Abstract
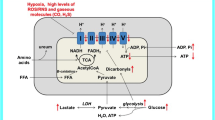
The mitochondrial pyruvate oxidation route is a tightly regulated process, which is essential for aerobic cellular energy production. Disruption of this pathway may lead to severe neurometabolic disorders with onset in early childhood. A frequent finding in these patients is acute and chronic lactic acidemia, which is caused by increased conversion of pyruvate via the enzyme lactate dehydrogenase. Under stable clinical conditions, this process may remain well compensated and does not require specific therapy. However, especially in situations with altered energy demands, such as febrile infections or longer periods of fasting, children with mitochondrial disorders have a high risk of metabolic decompensation with exacerbation of hyperlactatemia and severe metabolic acidosis. Unfortunately, no controlled studies regarding therapy of this critical condition are available and clinical outcome is often unfavorable. Therefore, the aim of this review was to formulate expert-based suggestions for treatment of these patients, including dietary recommendations, buffering strategies and specific drug therapy. However, it is important to keep in mind that a specific therapy for the underlying metabolic cause in children with mitochondrial diseases is usually not available and symptomatic therapy especially of severe lactic acidosis has its ethical limitations.

Similar content being viewed by others
References
Abdelmalak M, Lew A, Ramezani R et al (2013) Long-term safety of dichloroacetate in congenital lactic acidosis. Mol Genet Metab 109(2):139–143
Ahmad A, Kahler SG, Kishnani PS et al (1999) Treatment of pyruvate carboxylase deficiency with high doses of citrate and aspartate. Am J Med Genet 87(4):331–338
Andrade OV, Ihara FO, Troster EJ (2007) Metabolic acidosis in childhood: why, when and how to treat. J Pediatr (Rio J) 83(2 Suppl):S11–S21
Arieff AI (1994) Dialysis disequilibrium syndrome: current concepts on pathogenesis and prevention. Kidney Int 45(3):629–635
Arnon S, Litmanovits I, Regev R, Elpeleg O, Dolfin T (2001) Dichloroacetate treatment for severe refractory metabolic acidosis during neonatal sepsis. Pediatr Infect Dis J 20(2):218–219
Aschner JL, Poland RL (2008) Sodium bicarbonate: basically useless therapy. Pediatrics 122(4):831–835
Baertling F, Rodenburg RJ, Schaper J et al (2014) A guide to diagnosis and treatment of Leigh syndrome. J Neurol Neurosurg Psychiatry 85(3):257–265
Basu RK, Wheeler DS, Goldstein S, Doughty L (2011) Acute renal replacement therapy in pediatrics. Int J Nephrol 2011:785392
Berendzen K, Theriaque DW, Shuster J, Stacpoole PW (2006) Therapeutic potential of dichloroacetate for pyruvate dehydrogenase complex deficiency. Mitochondrion 6(3):126–135
Berg CS, Barnette AR, Myers BJ, Shimony MK, Barton AW, Inder TE (2010) Sodium bicarbonate administration and outcome in preterm infants. J Pediatr 157(4):684–687
Berthet C, Lei H, Thevenet J, Gruetter R, Magistretti PJ, Hirt L (2009) Neuroprotective role of lactate after cerebral ischemia. J Cereb Blood Flow Metab 29(11):1780–1789
Beveridge CJ, Wilkinson AR (2006) Sodium bicarbonate infusion during resuscitation of infants at birth. Cochrane Database Syst Rev 1:CD004864
Bousounis DP, Camfield PR, Wolf B (1993) Reversal of brain atrophy with biotin treatment in biotinidase deficiency. Neuropediatrics 24(4):214–217
Bridges BC, Askenazi DJ, Smith J, Goldstein SL (2012) Pediatric renal replacement therapy in the intensive care unit. Blood Purif 34(2):138–148
Cuthbert C, Alberti KG (1978) Acidemia and insulin resistance in the diabetic ketoacidotic rat. Metabolism 27(12 Suppl 2):1903–1916
Debray FG, Lambert M, Chevalier I et al (2007) Long-term outcome and clinical spectrum of 73 pediatric patients with mitochondrial diseases. Pediatrics 119(4):722–733
Distelmaier F, Huppke P, Pieperhoff P, et al (2013) Biotin-responsive basal ganglia disease: a treatable differential diagnosis of leigh syndrome. JIMD reports
Enns GM, Bennett MJ, Hoppel CL et al (2000) Mitochondrial respiratory chain complex I deficiency with clinical and biochemical features of long-chain 3-hydroxyacyl-coenzyme A dehydrogenase deficiency. J Pediatr 136(2):251–254
Felig P (1973) The glucose-alanine cycle. Metabolism 22(2):179–207
Ferriero R, Manco G, Lamantea E et al (2013) Phenylbutyrate therapy for pyruvate dehydrogenase complex deficiency and lactic acidosis. Sci Transl Med 5(175):175ra131
Gehlbach BK, Schmidt GA (2004) Bench-to-bedside review: treating acid-base abnormalities in the intensive care unit - the role of buffers. Crit Care 8(4):259–265
Gerards M, van den Bosch BJ, Danhauser K et al (2011) Riboflavin-responsive oxidative phosphorylation complex I deficiency caused by defective ACAD9: new function for an old gene. Brain 134(Pt 1):210–219
Glaser N, Barnett P, McCaslin I et al (2001) Risk factors for cerebral edema in children with diabetic ketoacidosis. The pediatric emergency medicine collaborative research committee of the American academy of pediatrics. N Engl J Med 344(4):264–269
Halperin ML, Cheema-Dhadli S, Halperin FA, Kamel KS (1994) Rationale for the use of sodium bicarbonate in a patient with lactic acidosis due to a poor cardiac output. Nephron 66(3):258–261
Hatherill M, Waggie Z, Purves L, Reynolds L, Argent A (2003) Mortality and the nature of metabolic acidosis in children with shock. Intensive Care Med 29(2):286–291
Herrera L, Kazemi H (1980) CSF bicarbonate regulation in metabolic acidosis: role of HCO3- formation in CNS. J Appl Physiol Respir Environ Exerc Physiol 49(5):778–783
Holmdahl MH, Wiklund L, Wetterberg T et al (2000) The place of THAM in the management of acidemia in clinical practice. Acta Anaesthesiol Scand 44(5):524–527
Honzik T, Tesarova M, Mayr JA et al (2010) Mitochondrial encephalocardio-myopathy with early neonatal onset due to TMEM70 mutation. Arch Dis Child 95(4):296–301
Huang YG, Wong KC, Yip WH, McJames SW, Pace NL (1995) Cardiovascular responses to graded doses of three catecholamines during lactic and hydrochloric acidosis in dogs. Br J Anaesth 74(5):583–590
Infante JP, Huszagh VA (2000) Secondary carnitine deficiency and impaired docosahexaenoic (22:6n-3) acid synthesis: a common denominator in the pathophysiology of diseases of oxidative phosphorylation and beta-oxidation. FEBS Lett 468(1):1–5
Jacobs SE, Berg M, Hunt R, Tarnow-Mordi WO, Inder TE, Davis PG (2013) Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev 1:CD003311
Kaufmann P, Engelstad K, Wei Y et al (2006) Dichloroacetate causes toxic neuropathy in MELAS: a randomized, controlled clinical trial. Neurology 66(3):324–330
Kecskes ZB, Davies MW (2002) Rapid correction of early metabolic acidaemia in comparison with placebo, no intervention or slow correction in LBW infants. Cochrane Database Syst Rev 1:CD002976
Kellum JA, Song M, Li J (2004a) Science review: extracellular acidosis and the immune response: clinical and physiologic implications. Crit Care 8(5):331–336
Kellum JA, Song M, Venkataraman R (2004b) Effects of hyperchloremic acidosis on arterial pressure and circulating inflammatory molecules in experimental sepsis. Chest 125(1):243–248
Kennedy AC, Linton AL, Eaton JC (1962) Urea levels in cerebrospinal fluid after haemodialysis. Lancet 1(7226):410–411
Klepper J, Leiendecker B, Bredahl R et al (2002) Introduction of a ketogenic diet in young infants. J Inherit Metab Dis 25(6):449–460
Koene S, Rodenburg RJ, van der Knaap MS et al (2012) Natural disease course and genotype-phenotype correlations in Complex I deficiency caused by nuclear gene defects: what we learned from 130 cases. J Inherit Metab Dis 35(5):737–747
Koopman WJ, Willems PH, Smeitink JA (2012) Monogenic mitochondrial disorders. N Engl J Med 366(12):1132–1141
Kowalchuk JM, Maltais SA, Yamaji K, Hughson RL (1989) The effect of citrate loading on exercise performance, acid-base balance and metabolism. Eur J Appl Physiol Occup Physiol 58(8):858–864
Kraut JA, Madias NE (2012) Treatment of acute metabolic acidosis: a pathophysiologic approach. Nat Rev Nephrol 8(10):589–601
Kurlemann G, Paetzke I, Moller H et al (1995) Therapy of complex I deficiency: peripheral neuropathy during dichloroacetate therapy. Eur J Pediatr 154(11):928–932
Lardner A (2001) The effects of extracellular pH on immune function. J Leukoc Biol 69(4):522–530
Leary SC, Antonicka H, Sasarman F et al (2013) Novel Mutations in SCO1 as a cause of fatal infantile encephalopathy and lactic acidosis. Hum Mutat 34(10):1366–1370
Lim S (2007) Metabolic acidosis. Acta Med Indones 39(3):145–150
Manzanares W, Hardy G (2011) Thiamine supplementation in the critically ill. Curr Opin Clin Nutr Metab Care 14(6):610–617
Mayr JA, Freisinger P, Schlachter K et al (2011) Thiamine pyrophosphokinase deficiency in encephalopathic children with defects in the pyruvate oxidation pathway. Am J Hum Genet 89(6):806–812
McBryde KD, Kershaw DB, Bunchman TE et al (2006) Renal replacement therapy in the treatment of confirmed or suspected inborn errors of metabolism. J Pediatr 148(6):770–778
McNaughton LR (1990) Sodium citrate and anaerobic performance: implications of dosage. Eur J Appl Physiol Occup Physiol 61(5–6):392–397
Mitchell JH, Wildenthal K, Johnson RL Jr (1972) The effects of acid-base disturbances on cardiovascular and pulmonary function. Kidney Int 1(5):375–389
Mizock BA (1989) Lactic acidosis. Dis Mon 35(4):233–300
Mochel F, DeLonlay P, Touati G et al (2005) Pyruvate carboxylase deficiency: clinical and biochemical response to anaplerotic diet therapy. Mol Genet Metab 84(4):305–312
Nahas GG, Sutin KM, Fermon C et al (1998) Guidelines for the treatment of acidaemia with THAM. Drugs 55(2):191–224
O’Regan RG, Majcherczyk S (1982) Role of peripheral chemoreceptors and central chemosensitivity in the regulation of respiration and circulation. J Exp Biol 100:23–40
Orchard CH, Cingolani HE (1994) Acidosis and arrhythmias in cardiac muscle. Cardiovasc Res 28(9):1312–1319
Orchard CH, Kentish JC (1990) Effects of changes of pH on the contractile function of cardiac muscle. Am J Physiol 258(6 Pt 1):C967–C981
Parikh S, Saneto R, Falk MJ, Anselm I, Cohen BH, Haas R, Medicine Society TM (2009) A modern approach to the treatment of mitochondrial disease. Curr Treat Options Neurol 11(6):414–30
Parker MJ, Parshuram CS (2013) Sodium bicarbonate use in shock and cardiac arrest: attitudes of pediatric acute care physicians*. Crit Care Med 41(9):2188–2195
Patel MS, Korotchkina LG (2006) Regulation of the pyruvate dehydrogenase complex. Biochem Soc Trans 34(Pt 2):217–222
Pfeffer G, Majamaa K, Turnbull DM, Thorburn D, Chinnery PF (2012) Treatment for mitochondrial disorders. Cochrane Database Syst Rev 4:CD004426
Pfeffer G, Horvath R, Klopstock T et al (2013) New treatments for mitochondrial disease-no time to drop our standards. Nat Rev Neurol 9(8):474–481
Prietsch V, Lindner M, Zschocke J, Nyhan WL, Hoffmann GF (2002) Emergency management of inherited metabolic diseases. J Inherit Metab Dis 25(7):531–546
Rango M, Arighi A, Bonifati C, Del Bo R, Comi G, Bresolin N (2014) The brain is hypothermic in patients with mitochondrial diseases. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 34(5):915–920
Roberton NR (1970) Apnoea after THAM administration in the newborn. Arch Dis Child 45(240):206–214
Robinson BH (2006) Lactic acidemia and mitochondrial disease. Mol Genet Metab 89(1–2):3–13
Roe CR, Mochel F (2006) Anaplerotic diet therapy in inherited metabolic disease: therapeutic potential. J Inherit Metab Dis 29(2–3):332–340
Rotig A (2014) Genetics of mitochondrial respiratory chain deficiencies. Revue neurologique
Rotig A, Appelkvist EL, Geromel V et al (2000) Quinone-responsive multiple respiratory-chain dysfunction due to widespread coenzyme Q10 deficiency. Lancet 356(9227):391–395
Sabatini S, Kurtzman NA (2009) Bicarbonate therapy in severe metabolic acidosis. J Am Soc Nephrol 20(4):692–695
Sato Y, Mutoh H, Suzuki M, Takekuma Y, Iseki K, Sugawara M (2013) Emulsification using highly hydrophilic surfactants improves the absorption of orally administered coenzyme Q10. Biol Pharm Bull 36(12):2012–2017
Schaefer AM, McFarland R, Blakely EL et al (2008) Prevalence of mitochondrial DNA disease in adults. Ann Neurol 63(1):35–39
Sheridan RL, Yu YM, Prelack K, Young VR, Burke JF, Tompkins RG (1998) Maximal parenteral glucose oxidation in hypermetabolic young children: a stable isotope study. JPEN J Parenter Enteral Nutr 22(4):212–216
Stacpoole PW (1989) The pharmacology of dichloroacetate. Metab Clin Exp 38(11):1124–1144
Stacpoole PW, Gilbert LR, Neiberger RE et al (2008a) Evaluation of long-term treatment of children with congenital lactic acidosis with dichloroacetate. Pediatrics 121(5):e1223–e1228
Stacpoole PW, Kurtz TL, Han Z, Langaee T (2008b) Role of dichloroacetate in the treatment of genetic mitochondrial diseases. Adv Drug Deliv Rev 60(13–14):1478–1487
Staub F, Winkler A, Haberstok J et al (1996) Swelling, intracellular acidosis, and damage of glial cells. Acta Neurochir Suppl 66:56–62
Tarnopolsky MA, Raha S (2005) Mitochondrial myopathies: diagnosis, exercise intolerance, and treatment options. Med Sci Sports Exerc 37(12):2086–2093
Thorburn DR (2004) Mitochondrial disorders: prevalence, myths and advances. J Inherit Metab Dis 27(3):349–362
Tsai IJ, Hwu WL, Huang SC et al (2014) Efficacy and safety of intermittent hemodialysis in infants and young children with inborn errors of metabolism. Pediatr Nephrol 29(1):111–116
Walker BG, Phear DN, Martin FI, Baird CW (1963) Inhibition of insulin by acidosis. Lancet 2(7315):964–965
Wexler ID, Hemalatha SG, McConnell J et al (1997) Outcome of pyruvate dehydrogenase deficiency treated with ketogenic diets. Studies in patients with identical mutations. Neurology 49(6):1655–1661
Wolf B (2012) Biotinidase deficiency: “if you have to have an inherited metabolic disease, this is the one to have”. Genet Med Off J Am Coll Med Genet 14(6):565–575
Wyss MT, Jolivet R, Buck A, Magistretti PJ, Weber B (2011) In vivo evidence for lactate as a neuronal energy source. J Neurosci 31(20):7477–7485
Funding source
This project was supported by the BMBF funded German Network for Mitochondrial Disorders (mitoNET #01GM1113C).
Conflict of interest
Katharina Danhauser, Peter Freisinger, Wolfgang Sperl, Hemmen Sabir, Dirk Klee, Berit Hadzik, Ertan Mayatepek, Eva Morava and Felix Distelmaier declare that they have no conflict of interest. Jan Smeitink is founder and CEO of Khondrion, a university spin-off company of the Radboud University Medical Centre, Nijmegen, The Netherlands.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Verena Peters
Rights and permissions
About this article
Cite this article
Danhauser, K., Smeitink, J.A.M., Freisinger, P. et al. Treatment options for lactic acidosis and metabolic crisis in children with mitochondrial disease. J Inherit Metab Dis 38, 467–475 (2015). https://doi.org/10.1007/s10545-014-9796-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9796-2