phenylketonuria in a mouse model. Clin Chim Acta 401:90–99
Pascucci T, Andolina D, Mela IL et al (2009) 5-Hydroxytryptophan rescues serotonin response to stress in prefrontal cortex of hyperphenylalaninaemic mice. Int J Neuropsychopharmacol 12:1067–1079
Pascucci T, Giacovazzo G, Andolina D et al (2012) In vivo catecholaminergic metabolism in the medial prefrontal cortex of ENU2 mice: an investigation of the cortical dopamine deficit in phenylketonuria. J Inherit Metab Dis 35:1001–1009
Pascucci T, Ventura R, Puglisi-Allegra S, Cabib S (2002) Deficits in brain serotonin synthesis in a genetic mouse model of phenylketonuria. Neuroreport 13:2561–2564
Pey AL, Stricher F, Serrano L, Martinez A (2007) Predicted effects of missense mutations on native-state stability account for phenotypic outcome in phenylketonuria, a paradigm of misfolding diseases. Am J Hum Genet 81:1006–1024
Pietz J, Kreis R, Rupp A et al (1999) Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J Clin Invest 103:1169–1178
Puglisi-Allegra S, Cabib S, Pascucci T, Ventura R, Cali F, Romano V (2000) Dramatic brain aminergic deficit in a genetic mouse model of phenylketonuria. Neuroreport 11:1361–1364
Rebuffat A, Harding CO, Ding Z, Thony B (2010) Comparison of adeno-associated virus pseudotype 1, 2, and 8 vectors administered by intramuscular injection in the treatment of murine phenylketonuria. Hum Gene Ther 21:463–477
Reinhard JF Jr, Smith GK, Nichol CA (1986) A rapid and sensitive assay for tyrosine-3-monooxygenase based upon the release of 3H2O and adsorption of [3H]-tyrosine by charcoal. Life Sci 39:2185–2189
Sarkissian CN, Boulais DM, McDonald JD, Scriver CR (2000) A heteroallelic mutant mouse model: a new orthologue for human hyperphenylalaninemia. Mol Genet Metab 69:188–194
Sarkissian CN, Ying M, Scherer T, Thöny B, Martinez A (2012) The mechanism of BH4 -responsive hyperphenylalaninemia--as it occurs in the ENU1/2 genetic mouse model. Hum Mutat 33:1464–1473
Shen N, Heintz C, Thiel C, Okun JG, Hoffmann GF, Blau N (2016) Co-expression of phenylalanine hydroxylase variants and effects of interallelic complementation on in vitro enzyme activity and genotype-phenotype correlation. Mol Genet Metab 117:328–335
Smith CB, Kang J (2000) Cerebral protein synthesis in a genetic mouse model of phenylketonuria. Proc Natl Acad Sci U S A 97:11014–11019
Thony B, Calvo AC, Scherer T et al (2008) Tetrahydrobiopterin shows chaperone activity for tyrosine hydroxylase. J Neurochem 106:672–681
van Spronsen FJ, Hoeksma M, Reijngoud DJ (2009) Brain dysfunction in phenylketonuria: is phenylalanine toxicity the only possible cause? J Inherit Metab Dis 32:46–51
Werner ER, Blau N, Thony B (2011) Tetrahydrobiopterin: biochemistry and pathophysiology. Biochem J 438:397–414
Wettstein S, Underhaug J, Perez B et al (2015) Linking genotypes database with locus-specific database and genotype-phenotype correlation in phenylketonuria. Eur J Hum Gen 23:302–309
Winn SR, Scherer T, Thöny B et al (2017) Blood phenylalanine reduction corrects CNS dopamine and serotonin deficiencies and partially improves behavioral performance in adult phenylketonuric mice. Mol Genet Metabol. https://doi.org/10.1016/j.ymgme.2017.10.009
Acknowledgements
We thank Dr. Ernst Werner for helpful discussions and the Institute for Animal Science at the University of Zürich for cooperation. Financial support was given by the Stiftung für wissenschaftliche Forschung University of Zürich (Baumgarten Stiftung to B.T.), the Swiss National Science Foundation (to B.T.) SNF 310030-162547, the NIH R01NS080866 to C.O.H., the Novartis “Stiftung für medizinisch-biologische Forschung” (to B.T.), The Research Council of Norway (Programmes FORNY and FRIMEDBIO to A.M.), and the K. G. Jebsen Centre for Neuropsychiatric Disorders (to A.M.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
T.Scherer, G.Allegri, C. Sarkissian, M. Ying, H.M. Grisch-Chan, A. Rassi, S. Winn, C. Harding, A. Martinez and B. Thöny declare that they have no conflict of interest.
Informed consent
This article does not contain any studies with human subjects performed by the any of the authors.
Animal rights
All institutional and national guidelines for the care and use of laboratory animals were followed (see also chapter “Materials, methods, and animal husbandry”).
Additional information
Communicated by: Viktor Kožich
Electronic supplementary material
Suppl. Fig. S1
Inhibition of GPTCH activity by 2,4,-diamino-6-hydroxypyrimidine (DAHP) in crude brain extracts from either wild-type or ENU2/2 (PKU) mice. Activity was assayed as the quantity of 7,8,-dihydroneopterin triphosphate produced by 80 mg of brain extract from either wild-type or ENU1/2 mice in the presence of increasing amounts of added DAHP. Assays were performed in triplicates. Two tailed Student‘s t-test, ***p < 0.005, **p < 0.05, *p < 0.1; n = 3 (wild-type and PKU) (GIF 10 kb)
Rights and permissions
About this article
Cite this article
Scherer, T., Allegri, G., Sarkissian, C.N. et al. Tetrahydrobiopterin treatment reduces brain L-Phe but only partially improves serotonin in hyperphenylalaninemic ENU1/2 mice. J Inherit Metab Dis 41, 709–718 (2018). https://doi.org/10.1007/s10545-018-0150-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-018-0150-y