Abstract
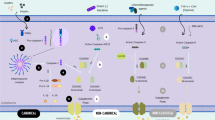
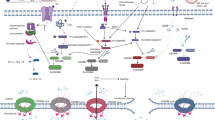
Pyroptosis is a gasdermin-mediated programmed cell death (PCD) pathway. It differs from apoptosis because of the secretion of inflammatory molecules. Pyroptosis is closely associated with various malignant tumors. Recent studies have demonstrated that pyroptosis can either inhibit or promote the development of malignant tumors, depending on the cell type (immune or cancer cells) and duration and severity of the process. This review summarizes the molecular mechanisms of pyroptosis, its relationship with malignancies, and focuses on current pyroptosis inducers and their significance in cancer treatment. The molecules involved in the pyroptosis signaling pathway could serve as therapeutic targets for the development of novel drugs for cancer therapy. In addition, we analyzed the potential of combining pyroptosis with conventional anticancer techniques as a promising strategy for cancer treatment.





Similar content being viewed by others
Data Availability
Not applicable.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 Countries[J]. CA Cancer J Clin 71(3):209–249
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation[J]. Cell 144(5):646–674
Philip M, Schietinger A (2022) CD8(+) T cell differentiation and dysfunction in cancer[J]. Nat Rev Immunol 22(4):209–223
Warburg O (1956) On the origin of cancer cells[J]. Science 123(3191):309–314
Koren E, Fuchs Y (2021) Modes of regulated cell death in Cancer[J]. Cancer Discov 11(2):245–265
Yu P, Zhang X, Liu N et al (2021) Pyroptosis: mechanisms and diseases[J]. Signal Transduct Target Ther 6(1):128
Zhang Y, Chen X, Gueydan C et al (2018) Plasma membrane changes during programmed cell deaths[J]. Cell Res 28(1):9–21
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics[J]. Br J Cancer 26(4):239–257
Medina CB, Mehrotra P, Arandjelovic S et al (2020) Metabolites released from apoptotic cells act as tissue messengers[J]. Nature 580(7801):130–135
Shi J, Gao W, Shao F, Pyroptosis (2017) Gasdermin-mediated programmed necrotic cell Death[J]. Trends Biochem Sci 42(4):245–254
Karmakar M, Minns M, Greenberg EN et al (2020) N-GSDMD trafficking to neutrophil organelles facilitates IL-1β release independently of plasma membrane pores and pyroptosis[J]. Nat Commun 11(1):2212
Kijima M, Mizuta R (2019) Histone H1 quantity determines the efficiencies of apoptotic DNA fragmentation and chromatin condensation[J]. Biomed Res 40(1):51–56
Zychlinsky A, Prevost MC, Sansonetti PJ (1992) Shigella flexneri induces apoptosis in infected macrophages[J]. Nature 358(6382):167–169
Hou J, Hsu JM, Hung MC (2021) Molecular mechanisms and functions of pyroptosis in inflammation and antitumor immunity[J]. Mol Cell 81(22):4579–4590
Chen S, Mei S, Luo Y et al (2018) Gasdermin Family: a promising therapeutic target for Stroke[J]. Transl Stroke Res 9(6):555–563
Broz P, Pelegrín P, Shao F (2020) The gasdermins, a protein family executing cell death and inflammation[J]. Nat Rev Immunol 20(3):143–157
Julien O, Wells JA (2017) Caspases and their substrates[J]. Cell Death Differ 24(8):1380–1389
Liu Z, Wang C, Yang J et al (2019) Crystal structures of the full-length murine and human gasdermin D reveal mechanisms of autoinhibition, lipid binding, and Oligomerization[J]. Immunity 51(1):43–49e4
Rogers C, Fernandes-Alnemri T, Mayes L et al (2017) Cleavage of DFNA5 by caspase-3 during apoptosis mediates progression to secondary necrotic/pyroptotic cell death[J]. Nat Commun 8:14128
Sharma M, De Alba E, Structure (2021) Activation and regulation of NLRP3 and AIM2 Inflammasomes[J]. Int J Mol Sci, 22(2)
Lamkanfi M, Dixit VM (2014) Mechanisms and functions of inflammasomes[J]. Cell 157(5):1013–1022
Broz P, Dixit VM (2016) Inflammasomes: mechanism of assembly, regulation and signalling[J]. Nat Rev Immunol 16(7):407–420
Rathinam VA, Jiang Z, Waggoner SN et al (2010) The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses[J]. Nat Immunol 11(5):395–402
Heilig R, Broz P (2018) Function and mechanism of the pyrin inflammasome[J]. Eur J Immunol 48(2):230–238
Aachoui Y, Sagulenko V, Miao EA et al (2013) Inflammasome-mediated pyroptotic and apoptotic cell death, and defense against infection[J]. Curr Opin Microbiol 16(3):319–326
Shi J, Zhao Y, Wang K et al (2015) Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death[J]. Nature 526(7575):660–665
Wang K, Sun Q, Zhong X et al (2020) Structural mechanism for GSDMD Targeting by Autoprocessed caspases in Pyroptosis[J]. Cell 180(5):941–955e20
Thi HTH, Hong S (2017) Inflammasome as a therapeutic target for Cancer Prevention and Treatment[J]. J Cancer Prev 22(2):62–73
Li D, Wu M (2021) Pattern recognition receptors in health and diseases[J]. Signal Transduct Target Ther 6(1):291
Zindel J, Kubes P (2020) DAMPs, PAMPs, and LAMPs in immunity and sterile Inflammation[J]. Annu Rev Pathol 15:493–518
Gong T, Liu L, Jiang W et al (2020) DAMP-sensing receptors in sterile inflammation and inflammatory diseases[J]. Nat Rev Immunol 20(2):95–112
Takeda K, Akira S (2015) Toll-like receptors[J]. Curr Protoc Immunol, 109: 14.12.1-14.12.10
Liston A, Masters SL (2017) Homeostasis-altering molecular processes as mechanisms of inflammasome activation[J]. Nat Rev Immunol 17(3):208–214
Liu X, Zhang Z, Ruan J et al (2016) Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores[J]. Nature 535(7610):153–158
He WT, Wan H, Hu L et al (2015) Gasdermin D is an executor of pyroptosis and required for interleukin-1β secretion[J]. Cell Res 25(12):1285–1298
Kayagaki N, Wong MT, Stowe IB et al (2013) Noncanonical inflammasome activation by intracellular LPS Independent of TLR4[J]. Science 341(6151):1246–1249
Kayagaki N, Stowe IB, Lee BL et al (2015) Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling[J]. Nature 526(7575):666–671
Kayagaki N, Warming S, Lamkanfi M et al (2011) Non-canonical inflammasome activation targets caspase-11[J]. Nature 479(7371):117–121
Yang D, He Y, Muñoz-Planillo R et al (2015) Caspase-11 Requires the Pannexin-1 Channel and the Purinergic P2X7 Pore to Mediate Pyroptosis and Endotoxic Shock[J]. Immunity 43(5):923–932
Muendlein HI, Jetton D, Connolly WM, Eidell KP, Magri Z, Smirnova I, Poltorak A (2020) cFLIPL protects macrophages from LPS-induced pyroptosis via inhibition of complex II formation. Science 367:1379–1384
Sarhan J, Liu BC, Muendlein HI et al (2018) Caspase-8 induces cleavage of gasdermin D to elicit pyroptosis during Yersinia infection[J]. Proc Natl Acad Sci U S A 115(46):E10888–e10897
Kambara H, Liu F, Zhang X et al (2018) Gasdermin D exerts anti-inflammatory effects by promoting Neutrophil Death[J]. Cell Rep 22(11):2924–2936
De Vasconcelos NM, Van Opdenbosch N, Van Gorp H et al (2019) Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture[J]. Cell Death Differ 26(1):146–161
Humphries F, Shmuel-Galia L, Ketelut-Carneiro N et al (2020) Succination inactivates gasdermin D and blocks pyroptosis[J]. Science 369(6511):1633–1637
Pickering RJ, Bryant CE (2020) Preventing pores and inflammation[J]. Science 369(6511):1564–1565
Rühl S, Shkarina K, Demarco B et al (2018) ESCRT-dependent membrane repair negatively regulates pyroptosis downstream of GSDMD activation[J]. Science 362(6417):956–960
Wang D, Zheng J, Hu Q et al (2020) Magnesium protects against sepsis by blocking gasdermin D N-terminal-induced pyroptosis[J]. Cell Death Differ 27(2):466–481
Yu P, Chen X, Peng C (2023) A new perspective in pyroptosis: lifting the veil on GSDMA activation[J]. Front Med,
Deng W, Bai Y, Deng F et al (2022) Streptococcal pyrogenic exotoxin B cleaves GSDMA and triggers pyroptosis[J]. Nature 602(7897):496–502
Larock DL, Johnson AF, Wilde S et al (2022) Group A Streptococcus induces GSDMA-dependent pyroptosis in keratinocytes[J]. Nature 605(7910):527–531
Wang Y, Gao W, Shi X et al (2017) Chemotherapy Drugs induce pyroptosis through caspase-3 cleavage of a gasdermin[J]. Nature 547(7661):99–103
Zheng Z, Bian Y, Zhang Y et al (2020) Metformin activates AMPK/SIRT1/NF-κB pathway and induces mitochondrial dysfunction to drive caspase3/GSDME-mediated cancer cell pyroptosis[J]. Cell Cycle 19(10):1089–1104
Tan G, Huang C, Chen J et al (2020) HMGB1 released from GSDME-mediated pyroptotic epithelial cells participates in the tumorigenesis of colitis-associated Colorectal cancer through the ERK1/2 pathway[J]. J Hematol Oncol 13(1):149
Li Y, Wang W, Li A et al (2021) Dihydroartemisinin induces pyroptosis by promoting the AIM2/caspase-3/DFNA5 axis in Breast cancer cells[J]. Chem Biol Interact 340:109434
Ding Q, Zhang W, Cheng C et al (2020) Dioscin inhibits the growth of human osteosarcoma by inducing G2/M-phase arrest, apoptosis, and GSDME-dependent cell death in vitro and in vivo[J]. J Cell Physiol 235(3):2911–2924
Cai J, Yi M, Tan Y et al (2021) Natural product triptolide induces GSDME-mediated pyroptosis in Head and Neck cancer through suppressing mitochondrial hexokinase-ΙΙ[J]. J Exp Clin Cancer Res 40(1):190
Zheng X, Zhong T, Ma Y et al (2020) Bnip3 mediates doxorubicin-induced cardiomyocyte pyroptosis via caspase-3/GSDME[J]. Life Sci 242:117186
Jiang M, Qi L, Li L et al (2020) The caspase-3/GSDME signal pathway as a switch between apoptosis and pyroptosis in cancer[J]. Cell Death Discov 6:112
Wang Y, Yin B, Li D et al (2018) GSDME mediates caspase-3-dependent pyroptosis in gastric cancer[J]. Biochem Biophys Res Commun 495(1):1418–1425
Ewen CL, Kane KP, Bleackley RC (2012) A quarter century of granzymes[J]. Cell Death Differ 19(1):28–35
Liu Y, Fang Y, Chen X et al (2020) Gasdermin E-mediated target cell pyroptosis by CAR T cells triggers cytokine release syndrome[J]. Sci Immunol, 5(43)
Zhang Z, Zhang Y, Xia S et al (2020) Gasdermin E suppresses tumour growth by activating anti-tumour immunity[J]. Nature 579(7799):415–420
Newton K, Wickliffe KE, Maltzman A et al (2019) Activity of caspase-8 determines plasticity between cell death pathways[J]. Nature 575(7784):679–682
Zhang JY, Zhou B, Sun RY et al (2021) The metabolite α-KG induces GSDMC-dependent pyroptosis through death receptor 6-activated caspase-8[J]. Cell Res 31(9):980–997
Hou J, Zhao R, Xia W et al (2020) PD-L1-mediated gasdermin C expression switches apoptosis to pyroptosis in cancer cells and facilitates tumour necrosis[J]. Nat Cell Biol 22(10):1264–1275
Zhou Z, He H, Wang K et al (2020) Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells[J]. Science, 368(6494)
Chauhan D, Demon D, Vande Walle L et al (2022) GSDMD drives canonical inflammasome-induced neutrophil pyroptosis and is dispensable for NETosis[J]. EMBO Rep 23(10):e54277
Chao KL, Kulakova L, Herzberg O (2017) Gene polymorphism linked to increased Asthma and IBD risk alters gasdermin-B structure, a sulfatide and phosphoinositide binding protein. Proc Natl Acad Sci USA 114:E1128–E1137
Hou J, Wang S, Miao R et al (2023) Detection of gasdermin C-Mediated Cancer Cell Pyroptosis[J]. Methods Mol Biol 2641:135–146
Yang F, Bettadapura SN, Smeltzer MS et al (2022) Pyroptosis and pyroptosis-inducing cancer drugs[J]. Acta Pharmacol Sin 43(10):2462–2473
Xia X, Wang X, Cheng Z et al (2019) The role of pyroptosis in cancer: pro-cancer or pro-host?[J]. Cell Death Dis 10(9):650
Lu X, Guo T, Zhang X (2021) Pyroptosis in Cancer: friend or foe?[J]. Cancers (Basel), 13(14)
Wang F, Li G, Ning J et al (2018) Alcohol accumulation promotes esophagitis via pyroptosis activation[J]. Int J Biol Sci 14(10):1245–1255
Wang Q, Wang Y, Ding J et al (2020) A bioorthogonal system reveals antitumour immune function of pyroptosis[J]. Nature 579(7799):421–426
Galluzzi L, Buqué A, Kepp O et al (2017) Immunogenic cell death in cancer and Infectious disease[J]. Nat Rev Immunol 17(2):97–111
Aizawa E, Karasawa T, Watanabe S et al (2020) GSDME-Dependent incomplete pyroptosis permits selective IL-1α release under Caspase-1 Inhibition[J]. iScience 23(5):101070
De Vasconcelos NM, Van Opdenbosch N, Van Gorp H et al (2020) An Apoptotic Caspase Network Safeguards Cell Death Induction in Pyroptotic Macrophages[J]. Cell Rep 32(4):107959
Schwarzer R, Jiao H, Wachsmuth L et al (2020) FADD and Caspase-8 regulate gut homeostasis and inflammation by Controlling MLKL- and GSDMD-Mediated death of intestinal epithelial Cells[J]. Immunity 52(6):978–993e6
Wang L, Sharif H, Vora SM et al (2021) Structures and functions of the inflammasome engine[J]. J Allergy Clin Immunol 147(6):2021–2029
Shen E, Han Y, Cai C et al (2021) Low expression of NLRP1 is associated with a poor prognosis and immune infiltration in lung adenocarcinoma patients[J]. Aging 13(5):7570–7588
Williams TM, Leeth RA, Rothschild DE et al (2015) The NLRP1 inflammasome attenuates Colitis and colitis-associated tumorigenesis[J]. J Immunol 194(7):3369–3380
Salaro E, Rambaldi A, Falzoni S et al (2016) Involvement of the P2X7-NLRP3 axis in leukemic cell proliferation and death[J]. Sci Rep 6:26280
Zaki MH, Vogel P, Body-Malapel M et al (2010) IL-18 production downstream of the Nlrp3 inflammasome confers protection against colorectal Tumor formation[J]. J Immunol 185(8):4912–4920
Markowitz GJ, Yang P, Fu J et al (2016) Inflammation-dependent IL18 Signaling restricts Hepatocellular Carcinoma Growth by enhancing the Accumulation and Activity of Tumor-infiltrating Lymphocytes[J]. Cancer Res 76(8):2394–2405
Daley D, Mani VR, Mohan N et al (2017) NLRP3 signaling drives macrophage-induced adaptive immune suppression in pancreatic carcinoma[J]. J Exp Med 214(6):1711–1724
Sharma D, Malik A, Guy CS et al (2018) Pyrin Inflammasome regulates tight Junction Integrity to restrict Colitis and Tumorigenesis[J]. Gastroenterology 154(4):948–964e8
Terme M, Ullrich E, Aymeric L et al (2011) IL-18 induces PD-1-dependent immunosuppression in cancer[J]. Cancer Res 71(16):5393–5399
Dupaul-Chicoine J, Arabzadeh A, Dagenais M et al (2015) The Nlrp3 Inflammasome suppresses Colorectal Cancer Metastatic Growth in the liver by promoting natural killer cell tumoricidal Activity[J]. Immunity 43(4):751–763
Dmitrieva-Posocco O, Dzutsev A, Posocco DF et al (2019) Cell-type-specific responses to Interleukin-1 Control Microbial Invasion and Tumor-elicited inflammation in Colorectal Cancer[J]. Immunity 50(1):166–180e7
Luo X, Zhang X, Gan L et al (2018) The outer membrane protein Tp92 of Treponema pallidum induces human mononuclear cell death and IL-8 secretion[J]. J Cell Mol Med 22(12):6039–6054
Kim MS, Chang X, Yamashita K et al (2008) Aberrant promoter methylation and Tumor suppressive activity of the DFNA5 gene in colorectal carcinoma[J]. Oncogene 27(25):3624–3634
De Beeck KO, Van Laer L, Van Camp G (2012) DFNA5, a gene involved in hearing loss and cancer: a review[J]. Ann Otol Rhinol Laryngol 121(3):197–207
Yokomizo K, Harada Y, Kijima K et al (2012) Methylation of the DFNA5 gene is frequently detected in Colorectal cancer[J]. Anticancer Res 32(4):1319–1322
Wang CJ, Tang L, Shen DW et al (2013) The expression and regulation of DFNA5 in human hepatocellular carcinoma DFNA5 in hepatocellular carcinoma[J]. Mol Biol Rep 40(12):6525–6531
Teng JF, Mei QB, Zhou XG et al (2020) Polyphyllin VI induces caspase-1-Mediated pyroptosis via the induction of ROS/NF-κB/NLRP3/GSDMD Signal Axis in Non-small Cell Lung Cancer[J]. Cancers (Basel), 12(1)
Zhang R, Chen J, Mao L et al (2020) Nobiletin triggers reactive oxygen species-mediated pyroptosis through regulating Autophagy in Ovarian Cancer Cells[J]. J Agric Food Chem 68(5):1326–1336
Chen J, Wang Z, Yu S (2017) AIM2 regulates viability and apoptosis in human Colorectal cancer cells via the PI3K/Akt pathway[J]. Onco Targets Ther 10:811–817
Wei Q, Mu K, Li T et al (2014) Deregulation of the NLRP3 inflammasome in hepatic parenchymal cells during Liver cancer progression[J]. Lab Invest 94(1):52–62
Ma X, Guo P, Qiu Y et al (2016) Loss of AIM2 expression promotes hepatocarcinoma progression through activation of mTOR-S6K1 pathway[J]. Oncotarget 7(24):36185–36197
Janowski AM, Colegio OR, Hornick EE et al (2016) NLRC4 suppresses Melanoma Tumor progression independently of inflammasome activation[J]. J Clin Invest 126(10):3917–3928
Rogers C, Erkes DA, Nardone A et al (2019) Gasdermin pores permeabilize mitochondria to augment caspase-3 activation during apoptosis and inflammasome activation[J]. Nat Commun 10(1):1689
Wang F, Liu W, Ning J et al (2018) Simvastatin suppresses Proliferation and Migration in Non-small Cell Lung Cancer via Pyroptosis[J]. Int J Biol Sci 14(4):406–417
Wang WJ, Chen D, Jiang MZ et al (2018) Downregulation of gasdermin D promotes gastric cancer proliferation by regulating cell cycle-related proteins[J]. J Dig Dis 19(2):74–83
Yue E, Tuguzbaeva G, Chen X et al (2019) Anthocyanin is involved in the activation of pyroptosis in oral squamous cell carcinoma[J]. Phytomedicine 56:286–294
Nakamura K, Kassem S, Cleynen A et al (2018) Dysregulated IL-18 is a key driver of Immunosuppression and a possible therapeutic target in the Multiple Myeloma Microenvironment[J]. Cancer Cell 33(4):634–648e5
Zhao X, Zhang C, Hua M et al (2017) NLRP3 inflammasome activation plays a carcinogenic role through effector cytokine IL-18 in lymphoma[J]. Oncotarget 8(65):108571–108583
Lu F, Zhao Y, Pang Y et al (2021) NLRP3 inflammasome upregulates PD-L1 expression and contributes to immune suppression in lymphoma[J]. Cancer Lett 497:178–189
Zienolddiny S, Ryberg D, Maggini V et al (2004) Polymorphisms of the interleukin-1 beta gene are associated with increased risk of non-small cell Lung cancer[J]. Int J Cancer 109(3):353–356
Kong H, Wang Y, Zeng X et al (2015) Differential expression of inflammasomes in Lung cancer cell lines and tissues[J]. Tumour Biol 36(10):7501–7513
Zhai Z, Liu W, Kaur M et al (2017) NLRP1 promotes Tumor growth by enhancing inflammasome activation and suppressing apoptosis in metastatic melanoma[J]. Oncogene 36(27):3820–3830
Tsai CY, Lee TS, Kou YR et al (2009) Glucosamine inhibits IL-1beta-mediated IL-8 production in Prostate cancer cells by MAPK attenuation[J]. J Cell Biochem 108(2):489–498
Zhu Y, Zhu M, Lance P (2012) IL1β-mediated stromal COX-2 signaling mediates proliferation and invasiveness of colonic epithelial cancer cells[J]. Exp Cell Res 318(19):2520–2530
Fathima Hurmath K, Ramaswamy P, Nandakumar DN (2014) IL-1β microenvironment promotes proliferation, migration, and invasion of human glioma cells[J]. Cell Biol Int 38(12):1415–1422
Weichand B, Popp R, Dziumbla S et al (2017) S1PR1 on tumor-associated macrophages promotes lymphangiogenesis and Metastasis via NLRP3/IL-1β[J]. J Exp Med 214(9):2695–2713
Hergueta-Redondo M, Sarrió D, Molina-Crespo Á et al (2014) Gasdermin-B promotes invasion and Metastasis in Breast cancer cells[J]. PLoS ONE 9(3):e90099
Komiyama H, Aoki A, Tanaka S et al (2010) Alu-derived cis-element regulates tumorigenesis-dependent gastric expression of GASDERMIN B (GSDMB)[J]. Genes Genet Syst 85(1):75–83
Lutkowska A, Roszak A, Lianeri M et al (2017) Analysis of rs8067378 polymorphism in the risk of uterine Cervical Cancer from a Polish Population and its impact on Gasdermin B Expression[J]. Mol Diagn Ther 21(2):199–207
Molina-Crespo Á, Cadete A, Sarrio D et al (2019) Intracellular delivery of an antibody targeting Gasdermin-B reduces HER2 Breast Cancer Aggressiveness[J]. Clin Cancer Res 25(15):4846–4858
Wei J, Xu Z, Chen X et al (2020) Overexpression of GSDMC is a prognostic factor for predicting a poor outcome in lung adenocarcinoma[J]. Mol Med Rep 21(1):360–370
Wang Y, Kong H, Zeng X et al (2016) Activation of NLRP3 inflammasome enhances the proliferation and migration of A549 Lung cancer cells[J]. Oncol Rep 35(4):2053–2064
Zhang M, Jin C, Yang Y et al (2019) AIM2 promotes non-small-cell Lung cancer cell growth through inflammasome-dependent pathway[J]. J Cell Physiol 234(11):20161–20173
Gao J, Qiu X, Xi G et al (2018) Downregulation of GSDMD attenuates Tumor proliferation via the intrinsic mitochondrial apoptotic pathway and inhibition of EGFR/Akt signaling and predicts a good prognosis in non–small cell Lung cancer[J]. Oncol Rep 40(4):1971–1984
Kolb R, Phan L, Borcherding N et al (2016) Obesity-associated NLRC4 inflammasome activation drives Breast cancer progression[J]. Nat Commun 7:13007
Guo B, Fu S, Zhang J et al (2016) Targeting inflammasome/IL-1 pathways for cancer immunotherapy[J]. Sci Rep 6:36107
Wang H, Luo Q, Feng X et al (2018) NLRP3 promotes Tumor growth and Metastasis in human oral squamous cell carcinoma[J]. BMC Cancer 18(1):500
Wang H, Wang Y, Du Q et al (2016) Inflammasome-independent NLRP3 is required for epithelial-mesenchymal transition in colon Cancer cells[J]. Exp Cell Res 342(2):184–192
Li L, Liu Y (2015) Aging-related gene signature regulated by Nlrp3 predicts glioma progression[J]. Am J Cancer Res 5(1):442–449
Wang WH, Huang JQ, Zheng GF et al (2003) Non-steroidal anti-inflammatory drug use and the risk of gastric cancer: a systematic review and meta-analysis[J]. J Natl Cancer Inst 95(23):1784–1791
Sanui T, Takeshita M, Fukuda T et al (2017) Anti-CD14 antibody-treated neutrophils respond to LPS: possible involvement of CD14 upregulated by Anti-CD14 antibody Binding[J]. Immunol Invest 46(2):190–200
Bouis DA, Popova TG, Takashima A et al (2001) Dendritic cells phagocytose and are activated by Treponema pallidum[J]. Infect Immun 69(1):518–528
Hawley KL, Martín-Ruiz I, Iglesias-Pedraz JM et al (2013) CD14 targets complement receptor 3 to lipid rafts during phagocytosis of Borrelia burgdorferi[J]. Int J Biol Sci 9(8):803–810
Norgard MV, Arndt LL, Akins DR et al (1996) Activation of human monocytic cells by Treponema pallidum and Borrelia burgdorferi lipoproteins and synthetic lipopeptides proceeds via a pathway distinct from that of lipopolysaccharide but involves the transcriptional activator NF-kappa B[J]. Infect Immun 64(9):3845–3852
Ju X, Yang Z, Zhang H et al (2021) Role of pyroptosis in cancer cells and clinical applications[J]. Biochimie 185:78–86
Ball B, Zeidan A, Gore SD et al (2017) Hypomethylating agent combination strategies in myelodysplastic syndromes: hopes and shortcomings[J]. Leuk Lymphoma 58(5):1022–1036
Chen C, Wang B, Sun J et al (2015) DAC can restore expression of NALP1 to suppress Tumor growth in colon cancer[J]. Cell Death Dis 6(1):e1602
Lu H, Zhang S, Wu J et al (2018) Molecular targeted therapies elicit concurrent apoptotic and GSDME-Dependent pyroptotic Tumor cell Death[J]. Clin Cancer Res 24(23):6066–6077
Zhang J, Chen Y, He Q (2020) Distinct characteristics of dasatinib-induced pyroptosis in gasdermin E-expressing human Lung cancer A549 cells and neuroblastoma SH-SY5Y cells[J]. Oncol Lett 20(1):145–154
Zhang CC, Li CG, Wang YF et al (2019) Chemotherapeutic paclitaxel and cisplatin differentially induce pyroptosis in A549 Lung cancer cells via caspase-3/GSDME activation[J]. Apoptosis 24(3–4):312–325
Hu L, Chen M, Chen X et al (2020) Chemotherapy-induced pyroptosis is mediated by BAK/BAX-caspase-3-GSDME pathway and inhibited by 2-bromopalmitate[J]. Cell Death Dis 11(4):281
Li Q, Wang M, Zhang Y et al (2020) BIX-01294-enhanced chemosensitivity in nasopharyngeal carcinoma depends on autophagy-induced pyroptosis[J]. Acta Biochim Biophys Sin (Shanghai) 52(10):1131–1139
Wu M, Wang Y, Yang D et al (2019) A PLK1 kinase inhibitor enhances the chemosensitivity of cisplatin by inducing pyroptosis in oesophageal squamous cell carcinoma[J]. EBioMedicine 41:244–255
Yu J, Li S, Qi J et al (2019) Cleavage of GSDME by caspase-3 determines lobaplatin-induced pyroptosis in colon Cancer cells[J]. Cell Death Dis 10(3):193
Westbom C, Thompson JK, Leggett A et al (2015) Inflammasome Modulation by Chemotherapeutics in Malignant Mesothelioma[J]. PLoS ONE 10(12):e0145404
Tian W, Wang Z, Tang NN et al (2020) Ascorbic acid sensitizes colorectal carcinoma to the cytotoxicity of Arsenic Trioxide via promoting reactive oxygen species-dependent apoptosis and Pyroptosis[J]. Front Pharmacol 11:123
Hage C, Hoves S, Strauss L et al (2019) Sorafenib induces pyroptosis in macrophages and triggers natural killer cell-mediated cytotoxicity against Hepatocellular Carcinoma[J]. Hepatology 70(4):1280–1297
Draganov D, Han Z, Rana A et al (2021) Ivermectin converts cold tumors hot and synergizes with immune checkpoint blockade for treatment of Breast cancer[J]. NPJ Breast Cancer 7(1):22
Erkes DA, Cai W, Sanchez IM et al (2020) Mutant BRAF and MEK inhibitors regulate the Tumor Immune Microenvironment via Pyroptosis[J]. Cancer Discov 10(2):254–269
Zhou B, Zhang JY, Liu XS et al (2018) Tom20 senses iron-activated ROS signaling to promote Melanoma cell pyroptosis[J]. Cell Res 28(12):1171–1185
Yang W, Liu S, Li Y et al (2020) Pyridoxine induces monocyte-macrophages death as specific treatment of acute Myeloid leukemia[J]. Cancer Lett 492:96–105
Gao W, Wang X, Zhou Y et al (2022) Autophagy, ferroptosis, pyroptosis, and necroptosis in Tumor immunotherapy[J]. Signal Transduct Target Ther 7(1):196
Xu-Monette ZY, Zhou J, Young KH (2018) PD-1 expression and clinical PD-1 blockade in B-cell lymphomas[J]. Blood 131(1):68–83
Wang X, Guo G, Guan H et al (2019) Challenges and potential of PD-1/PD-L1 checkpoint blockade immunotherapy for glioblastoma[J]. J Exp Clin Cancer Res 38(1):87
Huang MY, Jiang XM, Wang BL et al (2021) Combination therapy with PD-1/PD-L1 blockade in non-small cell Lung cancer: strategies and mechanisms[J]. Pharmacol Ther 219:107694
Han G, Yang G, Hao D et al (2021) 9p21 loss confers a cold Tumor immune microenvironment and primary resistance to immune checkpoint therapy[J]. Nat Commun 12(1):5606
Kumagai S, Togashi Y, Kamada T et al (2020) The PD-1 expression balance between effector and regulatory T cells predicts the clinical efficacy of PD-1 blockade therapies[J]. Nat Immunol 21(11):1346–1358
Lu C, Guo C, Chen H et al (2020) A novel chimeric PD1-NKG2D-41BB receptor enhances antitumor activity of NK92 cells against human Lung cancer H1299 cells by triggering pyroptosis[J]. Mol Immunol 122:200–206
Okondo MC, Johnson DC, Sridharan R et al (2017) DPP8 and DPP9 inhibition induces pro-caspase-1-dependent monocyte and macrophage pyroptosis[J]. Nat Chem Biol 13(1):46–53
Johnson DC, Taabazuing CY, Okondo MC et al (2018) DPP8/DPP9 inhibitor-induced pyroptosis for treatment of acute Myeloid leukemia[J]. Nat Med 24(8):1151–1156
Bai B, Chen Q, Jing R et al (2021) Molecular basis of Prostate Cancer and Natural products as potential chemotherapeutic and chemopreventive Agents[J]. Front Pharmacol 12:738235
Liu Y, Yang S, Wang K et al (2020) Cellular senescence and cancer: focusing on traditional Chinese medicine and natural products[J]. Cell Prolif 53(10):e12894
Xiang Y, Guo Z, Zhu P et al (2019) Traditional Chinese medicine as a cancer treatment: modern perspectives of ancient but advanced science[J]. Cancer Med 8(5):1958–1975
Wang Y, Zhang Q, Chen Y et al (2020) Antitumor effects of immunity-enhancing traditional Chinese medicine[J]. Biomed Pharmacother 121:109570
Chu Q, Jiang Y, Zhang W et al (2016) Pyroptosis is involved in the pathogenesis of human hepatocellular carcinoma[J]. Oncotarget 7(51):84658–84665
Zhang X, Zhang P, An L et al (2020) Miltirone induces cell death in hepatocellular carcinoma cell through GSDME-dependent pyroptosis[J]. Acta Pharm Sin B 10(8):1397–1413
Chen YF, Qi HY, Wu FL (2018) Euxanthone exhibits anti-proliferative and anti-invasive activities in hepatocellular carcinoma by inducing pyroptosis: preliminary results[J]. Eur Rev Med Pharmacol Sci 22(23):8186–8196
Yuan R, Zhao W, Wang QQ et al (2021) Cucurbitacin B inhibits non-small cell Lung cancer in vivo and in vitro by triggering TLR4/NLRP3/GSDMD-dependent pyroptosis[J]. Pharmacol Res 170:105748
Sannino F, Sansone C, Galasso C et al (2018) Pseudoalteromonas haloplanktis TAC125 produces 4-hydroxybenzoic acid that induces pyroptosis in human A459 lung adenocarcinoma cells[J]. Sci Rep 8(1):1190
Zou J, Yang Y, Yang Y et al (2018) Polydatin suppresses proliferation and Metastasis of non-small cell Lung cancer cells by inhibiting NLRP3 inflammasome activation via NF-κB pathway[J]. Biomed Pharmacother 108:130–136
Yu Q, Zhang M, Ying Q et al (2019) Decrease of AIM2 mediated by luteolin contributes to non-small cell Lung cancer treatment[J]. Cell Death Dis 10(3):218
Liang J, Zhou J, Xu Y et al (2020) Osthole inhibits ovarian carcinoma cells through LC3-mediated autophagy and GSDME-dependent pyroptosis except for apoptosis[J]. Eur J Pharmacol 874:172990
Tan JQ, Li Z, Chen G et al (2022) The natural compound from Garcinia Bracteata mainly induces GSDME-mediated pyroptosis in Esophageal cancer cells[J]. Phytomedicine 102:154142
Zhang Y, Li M, Gao X et al (2019) Nanotechnology in cancer diagnosis: progress, challenges and opportunities[J]. J Hematol Oncol 12(1):137
Nadeem S, Yang C, Du Y, Li F, Chen Z, Zhou Y et al (2021) A virus-spike tumor-activatable pyroptotic agent. Small 17:e2006599
Yao J, Feng X, Dai X et al (2022) TMZ magnetic temperature-sensitive liposomes-mediated magnetothermal chemotherapy induces pyroptosis in glioblastoma[J]. Nanomedicine 43:102554
Li J, Luo G, Zhang C et al (2022) In situ injectable hydrogel-loaded Drugs induce anti-tumor immune responses in Melanoma immunochemotherapy[J]. Mater Today Bio 14:100238
Balahura LR, Dinescu S, Balaș M et al (2021) Cellulose nanofiber-based hydrogels embedding 5-FU promote pyroptosis activation in Breast Cancer cells and support human adipose-derived stem cell proliferation, opening new perspectives for breast tissue Engineering[J]. Pharmaceutics, 13(8)
Hu J, Dong Y, Ding L et al (2019) Local delivery of arsenic trioxide nanoparticles for hepatocellular carcinoma treatment[J]. Signal Transduct Target Ther 4:28
Yu Z, Cao W, Han C et al (2022) Biomimetic Metal-Organic Framework nanoparticles for Synergistic Combining of SDT-Chemotherapy Induce pyroptosis in gastric Cancer[J]. Front Bioeng Biotechnol 10:796820
Zhao P, Wang M, Chen M et al (2020) Programming cell pyroptosis with biomimetic nanoparticles for solid Tumor immunotherapy[J]. Biomaterials 254:120142
Serna N, Álamo P, Ramesh P et al (2020) Nanostructured toxins for the selective destruction of drug-resistant human CXCR4(+) Colorectal cancer stem cells[J]. J Control Release 320:96–104
Ploetz E, Zimpel A, Cauda V et al (2020) Metal-Organic Framework nanoparticles induce pyroptosis in cells controlled by the Extracellular pH[J]. Adv Mater 32(19):e1907267
Jiang W, Yin L, Chen H et al (2019) NaCl nanoparticles as a Cancer Therapeutic[J]. Adv Mater 31(46):e1904058
Ding B, Sheng J, Zheng P et al (2021) Biodegradable Upconversion nanoparticles induce pyroptosis for Cancer Immunotherapy[J]. Nano Lett 21(19):8281–8289
Clerc P, Jeanjean P, Hallali N et al (2018) Targeted magnetic intra-lysosomal hyperthermia produces lysosomal reactive oxygen species and causes Caspase-1 dependent cell death[J]. J Control Release 270:120–134
Nadeem S, Yang C, Du Y et al (2021) A virus-spike tumor-activatable Pyroptotic Agent[J]. Small 17(8):e2006599
Liu Y, Zhen W, Wang Y et al (2020) Na(2)S(2)O(8) Nanoparticles Trigger Antitumor Immunotherapy through reactive oxygen species Storm and surge of Tumor Osmolarity[J]. J Am Chem Soc 142(52):21751–21757
CHEN D R, GAO Y, XIAO Y et al (2022) Engineered nanogels simultaneously implement HDAC inhibition and chemotherapy to boost antitumor immunity via pyroptosis[J]. Appl Mater Today, 26
XIONG H G, MA X B, WANG X L et al (2021) Inspired epigenetic modulation synergy with Adenosine Inhibition elicits pyroptosis and Potentiates Cancer Immunotherapy[J]. Adv Funct Mater, 31(20)
Wu D, Wang S, Yu G et al (2021) Cell death mediated by the pyroptosis pathway with the aid of nanotechnology: prospects for Cancer Therapy[J]. Angew Chem Int Ed Engl 60(15):8018–8034
Zhao H, Song Q, Zheng C, Zhao B, Wu L, Feng Q et al (2020) Implantable bioresponsive nanoarray enhances postsurgical immunotherapy by activating pyroptosis and remodeling Tumor microenvironment. Adv Funct Mater 30:2005747
Xiao Y, Zhang T, Ma X, Yang QC, Yang LL, Yang SC et al (2021) Microenvironment-responsive prodrug-induced pyroptosis boosts cancer immunotherapy. Adv Sci (Weinh) 8:e2101840
Fan JX, Deng RH, Wang H et al (2019) Epigenetics-based Tumor cells pyroptosis for enhancing the Immunological Effect of Chemotherapeutic Nanocarriers[J]. Nano Lett 19(11):8049–8058
Elrayess RA, Mohallal ME, Mobarak YM et al (2021) Scorpion venom antimicrobial peptides induce Caspase-1 dependant pyroptotic cell Death[J]. Front Pharmacol 12:788874
Derangère V, Chevriaux A, Courtaut F et al (2014) Liver X receptor β activation induces pyroptosis of human and murine colon Cancer cells[J]. Cell Death Differ 21(12):1914–1924
Pizato N, Luzete BC, Kiffer L et al (2018) Omega-3 docosahexaenoic acid induces pyroptosis cell death in triple-negative breast cancer cells[J]. Sci Rep, 8(1): 1952
Wei Q, Zhu R, Zhu J et al (2019) E2-Induced activation of the NLRP3 inflammasome triggers pyroptosis and inhibits autophagy in HCC Cells[J]. Oncol Res 27(7):827–834
Qiao L, Wu X, Zhang J et al (2019) α-NETA induces pyroptosis of epithelial Ovarian cancer cells through the GSDMD/caspase-4 pathway[J]. Faseb j 33(11):12760–12767
Wang L, Li K, Lin X et al (2019) Metformin induces human esophageal carcinoma cell pyroptosis by targeting the miR-497/PELP1 axis[J]. Cancer Lett 450:22–31
Jia Y, Cui R, Wang C et al (2020) Metformin protects against intestinal ischemia-reperfusion injury and cell pyroptosis via TXNIP-NLRP3-GSDMD pathway[J]. Redox Biol 32:101534
Wu M, Liu X, Chen H, Duan Y, Liu J, Pan Y et al (2021) Activation of pyroptosis by membrane-anchoring AIE photosensitizer design: new prospect for photodynamic cancer cell ablation. Angew Chem Int Ed Engl 60:9093–9098
Hu J, Dong Y, Ding L, Dong Y, Wu Z, Wang W et al (2019) Local delivery of arsenic trioxide nanoparticles for hepatocellular carcinoma treatment. Signal Transduct Target Ther 4:28
Tan YF, Wang M, Chen ZY, Wang L, Liu XH (2020) Inhibition of BRD4 prevents proliferation and epithelial-mesenchymal transition in renal cell carcinoma via NLRP3 inflammasome-induced pyroptosis. Cell Death Dis 11(4):239
Acknowledgements
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Funding
This study was financially supported by the National Natural Science Foundation of China (grant No. 81803916 and 82374223) and Natural Science Foundation of Jiangsu Province (grant No. BK20180128).
Author information
Authors and Affiliations
Contributions
Q Chen and Y Sun conceived the structure of manuscript and drafted the manuscript; Q Chen, S Wang and J Xu discussed and revised the manuscript. All authors read and approved final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Q., Sun, Y., Wang, S. et al. New prospects of cancer therapy based on pyroptosis and pyroptosis inducers. Apoptosis 29, 66–85 (2024). https://doi.org/10.1007/s10495-023-01906-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01906-5