Summary
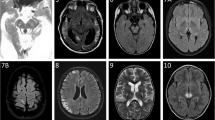
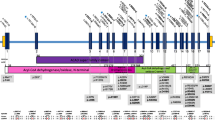
We report a 5-year-old child carrying polymerase gamma (POLG1) mutations, but strikingly normal oxidative phosphorylation analysis in muscle, fibroblasts and liver. Mutations in POLG1 have so far been described in children with severe combined oxidative phosphorylation (OXPHOS) deficiencies and with the classical Alpers–Huttenlocher syndrome. The patient presented with a delayed psychomotor development and ataxia during the first two years of life. From the third year of life he developed epilepsy and regression in development, together with symptoms of visual impairment and sensorineuronal deafness. Cerebrospinal fluid showed elevated lactic acid and protein concentrations. An elder brother had died due to combined OXPHOS deficiencies. Despite the clinical similarity with the elder brother, except for liver involvement, the OXPHOS system analysis in a frozen muscle biopsy was normal. For this reason a fresh muscle biopsy was performed, which has the advantage of the possibility of measuring the substrate oxidation rates and ATP production, part of the mitochondrial energy-generating system (MEGS). During the same session, biopsies of liver and fibroblasts were taken. These three tissues showed normal measurements of the MEGS capacity. Based on the phenotype of Alpers–Huttenlocher syndrome in the elder brother, we decided to screen the POLG1 gene. Mutation analysis showed compound heterozygosity with two known mutations, A467T and G848S. The normal MEGS capacity in this patient expands the already existing complexity and heterogeneity of the childhood POLG1 patients and, on the basis of the high frequency of POLG1 mutations in childhood, warrants a liberal strategy with respect to mutation analysis.
Similar content being viewed by others
Abbreviations
- ATP:
-
adenosine triphosphate
- COX:
-
cytochrome C oxidase
- CS:
-
citrate synthase
- CSF:
-
cerebrospinal fluid
- ECG:
-
electrocardiography
- EEG:
-
electroencephalography
- EPC:
-
epilepsia partialis continua
- MEGS:
-
mitochondrial energy-generating system
- MRI:
-
magnetic resonance imaging
- MRS:
-
magnetic resonance spectroscopy
- mtDNA:
-
mitochondrial DNA
- OXPHOS:
-
oxidative phosphorylation system
- PDHc:
-
pyruvate dehydrogenase complex
- POLG:
-
polymerase gamma
References
Davidzon G, Mancuso M, Ferraris S, et al (2005) POLG mutations and Alpers syndrome. Ann Neurol 57: 921–924.
De Vries MC, Rodenburg RJ, Morava E, et al (2007) Multiple oxidative phosphorylation deficiencies in severe childhood multi-system disorders due to polymerase gamma (POLG1) mutations. Eur J Pediatr 166: 229–234.
Ferrari G, Lamantea E, Donati A, et al (2005) Infantile hepatocerebral syndromes associated with mutations in the mitochondrial DNA polymerase-γa. Brain 128: 723–731.
González-Vioque E, Blázquez A, Fernández-Moreira D, et al (2006) Association of novel POLG mutations and multiple mitochondrial DNA deletions with variable clinical phenotypes in a Spanish population. Arch Neurol 63: 107–111.
Graziewicz MA, Longley MJ, Copeland WC (2006) DNA Polymerase γ in mitochondrial DNA replication and repair. Chem Rev 106: 383–405.
Horvath R, Hudson G, Ferrari G, et al (2006) Phenotypic spectrum associated with mutations of the mitochondrial polymerase γ gene. Brain 129: 1674–1684.
Janssen AJ, Smeitink JA, van den Heuvel LP (2003) Some practical aspects of providing a diagnostic service for respiratory chain defects. Ann Clin Biochem 40: 3–8.
Janssen AJ, Trijbels FJ, Sengers RC, et al (2006) Measurement of the energy-generating capacity of human muscle mitochondria: diagnostic procedure and application to human pathology. Clin Chem 52: 860–871.
Lamantea E, Tiranti V, Bordoni A, et al (2002) Mutations of mitochondrial DNA polymerase gamma A are a frequent cause of autosomal dominant or recessive progressive external ophthalmoplegia. Ann Neurol 52: 211–219.
Longley MJ, Graziewicz MA, Bienstock RJ, Copeland WC (2005) Consequences of mutations in human DNA polymerase γ. Gene 354: 125–131.
Naviaux RK, Nyhan WL, Barshop BA, et al (1999) Mitochondrial DNA polymerase gamma deficiency and mtDNA depletion in a child with Alpers syndrome. Ann Neurol 45: 54–58.
Naviaux RK, Nguyen KV (2004) POLG mutations associated with Alpers syndrome and mitochondrial DNA depletion. Ann Neurol 55: 706–712.
Nguyen KV, Østergaard E, Ravn S, et al (2005) POLG mutations in Alpers syndrome. Neurology 65: 1493–1495.
Sarzi E, Bourdon A, Chrétien D, et al (2007) Mitochondrial DNA depletion is a prevalent cause of multiple respiratory chain deficiency in childhood. J Pediatr 105: 531–534.
Smeitink J, Sengers R, Trijbels F, van den Heuvel L (2001) Human NADH: ubiquinone oxidoreductase. J Bioenerg Biomembr 33: 259–266.
Van Goethem G, Dermaut B, Löfgren A, Martin JJ, Van Broeckhoven C (2001) Mutation of POLG is associated with progressive external ophthalmoplegia characterized by mtDNA deletions. Nat Genet 28: 211–212.
Acknowledgements
Research at the NCMD is funded by the Radboud University Nijmegen Medical Centre, the Prinses Beatrix Fonds, the Dutch Scientific Organization, the Ministry of Economic Affairs and the European Community’s sixth Framework Programme for Research, Priority 1 ‘Life sciences, genomics and biotechnology for health’, contract number LSHM-CT-2004–503116.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicating editor: John Christodoulou
JIMD Short Report #108 (2008) Online
Competing interests: None declared
References to electronic databases: Alpers–Huttenlocher syndrome (OMIM #203700).
Rights and permissions
About this article
Cite this article
de Vries, M.C., Rodenburg, R.J., Morava, E. et al. Normal biochemical analysis of the oxidative phosphorylation (OXPHOS) system in a child with POLG mutations: A cautionary note. J Inherit Metab Dis 31 (Suppl 2), 299–302 (2008). https://doi.org/10.1007/s10545-008-0871-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-008-0871-4