Abstract
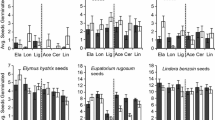
Allelopathy, the release of chemicals by plants that inhibit the germination and growth of competing species, can be an important trait for invasive success. However, little is known about potential biogeographical differences in allelopathy due to divergent regional eco-evolutionary histories. To test this, we examined the allelochemical potential of the highly invasive species Centaurea solstitialis from six world regions including native (Spain, Turkey) and non-native ranges (Argentina, Chile, California and Australia). Seeds from several populations in each region were collected and grown under common garden conditions. Allelopathic potential and chemical composition of three leaf extract concentrations of C. solstitialis from each region: 0.25%, 0.5% 0.75% (w/v−1) were assessed on the phytometer Lactuca sativa. The main allelochemicals present in the leaf-surface extract were sesquiterpene lactones that varied in major constitutive compounds across regions. These leaf extracts had strong inhibitory effects on L. sativa seed germination and net growth. Summed across regions, the 0.25% concentration suppressed germination by 72% and radicle elongation by 66%, relative to the controls. At the 0.5% concentration, no seeds germinated when exposed to extracts from the non-native ranges of Argentina and Chile, whereas germination and radicle growth were reduced by 98% and 89%, respectively, in the remaining regions, relative to controls. Germination and seedling growth were completely inhibited at the 0.75% concentration extract for all regions. Some non-native regions were characterized by relatively lower concentrations of allelochemicals, suggesting that there is biogeographical variation in allelopathic expression. These findings imply that rapid selection on the biochemical signatures of an exotic invasive plant species can be highly region-specific across the world.





Similar content being viewed by others
References
Almeida ABA, Miotto AM, Nunes DS, Spadari Bratifisch RC, Brito ARM (2002) Mechanism of antiulcerogenic activity of semi-synthetic crotonin obtained from Croton cajucara Benth. Rev Bras Farmacogn 12:105–110. https://doi.org/10.1590/S0102-695X2002000300050
Alvarez P (2008) Sheep grazing to control yellow starthistle: role of sesquiterpene lactones. PhD thesis, University of California
Aschehoug ET, Callaway RM, Newcombe G, Tharayil N, Chen S (2014) Fungal endophyte increases the allelopathic effects of an invasive forb. Oecologia 175:285–291. https://doi.org/10.1007/s00442-014-2891-0
Aschehoug ET, Brooker R, Atwater DZ, Maron JL, Callaway RM (2016) The mechanisms and consequences of interspecific competition among plants. Annu Rev Ecol Evol Syst 47:263–281. https://doi.org/10.1146/annurev-ecolsys-121415-032123
Bachelier A, Mayer R, Klein CD (2006) Sesquiterpene lactones are potent and irreversible inhibitors of the antibacterial target enzyme MurA. Bioorg Med Chem Lett 16:5605–5609. https://doi.org/10.1016/j.bmcl.2006.08.021
Barker BS, Andonian K, SwopE SM, Luster DG, Dlugosch KM (2017) Population genomic analysis reveal a history of range expansion and trait evolution across the native and invaded range of yellow starthistle (Centaurea solstitialis). Mol Ecol 26:1131–1147. https://doi.org/10.1111/mec.13998
Barto EK, Powell JR, Cipollini D (2010) How novel are the chemical weapons of garlic mustard in North American forest understories? Biol Invasions 12(10):3456–3471. https://doi.org/10.1007/s10530-010-9744-5
Bartoli A, Galati BG, Tortosa RD (2011) Anatomical studies of the secretory structures: glandular trichomes and ducts, in Grindelia pulchella Dunal (Astereae, Asteraceae). Flora 206:1063–1068. https://doi.org/10.1016/j.flora.2011.07.012
Beck JJ, Smith L, Merrill GB (2008) In situ volatile collection, analysis and comparison of three Centaurea species and their relationship to biocontrol with herbivorous insects. J Agric Food Chem 56:2759–2764. https://doi.org/10.1021/jf073383u
Belz RC, van der Laan M, Reinhardt CF, Hurle K (2009) Soil degradation of parthenin—does it contradict the role of allelopathy in the invasive weed Parthenium hysterophorus L.? J Chem Ecol 35:1137–1150. https://doi.org/10.1007/s10886-009-9698-1
Bhattacharyya PR, Barua NC, Ghosh AC (1996) Cynaropicrin from Trichlorepis glaberrima: a potential insect feeding deterrent compound. Ind Crop Prod 4:291–294. https://doi.org/10.1016/0926-6690(95)00044-5
Bruno M, Bancheva S, Rosselli S, Maggio A (2013) Sesquiterpenoids in subtribe Centaureinae (Cass.) Dumort (tribe Cardueae, Asteraceae): distribution, 13C NMR spectral data and biological properties. Phytochemistry 95:19–93. https://doi.org/10.1016/j.phytochem.2013.07.002
Burrows GE, Tyrl RJ (2013) Toxic plants of North America, Asteraceae Martinov, 2nd edn. Wiley-Blackwell, Iowa, pp 150–256
Callaway RM, Aschehoug ET (2000) Invasive plants versus their new old neighbors: a mechanism for exotic invasion. Science 290:521–523. https://doi.org/10.1126/science.290.5491.521
Callaway RM, Ridenour WM (2004) Novel weapons: invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436–443. https://doi.org/10.1890/1540-9295(2004)002[0436:NWISAT]2.0.CO;2
Carpenter DJ (2007) The role of the novel weapons and enemy release hypothesis on the invasiveness of knapweeds, Centaurea spp., in North America. Dissertation. Carleton University
Centaurea solstitialis L. in GBIF Secretariat (2017) GBIF Backbone Taxonomy. Checklist dataset. https://doi.org/10.15468/39omei. Accessed 20 July 2018
Chen S, Xiao S, Callaway RM (2013) Light intensity alters the allelopathic effects of an exotic invader. Plant Ecol Divers 5:521–526. https://doi.org/10.1080/17550874.2012.736548
Cheng CHK, Costal B, Hamburger M, Hostettmann K, Naylor RJ, Wang Y, Jenner P (1992) Toxic effects of solstitialin A 13 acetate and cynaropicrin from Centaurea solstitialis L (Asteraceae) in cell cultures of fetal rat brain. Neuropharmacology 31:271–277. https://doi.org/10.1016/0028-3908(92)90177-Q
Chon SU, Nelson CJ (2010) Allelopathy in Compositae plants. A review. Agron Sustain Dev 30:349–358. https://doi.org/10.1051/agro/2009027
Cis J, Nowak G, Kisiel W (2006) Antifeedant properties and chemotaxonomic implications of sesquiterpene lactones and syringing from Rhaponticum pulchrum. Biochem Syst Ecol 34:862–867. https://doi.org/10.1016/j.bse.2006.05.019
Dayan FE, Howell JL, Weidenhamer JD (2010) Dynamic root exudation of sorgoleone and its in planta mechanism of action. J Exp Bot 60:2107–2117. https://doi.org/10.1093/jxb/erp082
El Ayeb-Zakhama A, Sakka-Rouis L, Flamini G, Jannet HB, Harzallah-Skhiri F (2017) Chemical composition and allelopathic potential of essential oils from Citharexylum spinosum L. grown in Tunisia. Chem Biodivers 14(4):e1600225. https://doi.org/10.1002/cbdv.201600225
Eriksen RL, Hierro JL, Eren Ö, Andonian K, Török K, Becerra PI, Montesinos D, Khetsuriani L, Diaconu A, Kesseli R (2014) Dispersal pathways and genetic differentiation among worldwide populations of the invasive weed Centaurea solstitialis L. (Asteraceae). PLoS ONE 12:e114786. https://doi.org/10.1371/journal.pone.0114786
Filipe JC, Jorge A, Özkan E, Sotes G, Hierro J, Montesinos D (2016) Invasive and non-invasive congeneric Centaurea (Asteraceae) show contrasting patterns of herbivory by snails. Plant Ecol Evol 149:228–232. https://doi.org/10.5091/plecevo.2016.1231
Geppert B, Drozdz B, Kielczewski M, Holub M (1983) Sesquiterpene lactones. XXIII. Isolation of sesquiterpene lactones from Centaurea L. species. Acta Soc Bot Pol 52:23–34. https://doi.org/10.5586/asbp.1983.003
Gómez-González S, Cavieres LA, Torres Morales P, Torres-Díaz C (2009) Competitive effects of the alien invasive Centaurea solstitialis L. on two Chilean Baccharis species at different life-cycle stages. Gayana Bot 66:71–83. https://doi.org/10.4067/S0717-66432009000100007
Göpfert JC, Heil N, Conrad J, Spring O (2005) Cytological development and sesquiterpene lactone secretion in capitate glandular trichomes of sunflower. Plant Biol 7:148–155. https://doi.org/10.1055/s-2005-837575
Graebner RC, Callaway RM, Montesinos D (2012) Invasive species grows faster, competes better and shows greater evolution toward increased seed size and growth than exotic non-invasive congeners. Plant Ecol 213:545–553. https://doi.org/10.1007/s11258-012-0020-x
Gruntman M, Zieger S, Tielbörger K (2015) Invasive success and the evolution of enhanced weaponry. Oikos 125:59–65. https://doi.org/10.1111/oik.02109
Gruntman M, Segev U, Glauser G, Tielbörger K (2017) Evolution of plant defences along an invasion chronosequence: defence is lost due to enemy release—but not forever. J Ecol 105(1):255–264. https://doi.org/10.1111/1365-2745.12660
Hasan HAH (1998) Studies on toxigenic fungi in roasted food stuff (salted seed) and halotolerant activity of emodin-producing Aspergillus wentii. Folia Microbiol 43:383–391. https://doi.org/10.1007/BF02818578
Heywood VH, Harborne JB, Turner BL (1977) The biology and chemistry of the compositae. Academic Press Inc., New York
Hierro JL, Eren Ö, Villarreal D, Chiuffo MC (2013) Non-native conditions favor non-native populations of invasive plant: demographic consequences of seed size variation? Oikos. https://doi.org/10.1111/j.1600-0706.2012.00022.x
Inderjit, Dakshini KMM (1995) On laboratory bioassays in allelopathy. Bot Rev 61:28–44. https://doi.org/10.1007/bf02897150
Inoue N, Nishimura H, Li HH, Mizutani J (1992) Allelochemicals from Polygonum sachalinese Fr. Schm. (Polygonaceae). J Chem Ecol 18:1833–1840. https://doi.org/10.1007/BF02751107
ISO (1995) Soil quality-determination of the effects of pollutants on soil flora. Part 2: effects of chemicals on the emergence of higher plants. International Organization for Standardization, Genève
Izhaki I (2002) Emodin—a secondary metabolite with multiple ecological functions in higher plants. New Phytol 155:205–217. https://doi.org/10.1046/j.1469-8137.2002.00459.x
Jalaei Z, Fattahi M, Aramideh S (2015) Allelopathic and insecticidal activities of essential oil of Dracocephalum kotschyi Boiss. From Iran: a new chemotype with highest limonene-10-al and limonene. Ind Crops Prod 73:109–117. https://doi.org/10.1016/j.indcrop.2015.04.020
Kokalis-Burelle N, Rodríguez-Kábana R (2006) Allelochemicals as biopesticides for management of plant parasitic nematodes. In: Inderjit S, Mukerji KG (eds) Allelochemicals: biological control of plant pathogens and diseases. Springer, Dordrecht, pp 15–29
Lankau RA (2012) Coevolution between invasive and native plant driven by chemical competition and soil biota. PNAS 109:11240–11245. https://doi.org/10.1073/pnas.1201343109
Lankau RA, Nuzzo V, Spyreas G, Davis AS (2009) Evolutionary limits ameliorate the negative impact of an invasive plant. PNAS 106:15362–15367. https://doi.org/10.1073/pnas.0905446106
Levene H (1960) Robust testes for equality of variances. In: Olkin I (ed) Contributions to probability and statistics. Stanford University Press, Palo Alto, pp 278–292
Locken LJ (1985) Cnicin concentrations in spotted knapweed (Centaurea maculosa Lam.) and associated soils. Dissertation, University of Montana
Locken LJ, Kelsey RG (1987) Cnicin concentrations in Centaurea maculosa, spotted knapweed. Biochem Syst Ecol 15:313–320. https://doi.org/10.1016/0305-1978(87)90005-6
Lu-Irving P, Harenčár JG, Sounart H, Welles SR, Swope SM, Baltrus DA, Dlugosch KM (2019) Native and invading yellow starthistle (Centaurea solstitialis) microbiomes differ in composition and diversity of bacteria. mSphere 4:e00088-19. https://doi.org/10.1128/mSphere.00088-19
Maron JL, Montserrat V, Bommarco R, Elmendorf S, Beardsley P (2004) Rapid evolution of an invasive plant. Ecol Monogr 74(2):261–280. https://doi.org/10.1890/03-4027
Meepagala KM, Osbrink W, Sturtz G, Lax A (2006) Plant derived natural products exhibiting activity against Formosan subterranean termites (Coptotermes formosanus). Pest Manag Sci 62:565–570. https://doi.org/10.1002/ps.1214
Montesinos D, Callaway RM (2017) Inter-regional hybrids of native and invasive Centaurea solstitialis display intermediate competitive ability. Ecography 40:801–802. https://doi.org/10.1111/ecog.02653
Muir A, Majak W (1983) Allelopathic potential of diffuse knapweed (Centaurea diffusa) extracts. Can J Plant Sci 63:989–996. https://doi.org/10.4141/cjps83-124
Müller-Schärer H, Schaffner U, Steinger T (2004) Evolution in invasive plants: implications for biological control. Trends Ecol Evol 19:417–422. https://doi.org/10.1016/j.tree.2004.05.010
Ni G, Schaffner U, Peng S, Callaway RM (2010) Acroptilon repens, an Asian invader, has stronger competitive effects on species from America than species from its native range. Biol Invasions 12:3653–3663. https://doi.org/10.1007/s10530-010-9759-y
OECD (2003) Organisation for economic cooperation and development: OECD guideline 208. Terrestrial plant test. Seedling emergence and seedling growth test. OECD, Paris
Oksanen J, Blanchet GF, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Szoecs E, Wagner H (2018) vegan: community ecology package. R package version 2.5-4. https://CRAN.R-project.org/package=vegan. Accessed 14 July 2018
Oster M, Beck JJ, Furrow RE, Yeung K, Field CB (2015) In-field yellow star thistle (Centaurea solstitialis) volatile composition under elevated temperature and CO2 and implications for future control. Chemoecology 25:313–323. https://doi.org/10.1007/s00049-015-0200-y
Özçelik B, Gürbüz I, Karaoğlu T, Yeşilada E (2009) Antiviral and antimicrobial activities of three sesquiterpene lactones from Centaurea solstitialis L. ssp solstitialis. Microbiol Res 164:545–552. https://doi.org/10.1016/j.micres.2007.05.006
Picman AK (1987) Persistence and disappearance of the sesquiterpene lactone, isoalantolactone, in soils. Biochem Syst Ecol 15:361–363. https://doi.org/10.1016/0305-1978(87)90012-3
Qin B, Lau JA, Kopshever J, Callaway RM, McGray H, Perry LG, Weir TL, Paschke MW, Hierro JL, Vivanco JM, Strauss S (2007) No evidence for root-mediated allelopathy in Centaurea solstitialis, a species in a commonly allelopathic genus. Biol Invasions 9:897–907. https://doi.org/10.1007/s10530-007-9089-x
Quintana N, Weir TL, Du J, Broeckling CD, Rieder JP, Stermitz FR, Paschke MW, Vivanco JM (2008) Phytotoxic polyacetylenes from roots of Russian knapweed (Acroptilon repens (L.) DC.). Phytochemistry 69:2572–2578. https://doi.org/10.1016/j.phytochem.2008.07.015
Quintana N, El Kassis EG, Stermitz FR, Vivanco JM (2009) Phytotoxic compounds from roots of Centaurea diffusa Lam. Plant Signal Behav 4:9–14. https://doi.org/10.4161/psb.4.1.7487
Ranal MA, Garcia de Santana D, Resende Ferreira W, Mendes-Rodrigues C (2009) Calculating germination measurements and organizing spreadsheets. Rev Bras Bot 32:849–855. https://doi.org/10.1590/S0100-84042009000400022
Revelle W (2018). psych: procedures for personality and psychological research. Northwestern University, Evanston. R package version 1.8
Ridenour WM, Callaway RM (2001) The relative importance of allelopathy in interference: the effects of an invasive weed on a native bunchgrass. Oecologia 126:444–450. https://doi.org/10.1007/s004420000533
Riopelle RJ, Stevens KL (1993) In vitro neurotoxicity bioassay: neurotoxicity of sesquiterpene lactones. In: Colegate SM, Molyneux RJ (eds) Bioactive natural products detection, isolation and structural determination. CRC Press, Florida, pp 457–463
Roche BF Jr, Roche CT, Chapman RC (1994) Impacts of grassland habitat on yellow starthistle (Centaurea solstitialis L.) invasion. Northwest Sci 68:86–96
Rosche C, Hensen I, Lachmuth S (2018) Local pre-adaptation to disturbance and inbreeding–environment interactions affect colonisation abilities of diploid and tetraploid Centaurea stoebe. Plant Biol 20:75–84. https://doi.org/10.1111/plb.12628
Rosinski G, Bloszyk E, Harmatha J, Knapik A (1988) Changes produced by sesquiterpenes lactones in some physiological processes in Tenebrio molitor L. In: Sehnal F, Zabza A, Denlinger DL (eds) Endocrinological frontiers in physiological insect ecology. Wroclaw Technical University Press, Poland, pp 91–94
Sampaio BL, Edrada-Ebel RA, Batista Da Costa F (2016) Effect of the environment on the secondary metabolic profile of Tithonia diversifolia: a model for environmental metabolomics of plants. Sci Rep 6:29265. https://doi.org/10.1038/srep29265
Schemske DW (2010) Adaptation and the origin of species. Am Nat 176:S4–S25. https://doi.org/10.1086/657060
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591–611. https://doi.org/10.2307/2333709
Sheley RL, Larson LL, Jacobs JS (1999) Yellow starthistle. In: Sheley RL, Petroff JK (eds) Biology and management of noxious rangeland weeds. Oregon State University Press, Oregon, pp 408–416
Smith L, Beck JJ (2013) Effect of mechanical damage on emission of volatile organic compounds from plant leaves and implications for evaluation of host plant specificity of prospective biological control agents of weeds. Biocontrol Sci Technol 23:880–907. https://doi.org/10.1080/09583157.2013.807908
Sotes GJ, Cavieres LA, Montesinos D, Pereira Coutinho AX, Pelaez WJ, Lopes SM, Pinho e Melo TMMVD (2015) Inter-regional variation on leaf surface defenses in native and non-native Centaurea solstitialis plants. Biochem Syst Ecol 62:208–218. https://doi.org/10.1016/j.bse.2015.09.003
Stevens KL, Merrill GB (1985) Sesquiterpene lactones and allelochemicals from Centaurea species. In: Thompson AC (ed) ACS symposium series, the chemistry of allelopathy. American Chemical Society, Washington DC, pp 83–98
Stevens KL, Riopelle RJ, Wong RY (1990) Repin, a sesquiterpene lactone from Acroptilon repens possessing exceptional biological activity. J Nat Prod 53:218–221. https://doi.org/10.1021/np50067a038
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 18 June 2018
Tharayil N, Bhowmik P, Alpert P, Walker E, Amarasiriwardena D, Xing B (2009) Dual purpose secondary compounds: phytotoxin of Centaurea diffusa also facilitates nutrient uptake. New Phytol 181:424–434. https://doi.org/10.1111/j.1469-8137.2008.02647.x
Thelen GC, Vivanco JM, Newingham B, Good W, Bais HP, Landres P, Caesar A, Callaway RM (2005) Insect herbivory stimulates allelopathic exudation by an invasive plant and the suppression of natives. Ecol Lett 8:209–217. https://doi.org/10.1111/j.1461-0248.2004.00713.x
US EPA (1996) Ecological effects test guidelines (OPPTS 850.4200): seed germination/root elongation toxicity test. US Government Printing Office, Washington DC, pp 1–6
Uygur S, Smith L, Uygur FN, Cristofaro M, Balciunas J (2004) Population densitites of yellow starthistle (Centaurea solstitialis) in Turkey. Weed Sci 52:746–753
Vrchotová N, Ŝerá B, Krejčová J (2011) Allelopathic activity of extracts from Impatiens species. Plant Soil Environ 57:57–60. https://doi.org/10.17221/156/2010-PSE
Vu VQ (2011) ggbiplot: a ggplot2 based biplot. R package version 0.55. http://github.com/vqv/ggbiplot. Accessed 10 Mar 2019
Weir TL, Park SW, Vivanco JM (2004) Biochemical and physiological mechanisms mediated by allelochemicals. Curr Opin Plant Biol 7:472–479. https://doi.org/10.1016/j.pbi.2004.05.007
Yuan Y, Wang B, Zhang S, Tang J, Tu C, Hu S, Yong JWH, Chen X (2012) Enhanced allelopathy and competitive ability of invasive plant Solidago canadensis in its introduced range. J Plant Ecol 6(3):253–263. https://doi.org/10.1093/jpe/rts033
Zamora DL (1984) The allelopathic potential of yellow starthistle (Centaurea solstitialis L.). Dissertation, University of Idaho
Zhang H, Mallik A, Zeng RS (2013) Control of Panama disease of banana by rotating and intercropping with Chinese chive (Allium tuberosum Rottller): role of plant volatiles. J Chem Ecol 39:243–252. https://doi.org/10.1007/s10886-013-0243-x
Acknowledgements
Thanks to Soraia Branco, João Filipe and Andreia Jorge for help with the lab work. REI and DM thank the Portuguese Fundação para a Ciência e a Tecnologia (FCT) and the co-funding by the Fundo Europeu de Desenvolvimento Regional (FEDER), within the PT2020 and COMPETE 2020 program, for scholarships and grants: UID/BIA/04004/2013; IF-00066-2013; PTDC/BIA-PLA/0763/2014; SFRH/BD/110987/2015. SL and TPM thank Coimbra Chemistry Centre (CQC), supported by FCT through project POCI-01-0145-FEDER-007630, co-funded by COMPETE2020-UE. SL also acknowledge FCT for the post-doctoral research grant SFRH/BPD/84413/2012. RMC acknowledges support from the National Science Foundation EPSCoR Cooperative Agreement OIA-1757351. CJL is supported by a grant from the Natural Sciences and Engineering Research Council of Canada (NSERC) to examine plant dynamics in California, USA. LAC acknowledge funding from Fondo Nacional de Desarrollo Científico y Tecnológico (FONDECYT-1171005).
Author information
Authors and Affiliations
Contributions
REI and DM designed the study, SMML and TMVDPM performed the chemical analysis, GS, LAC, ÖE, CJL, KF, JLH, collected plant seeds, REI, SMML, CR, RMC and DM analyzed the data. REI and DM lead the writing of the manuscript. All authors contributed to earlier drafts of this manuscript and gave final approval for publication.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10530_2019_2038_MOESM1_ESM.docx
Additional information on the statistical tests conducted in this study are included in the Supporting Information for this manuscript. Data on the collinearity test is included in Appendix S1, Tukey post hoc tests may be found in Appendix S2 and the germination indices are included in the Appendix S3. (DOCX 6,113 kb)
Rights and permissions
About this article
Cite this article
Irimia, R.E., Lopes, S.M.M., Sotes, G. et al. Biogeographic differences in the allelopathy of leaf surface extracts of an invasive weed. Biol Invasions 21, 3151–3168 (2019). https://doi.org/10.1007/s10530-019-02038-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-019-02038-1