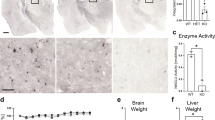
Abstract
Mucopolysaccharidosis (MPS) type IIIA, or Sanfilippo syndrome, is a neurodegenerative lysosomal storage disorder caused by a deficiency of the lysosomal enzyme N-sulfoglucosamine sulfohydrolase (SGSH), involved in the catabolism of heparan sulfate. The clinical spectrum is broad and the age of symptom onset and the degree of preservation of cognitive and motor functions appears greatly influenced by genotype. To explore this further, we generated a conditional knockout (Sgsh KO) mouse model with ubiquitous Sgsh deletion, and compared the clinical and pathological phenotype with that of the spontaneous Sgsh D31N MPS-IIIA mouse model. Phenotypic deficits were noted in Sgsh KO mice prior to Sgsh D31N mice, however these outcomes did not correlate with any shift in the time of appearance nor rate of accumulation of primary (heparan sulfate) or secondary substrates (GM2/GM3 gangliosides). Other disease lesions (elevations in lysosomal integral membrane protein-II expression, reactive astrocytosis and appearance of ubiquitin-positive inclusions) were also comparable between affected mouse strains. This suggests that gross substrate storage and these neuropathological markers are neither primary determinants, nor good biomarkers/indicators of symptom generation, confirming similar observations made recently in MPS-IIIA patients. The Sgsh KO mouse will be a useful tool for elucidation of the neurological basis of disease and assessment of the clinical efficacy of new treatments for Sanfilippo syndrome.




Similar content being viewed by others
References
Arfi A, Richard M, Gandolphe C, Bonnefont-Rousselot D, Therond P, Scherman D (2011) Neuroinflammatory and oxidative stress phenomena in MPS IIIA mouse model: the positive effect of long-term aspirin treatment. Mol Genet Metab 103:18–25
Bhaumik M, Muller VJ, Rozaklis T et al (1999) A mouse model for mucopolysaccharidosis type III a (Sanfilippo syndrome). Glycobiology 9:1389–1396
Crawley AC, Gliddon BL, Auclair D et al (2006) Characterization of a C57BL/6 congenic mouse strain of mucopolysaccharidosis type IIIA. Brain Res 1104:1–17
Elrick MJ, Pacheco CD, Yu T et al (2010) Conditional Niemann-Pick C mice demonstrate cell autonomous Purkinje cell neurodegeneration. Hum Mol Genet 19:837–847
Fraldi A, Hemsley K, Crawley A et al (2007) Functional correction of CNS lesions in a MPS-IIIA mouse model by intracerebral AAV-mediated delivery of sulfamidase and SUMF1 genes. Hum Mol Genet 16:2693–2702
Giulian D, Woodward J, Young DG, Krebs JF, Lachman LB (1988) Interleukin-1 injected into mammalian brain stimulates astrogliosis and neovascularization. J Neurosci 8:2485–2490
Griffey MA, Wozniak D, Wong M et al (2006) CNS-directed AAV2-mediated gene therapy ameliorates functional deficits in a murine model of infantile neuronal ceroid lipofuscinosis. Mol Ther 13:538–547
Haurigot V, Marco S, Ribera A et al (2013) Whole body correction of mucopolysaccharidosis IIIA by intracerebrospinal fluid gene therapy. J Clin Invest 123:3254–3271
Hemsley KM, Beard H, King BM, Hopwood JJ (2008) Effect of high dose, repeated intra-CSF injection of sulphamidase on neuropathology in MPS IIIA mice. Genes Brain Behav 7:740–753
Hemsley KM, Hopwood JJ (2005) Development of motor deficits in a murine model of mucopolysaccharidosis type IIIA (MPS-IIIA). Behav Brain Res 158:191–199
Hemsley KM, Luck AJ, Crawley AC et al (2009) Examination of intravenous and intra-CSF protein delivery for treatment of neurological disease. Eur J Neurosci 29:1197–1214
Hopwood JJ, Elliott H (1982) Diagnosis of Sanfilippo a syndrome by estimation of sulphamidase activity using a radiolabelled tetrasaccharide substrate. Clin Chim Acta 123:241–250
Keating DJ, Winter MA, Hemsley KM et al (2012) Exocytosis is impaired in mucopolysaccharidosis IIIA mouse chromaffin cells. Neuroscience 227:110–118
Lau AA, Crawley AC, Hopwood JJ, Hemsley KM (2008) Open field locomotor activity and anxiety-related behaviors in mucopolysaccharidosis type IIIA mice. Behav Brain Res 91:130–136
Lau AA, Hopwood JJ, Kremer EJ, Hemsley KM (2010) SGSH gene transfer in mucopolysaccharidosis type IIIA mice using canine adenovirus vectors. Mol Genet Metab 100:168–175
Lau AA, Shamsani NJ, Winner LK et al (2013) Neonatal bone marrow transplantation in MPS IIIA mice. JIMD reports 8:121–132
Marco S, Pujol A, Roca C, et al (2016) Progressive neurologic and somatic disease in a novel model of human mucopolysaccharidosis type IIIC. Dis Model Mech doi:10.1242/dmm.025171
Marshall NR, Hassiotis S, King B et al (2015) Delivery of therapeutic protein for prevention of neurodegenerative changes: comparison of different CSF-delivery methods. Exp Neurol 263:79–90
Martins C, Hulkova H, Dridi L et al (2015) Neuroinflammation, mitochondrial defects and neurodegeneration in mucopolysaccharidosis III type C mouse model. Brain 138:336–355
Matern D, Gavrilov D, Oglesbee D, Raymond K, Rinaldo P, Tortorelli S (2015) Newborn screening for lysosomal storage disorders. Semin Perinatol 39:206–216
Russell C (2003) Transgenic expression of human alpha-L-iduronidase in mouse and characterization of the long term pathophysiology of murine alpha-L-iduronidase deficiency. In: Transgenic expression of human alpha-L-iduronidase in mouse and characterization of the long term pathophysiology of murine alpha-L-iduronidase deficiency. University of British Colombia, p 177
Savas PS, Hemsley KM, Hopwood JJ (2004) Intracerebral injection of sulfamidase delays neuropathology in murine MPS-IIIA. Mol Genet Metab 82:273–285
Sergijenko A, Langford-Smith A, Liao AY et al (2013) Myeloid/microglial driven autologous hematopoietic stem cell gene therapy corrects a neuronopathic lysosomal disease. Mol Ther 21:1938–1949
Shapiro EG, Nestrasil I, Delaney KA et al (2016) A prospective natural history study of mucopolysaccharidosis type IIIA. J Pediatr 170(278–287):e274
Sorrentino NC, D’Orsi L, Sambri I et al (2013) A highly secreted sulphamidase engineered to cross the blood-brain barrier corrects brain lesions of mice with mucopolysaccharidoses type IIIA. EMBO molecular medicine 5:675–690
Trim PJ, Hopwood JJ, Snel MF (2015) Butanolysis derivatization: improved sensitivity in LC-MS/MS quantitation of heparan sulfate in urine from Mucopolysaccharidosis patients. Anal Chem 87:9243–9250
Valstar MJ, Marchal JP, Grootenhuis M, Colland V, Wijburg FA (2011) Cognitive development in patients with mucopolysaccharidosis type III (Sanfilippo syndrome). Orphanet journal of rare diseases 6:43
Valstar MJ, Neijs S, Bruggenwirth HT et al (2010) Mucopolysaccharidosis type IIIA: clinical spectrum and genotype-phenotype correlations. Ann Neurol 68:876–887
Valstar MJ, Ruijter GJ, van Diggelen OP, Poorthuis BJ, Wijburg FA (2008) Sanfilippo syndrome: a mini-review. J Inherit Metab Dis 31:240–252
Van Hove JL, Wevers RA, Van Cleemput J et al (2003) Late-onset visceral presentation with cardiomyopathy and without neurological symptoms of adult Sanfilippo a syndrome. Am J Med Genet A 118:382–387
Villani GR, Gargiulo N, Faraonio R, Castaldo S, Gonzalez YRE, Di Natale P (2007) Cytokines, neurotrophins, and oxidative stress in brain disease from mucopolysaccharidosis IIIB. J Neurosci Res 85:612–622
Vitry S, Ausseil J, Hocquemiller M, Bigou S, Dos Santos CR, Heard JM (2009) Enhanced degradation of synaptophysin by the proteasome in mucopolysaccharidosis type IIIB. Mol Cell Neurosci 41:8–18
Whyte L, Hopwood J, Hemsley K, Lau A (2015) Variables influencing fluorimetric N-sulfoglucosamine sulfohydrolase (SGSH) activity measurement in brain homogenates. Molecular Genetics and Metabolism Reports 5:60–62
Wilkinson FL, Holley RJ, Langford-Smith KJ et al (2012) Neuropathology in mouse models of mucopolysaccharidosis type I, IIIA and IIIB. PLoS One 7:e35787
Acknowledgements
This work was supported by the Lysosomal Diseases Research Unit (SAHMRI, Australia). The Monash Gene Targeting Facility was contracted to generate the targeting construct and mice containing the targeted allele. We thank Meghan Setford and Andrew Shoubridge for assistance in genotyping and mouse husbandry; Lynn Garrard and staff at the Women’s and Children’s Health Network Animal Care Facility for care of the mice; Dr. Mark Corbett (Adelaide Neurogenetics Research Group; The University of Adelaide) for providing the CMV-Cre mice, Dr. Mark Adams (Evolutionary Biology Unit, South Australian Museum) for the allozyme electrophoresis and the Australian Genome Research Facility Ltd. for SNP genotyping.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest statement
None.
Animal rights
All institutional and national guidelines for the care and use of laboratory animals were followed.
Informed consent
This article does not contain any studies with human subjects performed by any of the authors.
Funding details
This work was supported by the Lysosomal Diseases Research Unit (SAHMRI, Australia).
Additional information
Communicated by: Carla E. Hollak
Electronic supplementary material
ESM 1
(DOCX 608 kb)
Rights and permissions
About this article
Cite this article
Lau, A.A., King, B.M., Thorsen, C.L. et al. A novel conditional Sgsh knockout mouse model recapitulates phenotypic and neuropathic deficits of Sanfilippo syndrome. J Inherit Metab Dis 40, 715–724 (2017). https://doi.org/10.1007/s10545-017-0044-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-017-0044-4