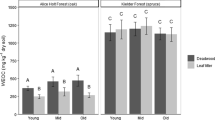
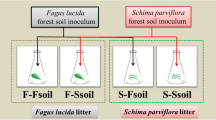
Abstract
In forest ecosystems, deadwood can improve carbon storage, nutrient availability, and water holding capacity in soils. Yet the effect of organic matter from deadwood such as lignin on these soil functions and their regulators are unknown. We hypothesized that carbon storage, exchangeable cations, and pore space increase with the quantity of lignin-derived phenolic acids from beech deadwood. We also hypothesize that the most pronounced differences occur in more advanced decay classes, in the forest floor at sites with moder forest floors, and in the Ah horizon at sites with mull forest floors. Cupric oxide-oxidation products were used to determine lignin concentration, composition, and oxidation from paired reference and test samples next to 42 downed European beech (Fagus sylvatica L.) deadwood logs in ten stands in Southwest Germany. Compared to reference points, the sum of vanillyl, syringyl and cinnamyl lignin-derived phenols increased next to beech deadwood (within 10–20 cm). The composition and oxidation of lignin-derived phenols also changed near beech deadwood: syringyl/vanillyl ratios increased while cinnamyl/vanillyl and aldehyde/acid ratios for vanillyl decreased. Water-extractable organic carbon (OC) and its aromaticity also increased next to beech deadwood as did total OC and particulate OC separated by density fractionation relative to total and mineral-bound OC. These changes occurred namely in the organic horizons of moder forest floors, and in the Ah horizon underneath mull forest floors. These observations indicated that phenols predominantly entered soil in fluxes of fragmented and dissolved organic matter from beech deadwood. Changes to soil nutrient availability and porosity were linked to increasing lignin-derived phenols from beech deadwood especially in nutrient-poor soils and near heavily decayed deadwood. This is evidence that soils close to beech deadwood, a substrate, are spatially limited pedogenic hot-spots that have increased soil carbon, available nutrients, and pore space depending on the forest floor and parent material.



Similar content being viewed by others
Notes
All samples from “mull-calcareous” sites had substantial peaks at 1.77–1.78 ppm \(^\delta \hbox {H}\). Spectra for eight of 14 samples from Kohltal had inverse peaks associated with exceedingly large 1.77–1.78 \(^\delta \hbox {H}\) peaks when acquired with “presaturation” and “PURGE” techniques; repeat measurements exhibited the same inverse peaks. The source of these peaks is unknown, especially as acetone was not used during sample preparation.
Abbreviations
- (Al/Ac)S:
-
Aldehyde/acid ratio for syringyl phenols
- (Al/Ac)V:
-
Aldehyde/acid ratio for vanillyl phenols
- AWC:
-
Available water capacity
- CEC:
-
Cation exchange capacity
- \(k_w\) :
-
Intrinsic air permeability
- LME:
-
Linear mixed effects
- PCA:
-
Principle component analysis
- POC:
-
Particulate organic carbon (filtered \(>\,1.5\,\upmu \hbox {m}\))
- \(\uprho\) :
-
Spearman’s rank correlation coefficient
- SOM:
-
Soil organic matter
- \(\hbox {SUVA}_{{280}}\) :
-
Specific UV absorbance at 280 nm
- VSC:
-
Sum of vanillyl (V), syringyl (S), and cinnamyl (C) phenols
- WEOC:
-
Water-extractable organic carbon (filtered \(<\,0.45\,\upmu \hbox {m}\))
References
Amelung W, Flach KW, Zech W (1999) Lignin in particle-size fractions of native grassland soils as influenced by climate. Soil Sci Soc Am J 63(5):1222–1228. https://doi.org/10.2136/sssaj1999.6351222x
Angst G, Mueller KE, Kögel-Knabner I, Freeman KH, Mueller CW (2017) Aggregation controls the stability of lignin and lipids in clay-sized particulate and mineral associated organic matter. Biogeochemistry 132(3):307–324. https://doi.org/10.1007/s10533-017-0304-2
Ball BC, Harris W, Burford JR (1981) A laboratory method to measure gas diffusion and flow in soil and other porous materials. J Soil Sci 32(3):323–334. https://doi.org/10.1111/j.1365-2389.1981.tb01709.x
Bantle A, Borken W, Ellerbock RH, Schulze ED, Weisser WW, Matzner E (2014) Quantity and quality of dissolved organic carbon released from coarse woody debris of different species in the early phase of decomposition. For Ecol Manag 329:287–294. https://doi.org/10.1016/j.foreco.2014.06.035
Buondonno A, Capra GF, Coppola E, Dazzi C, Grilli E, Odierna P, Rubino M, Vacca S (2014) Aspects of soil phenolic matter (SPM): an expolorative investigation in agricultural, agroforestry and wood ecosystems. Geoderma 231:235–244. https://doi.org/10.1016/j.geoderma.2013.08.026
Cassel DK, Nielsen DR (1986) Field capacity and available water capacity. In: Klute A (ed) Methods of soil analysis: part 1—physical and mineralogical methods, 2nd edn. Soil Science Society of America book series, no. 5. American Society of Agronomy, Inc. & Soil Science Society of America, Inc., Madison, pp 901–926. https://doi.org/10.2136/sssabookser5.1.2ed.c36
Chin YP, Alken G, O’Loughlin E (1994) Molecular weight, polydispersity, and spectroscopic properties of aquatic humic substances. Environ Sci Technol 28(11):1853–1858. https://doi.org/10.1021/es00060a015
Corey AT (1986) Air permeability. In: Klute A (ed) Methods of soil analysis: part 1—physical and mineralogical methods, 2nd edn. Soil Science Society of America book series, no. 5. American Society of Agronomy, Inc. & Soil Science Society of America, Inc., Madison, pp 1121–1136. https://doi.org/10.2136/sssabookser5.1.2ed.c48
Danielson RE, Sutherland PL (1986) Porosity. In: Klute A (ed) Methods of soil analysis: part 1—physical and mineralogical methods, 2nd edn. Soil Science Society of America book series, no. 5. American Society of Agronomy, Inc. & Soil Science Society of America, Inc., Madison, pp 443–461. https://doi.org/10.2136/sssabookser5.1.2ed.c18
Erickson M, Miksche GE, Somfai I (1973) Charakterisierung der Lignine von Angiospermen durch oxydativen Abbau. I. Dikotylen. Holzforschung 27(4):113–117. https://doi.org/10.1515/hfsg.1973.27.4.113
Ertel JR, Hedges JI (1984) The lignin component of humic substances: distribution among soil and sedimentary humic, fulvic, and base-insoluble fractions. Geochim Cosmochim Acta 48(10):2065–2074. https://doi.org/10.1016/0016-7037(84)90387-9
Fravolini G, Tognetti R, Lombardi F, Egli M, Ascher-Jenull J, Arfaioli P, Bardelli T, Cherubini P, Marchetti M (2018) Quantifying decay progression of deadwood in Mediterranean mountain forests. For Ecol Manag 408:228–237. https://doi.org/10.1016/j.foreco.2017.10.031
Gangloff S, Stille P, Schmitt AD, Chabaux F (2016) Factors controlling the chemical composition of colloidal and dissolved fractions in soil solutions and the mobility of trace elements in soils. Geochim Cosmochim Acta 189:37–57. https://doi.org/10.1016/j.gca.2016.06.009
Giannopoulos G, Pulleman MM, Van Groenigen JW (2010) Interactions between residue placement and earthworm ecological strategy affect aggregate turnover and \({\rm N}_2\)O dynamics in agricultural soil. Soil Biol Biochem 42(4):618–625. https://doi.org/10.1016/j.soilbio.2009.12.015
Graf-Rosenfellner M, Cierjacks A, Kleinschmit B, Lang F (2016) Soil formation and its implications for stabilization of soil organic matter in the riparian zone. Catena 139:9–18. https://doi.org/10.1016/j.catena.2015.11.010
Grünewald G, Kaiser K, Jahn R, Guggenberger G (2006) Organic matter stabilization in young calcareous soils as revealed by density fractionation and analysis of lignin-derived constituents. Org Geochem 37(11):1573–1589. https://doi.org/10.1016/j.orggeochem.2006.05.002
Gutachterausschuss Forstliche Analytik (2014) Handbuch Forstliche Analytik: Eine Loseblatt-Sammlung der Analysemethoden im Forstbereich. Bundesministerium für Verbraucherschutz, Ernährung und Landwirtschaft, Bonn, 5th edn. http://www.bmel.de/SharedDocs/Downloads/Landwirtschaft/Wald-Jagd/Bodenzustandserhebung/Handbuch/HandbuchForstanalytikKomplett.html, (In German; last accessed 13 September, 2018)
Hafner SD, Groffman PM, Mitchell MJ (2005) Leaching of dissolved organic carbon, dissolved organic nitrogen, and other solutes from coarse woody debris and litter in a mixed forest in New York State. Biogeochemistry 74(2):257–282. https://doi.org/10.1007/s10533-004-4722-6
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW, Cromack K Jr, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–302. https://doi.org/10.1016/S0065-2504(08)60121-X
Hedges JI, Ertel JR (1982) Characterization of lignin by gas capillary chromatography of cupric oxide oxidation products. Anal Chem 54(2):174–178. https://doi.org/10.1021/ac00239a007
Hedges JI, Mann DC (1979) The characterization of plant tissues by their lignin oxidation products. Geochim Cosmochim Acta 43(11):1803–1807. https://doi.org/10.1016/0016-7037(79)90028-0
Heinze S, Ludwig B, Piepho HP, Mikutta R, Don A, Wordell-Dietrich P, Helfrich M, Hertel D, Leuschner C, Kirfel K, Kandeler E, Preusser S, Guggenberger G, Leinemann T, Marschner B (2018) Factors controlling the variability of organic matter in the top- and subsoil of a sandy dystric cambisol under beech forest. Geoderma 311:37–44. https://doi.org/10.1016/j.geoderma.2017.09.028
Hernes PJ, Robinson AC, Aufdenkampe AK (2007) Fractionation of lignin during leaching and sorption and implications for organic matter “freshness”. Geophys Res Lett 34(17):L17,401. https://doi.org/10.1029/2007GL031017
Hernes PJ, Kaiser K, Dyda RY, Cerli C (2013) Molecular trickery in soil organic matter: hidden lignin. Environ Sci Technol 47(16):9077–9085. https://doi.org/10.1021/es401019n
Herrmann S, Bauhus J (2018) Nutrient retention and release in coarse woody debris of three important central European tree species and the use of NIRS to determine deadwood chemical properties. For Ecosyst 5:22. https://doi.org/10.1186/s40663-018-0140-4
Herrmann S, Kahl T, Bauhus J (2015) Decomposition dynamics of coarse woody debris of three important central European tree species. For Ecosyst 2:27. https://doi.org/10.1186/s40663-015-0052-5
Higuchi T (1997) Biochemistry and molecular biology of wood. Springer, Berlin. https://doi.org/10.1007/978-3-642-60469-0
Kahl T, Mund M, Bauhus J, Schulze ED (2012) Dissolved organic carbon from European beech logs: patterns of input to and retention by surface soil. Ecoscience 19(4):364–373. https://doi.org/10.2980/19-4-3501
Kahl T, Arnstadt T, Baber K, Bässler C, Bauhus J, Borken W, Buscot F, Floren A, Heibl C, Hessenmöller D, Hofrichter M, Hoppe B, Kellner H, Krüger D, Linsemair KE, Matzner E, Otto P, Purahong W, Seilwinder C, Schulze ED, Wende B, Weisser WW, Gossner MM (2017) Wood decay rates of 13 temperate tree species in relation to wood properties, enzyme activities and organismic diversities. For Ecol Manage 391:86–95. https://doi.org/10.1016/j.foreco.2017.02.012
Kappes H, Catalano C, Topp W (2007) Coarse woody debris ameliorates chemical and biotic soil parameters of acidified broad-leaved forests. Appl Soil Ecol 36(2–3):190–198. https://doi.org/10.1016/j.apsoil.2007.02.003
Karroum M, Guillet B, Laggoun-Défarge F, Disnar JR, Lottier N, Villemin G, Toutain F (2005) Morphological evolution of beech litter (Fagus sylvatica L.) and biopolymer transformation (lignin, polysaccharides) in a mull and a moder, under temperate climate (Fougéres, Britany, France). Can J Soil Sci 85(3):405–416. https://doi.org/10.4141/S03-021
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Annu Rev Microbiol 41:465–501. https://doi.org/10.1146/annurev.mi.41.100187.002341
Klotzbücher T, Kaiser K, Guggenberger G, Gatzek C, Kalbitz K (2011) A new conceptual model for the fate of lignin in decomposing plant litter. Ecology 92(5):1052–1062. https://doi.org/10.1890/10-1307.1
Klotzbücher T, Kaiser K, Filley TR, Kalbitz K (2013a) Processes controlling the production of aromatic water-soluble organic matter during litter decomposition. Soil Biol Biochem 67:133–139. https://doi.org/10.1016/j.soilbio.2013.08.003
Klotzbücher T, Strohmeier S, Kaiser K, Bowden RD, Lajtha K, Ohm H, Kalbitz K (2013b) Lignin properties in topsoils of a beech/oak forest after 8 years of manipulated litter fall: relevance of altered input and oxidation of lignin. Plant Soil 367:579–589. https://doi.org/10.1007/s11104-012-1489-8
Klotzbücher T, Kalbitz K, Cerli C, Hernes PJ, Kaiser K (2016) Gone or just out of sight? The apparent disappearance of aromatic litter components in soils. Soil 2(3):325–335. https://doi.org/10.5194/soil-2-325-2016
Kögel I (1986) Estimation and decomposition pattern of the lignin component in forest humus layers. Soil Biol Biochem 18(6):589–594. https://doi.org/10.1016/0038-0717(86)90080-5
Kögel-Knabner I, Rumpel C (2018) Advances in molecular approaches for understanding soil organic matter composition, origin, and turnover: a historical overview. Adv Agron 149:1–48. https://doi.org/10.1016/bs.agron.2018.01.003
Krueger I, Schulz C, Borken W (2017) Stocks and dynamics of soil organic carbon and coarse woody debris in three managed and unmanaged temperate forests. Eur J For Res 136(1):123–137. https://doi.org/10.1007/s10342-016-1013-4
Krzyszowska-Waitkus A, Vance GF, Preston CM (2006) Influence of coarse wood and fine litter on forest organic matter composition. Can J Soil Sci 86(1):35–46. https://doi.org/10.4141/S05-040
Kuehne C, Donath C, Müller-Using SI, Bartsch N (2008) Nutrient fluxes via leaching from coarse woody debris in a Fagus sylvatica forest in the Solling Mountains, Germany. Can J For Res 38(9):2405–2413. https://doi.org/10.1139/X08-088
Lachat T, Brang P, Bolliger M, Bollmann K, Brändli UB, Bütler R, Hermann S, Schneider O, Wermelinger B (2014) Totholz im Wald. Entstehung, Bedeutung und Förderung. Merkbl Prax 52:12 S (in German)
Laiho R, Prescott CE (2004) Decay and nutrient dynamics of coarse woody debris in northern coniferous forests: a synthesis. Can J For Res 34:763–777. https://doi.org/10.1139/X03-241
Lang F, Krüger J, Amelung W, Willbold S, Frossard E, Bünemann EK, Bauhus J, Nitschke R, Kandeler E, Marhan S, Schulz S, Bergkemper F, Schloter M, Luster J, Guggisberg F, Kaiser K, Mikutta R, Guggenberger G, Polle A, Pena R, Prietzel J, Rodionov A, Talkner U, Messenburg H, von Wilpert K, Hölscher A, Dietrich HP, Chmara I (2017) Soil phosphorus supply controls P nutrition strategies of beech forest ecosystems in Central Europe. Biogeochemistry 136(1):5–29. https://doi.org/10.1007/s10533-017-0375-0
Lauber CL, Strickland MS, Bradford MA, Fierer N (2008) The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biol Biochem 40(9):2407–2415. https://doi.org/10.1016/j.soilbio.2008.05.021
Magnússon RI, Tietema A, Cornelissen JHC, Hefting MM, Kalbitz K (2016) Tamm review: sequestration of carbon from coarse woody debris in forest soils. For Ecol Manage 377:1–15. https://doi.org/10.1016/j.foreco.2016.06.033
Martens DA (2002) Relationship between plant phenolic acids released during soil mineralization and aggregate stabilization. Soil Sci Soc Am J 66(6):1857–1867. https://doi.org/10.2136/sssaj2002.1857
Mikutta R, Mikutta C, Kalbitz K, Scheel T, Kaiser K, Jahn R (2007) Biodegradation of forest floor organic matter bound to minerals via different binding mechanisms. Geochim Cosmochim Acta 71(10):2569–2590. https://doi.org/10.1016/j.gca.2007.03.002
Mosier SL, Kane ES, Richter DL, Lilleskov EA, Jurgensen MF, Burton AJ, Resh SC (2017) Interacting effects of climate change and fungal communities on wood-derived carbon in forest soils. Soil Biol Biochem 115:297–309. https://doi.org/10.1016/j.soilbio.2017.08.028
Nelson DW, Sommers LE (1996) Total carbon, organic carbon, and organic matter. In: Sparks DL (ed) Methods of soil analysis: part 3—chemical methods, Soil Science Society of America book series, no. 5. Soil Science Society of America, Inc. & American Society of Agronomy, Inc., Madison, pp 961–1010. https://doi.org/10.2136/sssabookser5.3.c34
Novo-Uzal E, Pomar F, Espiñeira JM (2012) Evolutionary history of lignins. In: Jouanin L, Lapierre C (eds) Lignins: biosynthesis, biodegradation and bioengineering, vol 61. Advances in botanical research. Academic Press, London, pp 311–350. https://doi.org/10.1016/B978-0-12-416023-1.00009-4
Peršoh D, Borken W (2017) Impact of woody debris of different tree species on the microbial activity and community of an underlying organic horizon. Soil Biol Biochem 115:516–525. https://doi.org/10.1016/j.soilbio.2017.09.017
Phillips JD, Marion DA (2004) Pedological memory in forest soil development. For Ecol Manage 188(1–3):363–380. https://doi.org/10.1016/j.foreco.2003.08.007
Santos F, Russell D, Berhe AA (2016) Thermal alteration of water extractable organic matter in climosequence soils from the Sierra Nevada, California. J Geophys Res 121(11):2877–2885. https://doi.org/10.1002/2016JG003597
Sarker TC, Incerti G, Spaccini R, Piccolo A, Mazzoleni S, Bonanomi G (2018) Linking organic matter chemistry with soil aggregate stability: insight from \(^{13}\)C NMR spectroscopy. Soil Biol Biochem 117:175–184. https://doi.org/10.1016/j.soilbio.2017.11.011
Spielvogel S, Prietzel J, Auerswald K, Kögel-Knabner I (2009) Site-specific patterns of soil organic carbon stocks in different landscape units of a high-elevation forest including a site with forest dieback. Geoderma 152(3–4):218–230. https://doi.org/10.1016/j.geoderma.2009.03.009
Spielvogel S, Prietzel J, Kögel-Knabner I (2016) Stand scale variability of topsoil organic matter composition in a high-elevation Norway spruce forest ecosystem. Geoderma 267:112–122. https://doi.org/10.1016/j.geoderma.2015.12.001
Stahr S, Graf-Rosenfellner M, Klysubun W, Mikutta R, Prietzel J, Lang F (2018) Phosphorus speciation and C:N:P stoichiometry of functional organic matter fractions in temperate forest soils. Plant Soil 427(1–2):53–69. https://doi.org/10.1007/s11104-017-3394-7
Strukelji M, Brais S, Quideau SA, Angers VA, Kebli H, Drapeau P, Oh SW (2013) Chemical transformations in downed logs and snags of mixed boreal species during decomposition. Can J For Res 43(9):785–798. https://doi.org/10.1139/cjfr-2013-0086
Stutz KP, Lang F (2017) Potentials and unknowns in managing coarse woody debris for soil functioning. Forests 8(2):37. https://doi.org/10.3390/f8020037
Stutz KP, Dann D, Wambsganss J, Scherer-Lorenzen M, Lang F (2017) Phenolic matter from deadwood can impact forest soil properties. Geoderma 288:204–212. https://doi.org/10.1016/j.geoderma.2016.11.014
Tatti D, Fatton V, Sartori L, Gobat JM, Le Bayon RC (2018) What does ‘lignoform’ really mean? Appl Soil Ecol 123:632–645. https://doi.org/10.1016/j.apsoil.2017.06.037
Thevenot M, Dignac MF, Rumpel C (2010) Fate of lignins in soils: a review. Soil Biol Biochem 42(8):1200–1211. https://doi.org/10.1016/j.soilbio.2010.03.017
Trum F, Titeux H, Ranger J, Delvaux B (2011) Influence of tree species on carbon and nitrogen transformation patterns in forest floor profiles. Ann For Sci 68(4):837–847. https://doi.org/10.1007/s13595-011-0080-4
vanden Enden L, Frey SD, Nadelhoffer KJ, Le Moine JM, Lajtha K, Simpson MJ (2018) Molecular-level changes in soil organic matter composition after 10 years of litter, root and nitrogen manipulation in a temperate forest. Biogeochemistry 141(2):183–197. https://doi.org/10.1007/s10533-018-0512-4
Wambsganss J, Stutz KP, Lang F (2017) European beech deadwood can increase soil organic carbon sequestration in forest topsoils. For Ecol Manage 405:200–209. https://doi.org/10.1016/j.foreco.2017.08.053
Weishaar JL, Aiken GR, Bergamaschi BA, Fram MS, Fujii R, Mopper K (2003) Evaluation of specific ultraviolet absorbance as an indicator of the chemical composition and reactivity of dissolved organic carbon. Environ Sci Technol 37(20):4702–4708. https://doi.org/10.1021/es030360x
WRB IWG (2014) World reference base for soil resources 2014. International soil classification system for naming soils and creating legends for soil maps. No. 106 in World Soil Resources Reports, FAO, Rome
Zalamea M, González G, Lodge DJ (2016) Physical, chemical, and biological properties of soil under decaying wood in a tropical wet forest in Puerto Rico. Forests 7(8):168. https://doi.org/10.3390/f7080168
Zar JH (1999) Biostatistical analysis, 4th edn. Prentice-Hall International Inc, Upper River Saddle
Acknowledgements
We sincerely thank Daniel Dann, Markus Graf-Rosenfellner, Petra Grossmann, Gudrun Nemson-von Koch, Anna Ortmann, Christina Petschke, Camille Puverel, David Rice, Rabea Saad, Helmer Schack-Kirchner, Raphael Schönle, Hannah Simon, Nicole Specht, Jasmin Steininger, Angela Thiemann, Petra Wiedemer, and Niklas Wisskirchen for their assistance. Two anonymous reviewers additionally provided constructive and beneficial comments. We also thank the Forstliche Versuchs- und Forschungsanstalt Baden-Württemberg, the Forschungsanstalt für Waldökologie und Forstwirtschaft, ForstBW, and Landesforsten RLP for providing access to the study sites. This work was supported by a Grant from the Ministry of Science, Research and the Arts of Baden-Württemberg (Az: 33-7533-10-5/81) to Kenton Stutz and Friederike Lang.
Author information
Authors and Affiliations
Contributions
KPS and FL conceived and designed the study; KPS and JW collected field samples and, along with FS, performed the laboratory trials; KPS, KK, and FS analyzed the data; KPS wrote the paper; and all authors developed and revised the paper.
Corresponding author
Additional information
Responsible Editor: Jan Mulder.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Stutz, K.P., Kaiser, K., Wambsganss, J. et al. Lignin from white-rotted European beech deadwood and soil functions. Biogeochemistry 145, 81–105 (2019). https://doi.org/10.1007/s10533-019-00593-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-019-00593-2