Abstract
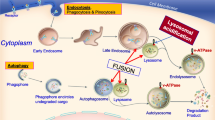
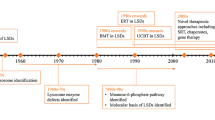
Lysosomal storage diseases (LSD) are inborn errors of metabolism resulting in multisystem disease. Central nervous system involvement, often with progressive neurodegeneration, accounts for a large portion of the morbidity and mortality seen in many LSD. Available treatments fail to prevent or correct neurologic symptoms and decline. Emerging evidence points to an important role for mitochondrial dysfunction in the pathogenesis and progression of LSD-associated neurodegeneration. Mitochondrial dysfunction in LSD is characterized by alterations in mitochondrial mass, morphology and function. Disturbed mitochondrial metabolism in the CNS may lead to excessive production of mitochondrial reactive oxygen species and dysregulated calcium homeostasis. These metabolic disturbances ultimately result in mitochondria-induced apoptosis and neuronal degeneration. Here, we review the current evidence for mitochondrial dysfunction in neuronal models of seven LSD, including GM1-gangliosidosis, mucopolysaccharidosis IIIC, multiple sulfatase deficiency, Krabbe disease, Gaucher disease, Niemann Pick disease type C and the neural ceroid lipofuscinoses and outline current experimental therapies aimed at restoring mitochondrial function and neuroprotection in LSD.

Similar content being viewed by others
References
Alavian KN, Beutner G, Lazrove E et al (2014) An uncoupling channel within the c-subunit ring of the F1FO ATP synthase is the mitochondrial permeability transition pore. Proc Natl Acad Sci U S A 111:10580–10585
Bahat-Stroomza M, Gilgun-Sherki Y, Offen D et al (2005) A novel thiol antioxidant that crosses the blood brain barrier protects dopaminergic neurons in experimental models of Parkinson’s disease. Eur J Neurosci 21:637–646
Bove J, Martinez-Vicente M, Vila M (2011) Fighting neurodegeneration with rapamycin: mechanistic insights. Nat Rev Neurosci 12:437–452
Cali T, Ottolini D, Negro A, Brini M (2012) Alpha-Synuclein controls mitochondrial calcium homeostasis by enhancing endoplasmic reticulum-mitochondria interactions. J Biol Chem 287:17914–17929
Cantuti Castelvetri L, Givogri MI, Hebert A et al (2013) The sphingolipid psychosine inhibits fast axonal transport in Krabbe disease by activation of GSK3beta and deregulation of molecular motors. J Neurosci 33:10048–10056
Cao Y, Staropoli JF, Biswas S et al (2011) Distinct early molecular responses to mutations causing vLINCL and JNCL presage ATP synthase subunit C accumulation in cerebellar cells. PLoS One 6:e17118
Carraro M, Giorgio V, Sileikyte J et al (2014) Channel formation by yeast F-ATP synthase and the role of dimerization in the mitochondrial permeability transition. J Biol Chem 289:15980–15985
Chen S, Owens GC, Crossin KL, Edelman DB (2007) Serotonin stimulates mitochondrial transport in hippocampal neurons. Mol Cell Neurosci 36:472–483
Dasgupta N, Xu YH, Li R et al (2015) Neuronopathic Gaucher disease: dysregulated mRNAs and miRNAs in brain pathogenesis and effects of pharmacologic chaperone treatment in a mouse model. Hum Mol Genet 24:7031–7048
Decressac M, Mattsson B, Weikop P, Lundblad M, Jakobsson J, Bjorklund A (2013) TFEB-mediated autophagy rescues midbrain dopamine neurons from alpha-synuclein toxicity. Proc Natl Acad Sci U S A 110:E1817–E1826
Eldar-Finkelman H, Martinez A (2011) GSK-3 inhibitors: preclinical and clinical focus on CNS. Front Mol Neurosci 4:32
Fernandez A, Llacuna L, Fernandez-Checa JC, Colell A (2009) Mitochondrial cholesterol loading exacerbates amyloid beta peptide-induced inflammation and neurotoxicity. J Neurosci 29:6394–6405
Fossale E, Wolf P, Espinola JA et al (2004) Membrane trafficking and mitochondrial abnormalities precede subunit c deposition in a cerebellar cell model of juvenile neuronal ceroid lipofuscinosis. BMC Neurosci 5:57
Fu R, Wassif CA, Yanjanin NM et al (2013) Efficacy of N-acetylcysteine in phenotypic suppression of mouse models of Niemann-Pick disease, type C1. Hum Mol Genet 22:3508–3523
Futerman AH, Boldin SA, Brann AB, Pelled D, Meivar-Levy I, Zisling R (1999) Regulation of sphingolipid and glycosphingolipid metabolism during neuronal growth and development. Biochem Soc Trans 27:432–437
Giorgio V, von Stockum S, Antoniel M et al (2013) Dimers of mitochondrial ATP synthase form the permeability transition pore. Proc Natl Acad Sci U S A 110:5887–5892
Gorlach A, Bertram K, Hudecova S, Krizanova O (2015) Calcium and ROS: a mutual interplay. Redox Biol 6:260–271
Hamacher-Brady A, Brady NR (2016) Mitophagy programs: mechanisms and physiological implications of mitochondrial targeting by autophagy. Cell Mol Life Sci 73:775–795
Haq E, Giri S, Singh I, Singh AK (2003) Molecular mechanism of psychosine-induced cell death in human oligodendrocyte cell line. J Neurochem 86:1428–1440
Hoglinger D, Haberkant P, Aguilera-Romero A et al (2015) Intracellular sphingosine releases calcium from lysosomes. elife 4:e10616
Hooper C, Killick R, Lovestone S (2008) The GSK3 hypothesis of Alzheimer’s disease. J Neurochem 104:1433–1439
Huang Z, Hou Q, Cheung NS, Li QT (2006) Neuronal cell death caused by inhibition of intracellular cholesterol trafficking is caspase dependent and associated with activation of the mitochondrial apoptosis pathway. J Neurochem 97:280–291
Johnson SC, Yanos ME, Kayser EB et al (2013) mTOR inhibition alleviates mitochondrial disease in a mouse model of Leigh syndrome. Science 342:1524–1528
Karatas H, Aktas Y, Gursoy-Ozdemir Y et al (2009) A nanomedicine transports a peptide caspase-3 inhibitor across the blood-brain barrier and provides neuroprotection. J Neurosci 29:13761–13769
Kennedy BE, LeBlanc VG, Mailman TM et al (2013) Pre-symptomatic activation of antioxidant responses and alterations in glucose and pyruvate metabolism in Niemann-Pick type C1-deficient murine brain. PLoS One 8:e82685
Kiselyov K, Muallem S (2008) Mitochondrial Ca2+ homeostasis in lysosomal storage diseases. Cell Calcium 44:103–111
Kolikova J, Afzalov R, Surin A, Lehesjoki AE, Khiroug L (2011) Deficient mitochondrial ca(2+) buffering in the Cln8(mnd) mouse model of neuronal ceroid lipofuscinosis. Cell Calcium 50:491–501
Liu G, Park SH, Imbesi M et al (2016) Loss of NAD-dependent protein deacetylase Sirtuin-2 alters mitochondrial protein acetylation and dysregulates Mitophagy. Antioxid Redox Signal. doi:10.1089/ars.2016.6662
Lloyd-Evans E, Morgan AJ, He X et al (2008) Niemann-Pick disease type C1 is a sphingosine storage disease that causes deregulation of lysosomal calcium. Nat Med 14:1247–1255
Lojewski X, Staropoli JF, Biswas-Legrand S et al (2014) Human iPSC models of neuronal ceroid lipofuscinosis capture distinct effects of TPP1 and CLN3 mutations on the endocytic pathway. Hum Mol Genet 23:2005–2022
Luiro K, Kopra O, Blom T et al (2006) Batten disease (JNCL) is linked to disturbances in mitochondrial, cytoskeletal, and synaptic compartments. J Neurosci Res 84:1124–1138
Martins C, Hulkova H, Dridi L et al (2015) Neuroinflammation, mitochondrial defects and neurodegeneration in mucopolysaccharidosis III type C mouse model. Brain 138:336–355
Medina DL, Fraldi A, Bouche V et al (2011) Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev Cell 21:421–430
Medina DL, Di Paola S, Peluso I et al (2015) Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB. Nat Cell Biol 17:288–299
Nath S, Goodwin J, Engelborghs Y, Pountney DL (2011) Raised calcium promotes alpha-synuclein aggregate formation. Mol Cell Neurosci 46:516–526
Nunnari J, Suomalainen A (2012) Mitochondria: in sickness and in health. Cell 148:1145–1159
Okada R, Wu Z, Zhu A et al (2015) Cathepsin D deficiency induces oxidative damage in brain pericytes and impairs the blood-brain barrier. Mol Cell Neurosci 64:51–60
Osellame LD, Rahim AA, Hargreaves IP et al (2013) Mitochondria and quality control defects in a mouse model of Gaucher disease--links to Parkinson’s disease. Cell Metab 17:941–953
de Pablo-Latorre R, Saide A, Polishhuck EV, Nusco E, Fraldi A, Ballabio A (2012) Impaired parkin-mediated mitochondrial targeting to autophagosomes differentially contributes to tissue pathology in lysosomal storage diseases. Hum Mol Genet 21:1770–1781
Palmieri M, Pal R, Nelvagal HR et al (2017) mTORC1-independent TFEB activation via Akt inhibition promotes cellular clearance in neurodegenerative storage diseases. Nat Commun 8:14338
Pena-Llopis S, Vega-Rubin-de-Celis S, Schwartz JC et al (2011) Regulation of TFEB and V-ATPases by mTORC1. EMBO J 30:3242–3258
Pickrell AM, Youle RJ (2015) The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron 85:257–273
Plotegher N, Duchen MR (2017) Mitochondrial dysfunction and neurodegeneration in lysosomal storage disorders. Trends Mol Med 23:116–134
Raffaello A, Mammucari C, Gherardi G, Rizzuto R (2016) Calcium at the Center of Cell Signaling: interplay between endoplasmic reticulum, mitochondria, and lysosomes. Trends Biochem Sci 41:1035–1049
Rama Rao KV, Kielian T (2016) Astrocytes and lysosomal storage diseases. Neuroscience 323:195–206
Sano R, Annunziata I, Patterson A et al (2009) GM1-ganglioside accumulation at the mitochondria-associated ER membranes links ER stress to ca(2+)-dependent mitochondrial apoptosis. Mol Cell 36:500–511
Sanz-Blasco S, Valero RA, Rodriguez-Crespo I, Villalobos C, Nunez L (2008) Mitochondrial Ca2+ overload underlies Abeta oligomers neurotoxicity providing an unexpected mechanism of neuroprotection by NSAIDs. PLoS One 3:e2718
Sato S, Koike M, Funayama M et al (2016) Lysosomal storage of subunit c of mitochondrial ATP synthase in brain-specific Atp13a2-deficient mice. Am J Pathol 186(12):3074–3082
Settembre C, Fraldi A, Jahreiss L et al (2008) A block of autophagy in lysosomal storage disorders. Hum Mol Genet 17:119–129
Sidransky E, Nalls MA, Aasly JO et al (2009) Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N Engl J Med 361:1651–1661
Takamura A, Higaki K, Kajimaki K et al (2008) Enhanced autophagy and mitochondrial aberrations in murine G(M1)-gangliosidosis. Biochem Biophys Res Commun 367:616–622
Tang BL (2016) Sirt1 and the mitochondria. Mol Cells 39:87–95
Torres S, Matias N, Baulies A et al (2016) Mitochondrial GSH replenishment as a potential therapeutic approach for Niemann Pick type C disease. Redox Biol 11:60–72
Verity C, Winstone AM, Stellitano L, Will R, Nicoll A (2010) The epidemiology of progressive intellectual and neurological deterioration in childhood. Arch Dis Child 95:361–364
Voccoli V, Tonazzini I, Signore G, Caleo M, Cecchini M (2014) Role of extracellular calcium and mitochondrial oxygen species in psychosine-induced oligodendrocyte cell death. Cell Death Dis 5:e1529
Walkley SU (2004) Secondary accumulation of gangliosides in lysosomal storage disorders. Semin Cell Dev Biol 15:433–444
Wei H, Zhang Z, Saha A et al (2011) Disruption of adaptive energy metabolism and elevated ribosomal p-S6K1 levels contribute to INCL pathogenesis: partial rescue by resveratrol. Hum Mol Genet 20:1111–1121
Xu YH, Xu K, Sun Y et al (2014) Multiple pathogenic proteins implicated in neuronopathic Gaucher disease mice. Hum Mol Genet 23:3943–3957
Yamada T, McGeer PL, Baimbridge KG, McGeer EG (1990) Relative sparing in Parkinson’s disease of substantia nigra dopamine neurons containing calbindin-D28K. Brain Res 526:303–307
Yu W, Gong JS, Ko M, Garver WS, Yanagisawa K, Michikawa M (2005) Altered cholesterol metabolism in Niemann-Pick type C1 mouse brains affects mitochondrial function. J Biol Chem 280:11731–11739
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human or animal subjects performed by the author.
Conflict of interest
None.
Funding
Darius Ebrahimi-Fakhari has received funding from the Daimler and Benz Foundation (Daimler und Benz Stiftung, Ladenburg, Germany) and the Reinhard-Frank Foundation (Reinhard-Frank-Stiftung, Hamburg, Germany) and awards from the Child Neurology Society and the International Parkinson and Movement Disorder Society.
Additional information
Responsible editor: William Ross Wilcox
Rights and permissions
About this article
Cite this article
Saffari, A., Kölker, S., Hoffmann, G.F. et al. Linking mitochondrial dysfunction to neurodegeneration in lysosomal storage diseases. J Inherit Metab Dis 40, 631–640 (2017). https://doi.org/10.1007/s10545-017-0048-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-017-0048-0