Abstract
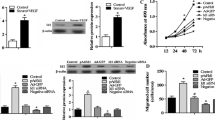
Angiogenesis is a complicated process including endothelial cell proliferation, migration and tube formation. AAMP plays a role in regulating cell migration of multiple cell types. The purpose of this study was to investigate whether AAMP regulates angiogenesis, and to clarify the role of AAMP in the VEGF-induced angiogenesis. We found that AAMP expressed in multiple cell types and mainly localized in cytoplasm and membrane in vascular endothelial cells. Using tube formation assay in vitro and aortic ring assay, siRNA-mediated knockdown and antibody blockade of AAMP impaired VEGF-induced endothelial cell tube formation and aortic ring angiogenic sprouting. Mechanistic studies showed that AAMP expression was significantly upregulated by VEGF in a concentration and time-dependent manner. Moreover, VEGF recruited AAMP to the cell membrane protrusions. AAMP regulates angiogenesis by mediating the spreading and migration of vascular endothelial cells. AAMP knock-down reduced VEGF-induced actin stress fibers and collagen gel contraction. Furthermore, we identified RhoA/Rho kinase signaling as an important factor that contributes to the action of AAMP in regulating endothelial cell migration and angiogenesis. Altogether, these data demonstrated the critical role of AAMP in angiogenesis and suggested blocking AAMP could serve as a potential therapeutic strategy for angiogenesis-related diseases.






Similar content being viewed by others
References
Adeyinka, A., E. Emberley, Y. Niu, L. Snell, L. C. Murphy, H. Sowter, C. C. Wykoff, A. L. Harris, and P. R. H. Watson. Analysis of gene expression in ductal carcinoma in situ of the breast. Clin. Cancer Res. 8:3788–3795, 2002.
Allander, S. V., N. N. Nupponen, M. Ringnér, G. Hostetter, G. W. Maher, N. Goldberger, Y. Chen, J. Carpten, A. G. Elkahloun, and P. S. Meltzer. Gastrointestinal stromal tumors with KIT mutations exhibit a remarkably homogeneous gene expression profile. Cancer Res. 61:8624–8628, 2001.
Baker, M., S. D. Robinson, T. Lechertier, P. R. Barber, B. Tavora, G. D’Amico, D. T. Jones, B. Vojnovic, and K. Hodivala-Dilke. Use of the mouse aortic ring assay to study angiogenesis. Nat. Protoc. 7(1):89–104, 2012.
Beckner, M. E., H. C. Krutzsch, M. L. Stracke, S. T. Williams, J. A. Gallardo, and L. A. Liotta. Identification of a new immunoglobulin superfamily protein expressed in blood vessels with a heparin-binding consensus sequence. Cancer Res. 55:2140–2149, 1995.
Beckner, M. E., and L. A. Liotta. AAMP, a conserved protein with immunoglobulin and WD40 domains, regulates endothelial tube formation in vitro. Lab. Invest. 75:97–107, 1996.
Beckner, M. E., V. A. Peterson, and D. E. Moul. Angio-associated migratory cell protein is expressed as an extracellular protein by blood-vessel-associated mesenchymal cells. Microvasc. Res. 57:347–352, 1999.
Beckner, M. E., S. Jagannathan, and V. A. Peterson. Extracellular angio-associated migratory cell protein plays a positive role in angiogenesis and is regulated by astrocytes in coculture. Microvasc. Res. 63:259–269, 2002.
Bielig, H., B. Zurek, A. Kutsch, M. Menning, D. J. Philpott, P. J. Sansonetti, and T. A. Kufer. A function for AAMP in Nod2-mediated NF-kappaB activation. Mol. Immunol. 46(13):2647–2654, 2009.
Birk, D. M., J. Barbato, L. Mureebe and R.A. Chaer. Current insights on the biology and clinical aspects of VEGF regulation. Vasc. Endovascular. Surg. 42(6):517-530, 2008-2009.
Blanchoin, L., R. Boujemaa-Paterski, C. Sykes, and J. Plastino. Actin dynamics, architecture, and mechanics in cell motility. Physiol. Rev. 94(1):235–263, 2014.
Blanco, R., and H. Gerhardt. VEGF and Notch in tip and stalk cell selection. Cold Spring Harb. Perspect. Med. 3(1):a006569, 2013.
Bryan, B. A., E. Dennstedt, D. C. Mitchell, T. E. Walshe, K. Noma, R. Loureiro, M. Saint-Geniez, J. P. Campaigniac, J. K. Liao, and P. A. D’Amore. RhoA/ROCK signaling is essential for multiple aspects of VEGF-mediated angiogenesis. FASEB J. 24(9):3186–3195, 2010.
Carmeliet, P., and R. K. Jain. Molecular mechanisms and clinical applications of angiogenesis. Nature 473(7347):298–307, 2011.
Chen, W., K. Mao, Z. Liu, and A. T. Dinh-Xuan. The role of the RhoA/Rho kinase pathway in angiogenesis and its potential value in prostate cancer. Oncol. Lett. 8(5):1907–1911, 2014.
Chi, Q., T. Yin, H. Gregersen, X. Deng, Y. Fan, J. Zhao, D. Liao, and G. Wang. Rear actomyosin contractility-driven directional cell migration in three-dimensional matrices: a mechano-chemical coupling mechanism. J. R. Soc. Interface 11(95):20131072, 2014.
Ferrara, N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am. J. Physiol. Cell Physiol. 280(6):C1358–C1366, 2001.
Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1(1):27–31, 1995.
Fong, G. H., J. Rossant, M. Gertsenstein, and M. L. Breitman. Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 376:66–70, 1995.
Frelin, C., A. Ladoux, and G. D’angelo. Vascular endothelial growth factors and angiogenesis. Ann. Endocrinol (Paris) 61:70–74, 2000.
Hoang, M. V., M. C. Whelan, and D. R. Senger. Rho activity critically and selectively regulates endothelial cell organization during angiogenesis. Proc. Natl. Acad. Sci. USA 101(7):1874–1879, 2004.
Holvoet, P., and P. Sinnaeve. Angio-associated migratory cell protein and smooth muscle cell migration in development of restenosis and atherosclerosis. J. Am. Coll. Cardiol. 52:312–314, 2008.
Lamalice, L., F. Le Boeuf, and J. Huot. Endothelial cell migration during angiogenesis. Circ. Res. 100(6):782–794, 2007.
Liekens, S., E. De Clercq, and J. Neyts. Angiogenesis: regulators and clinical applications. Biochem. Pharmacol. 61:253–270, 2001.
Machacek, M., L. Hodgson, C. Welch, H. Elliott, O. Pertz, P. Nalbant, A. Abell, G. L. Johnson, K. M. Hahn, and G. Danuser. Coordination of Rho GTPase activities during cell protrusion. Nature 461(7260):99–103, 2009.
Ngo, P., P. Ramalingam, J. A. Phillips, and G. T. Furuta. Collagen gel contraction assay. Methods Mol. Biol. 341:103–109, 2006.
Pertz, O., L. Hodgson, R. L. Klemke, and K. M. Hahn. Spatiotemporal dynamics of RhoA activity in migrating cells. Nature 440(7087):1069–1072, 2006.
Phng, L. K., F. Stanchi, and H. Gerhardt. Filopodia are dispensable for endothelial tip cell guidance. Development 140(19):4031–4040, 2013.
Potente, M., H. Gerhardt, and P. Carmeliet. Basic and therapeutic aspects of angiogenesis. Cell 146(6):873–887, 2011.
Qiu, J., G. Wang, Y. Zheng, J. Hu, Q. Peng, and T. Yin. Coordination of Id1 and p53 activation by oxidized LDL regulates endothelial cell proliferation and migration. Ann. Biomed. Eng. 39(12):2869–2878, 2011.
Qiu, J., G. Wang, Q. Peng, J. Hu, X. Luo, Y. Zheng, Y. Teng, and C. Tang. Id1 induces tubulogenesis by regulating endothelial cell adhesion and cytoskeletal organization through β1-integrin and Rho-kinase signalling. Int. J. Mol. Med. 28(4):543–548, 2011.
Qiu, J., Q. Peng, Y. Zheng, J. Hu, X. Luo, Y. Teng, T. Jiang, T. Yin, C. Tang, and G. Wang. OxLDL stimulates Id1 nucleocytoplasmic shuttling in endothelial cell angiogenesis via PI3 K pathway. Biochim. Biophys. Acta. 1821(10):1361–1369, 2012.
Reid, H. M., K. Wikström, D. J. Kavanagh, E. P. Mulvaney, and B. T. Kinsella. Interaction of angio-associated migratory cell protein with the TPα and TPβ isoforms of the human thromboxane A2 receptor. Cell Signal 23(4):700–717, 2011.
Reymann, A. C., R. Boujemaa-Paterski, J. L. Martiel, C. Guérin, W. Cao, H. F. Chin, E. M. De La Cruz, M. Théry, and L. Blanchoin. Actin network architecture can determine myosin motor activity. Science 336(6086):1310–1314, 2012.
Sakakura, K., M. Nakano, F. Otsuka, E. Ladich, F. D. Kolodgie, and R. Virmani. Pathophysiology of atherosclerosis plaque progression. Heart Lung Circ. 22(6):399–411, 2013.
Sharghi-Namini, S., E. Tan, L. L. Ong, R. Ge, and H. H. Asada. Dll4-containing exosomes induce capillary sprout retraction in a 3D microenvironment. Sci. Rep. 4:4031, 2014.
van Nieuw Amerongen, G. P., M. A. Vermeer, P. Nègre-Aminou, J. Lankelma, J. J. Emeis, and V. W. van Hinsbergh. Simvastatin improves disturbed endothelial barrier function. Circulation 102(23):2803–2809, 2000.
van Nieuw Amerongen, G. P., P. Koolwijk, A. Versteilen, and V. W. van Hinsbergh. Involvement of RhoA/Rho kinase signaling in VEGF-induced endothelial cell migraton and angiogenesis in vitro. Arterioscler. Thromb. Vasc. Biol. 23(2):211–217, 2003.
Vogt, F., A. Zernecke, M. Beckner, N. Krott, A. K. Bosserhoff, R. Hoffmann, M. A. Zandvoort, T. Jahnke, M. Kelm, C. Weber, and R. Blindt. Blockade of angio-associated migratory cell protein inhibits smooth muscle cell migration and neointima formation in accelerated atherosclerosis. J. Am. Coll. Cardiol. 52:302–311, 2008.
Acknowledgement
This study was supported by grants from the National Natural Science Foundation of China (31370949, 11332003, 81400329), the Fundamental Research Funds for the Central Universities (CQDXWL-2012-120), Chongqing Science and Technology Commission (cstc2013kjrc-ljrccj10003) and we also thanks for the support from the Chongqing Engineering Laboratory in Vascular Implants,the National “111 Plan” Base (B06023) and the Public Experiment Center of State Bioindustrial Base (Chongqing), China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Michael Gower oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, J., Qiu, J., Zheng, Y. et al. AAMP Regulates Endothelial Cell Migration and Angiogenesis Through RhoA/Rho Kinase Signaling. Ann Biomed Eng 44, 1462–1474 (2016). https://doi.org/10.1007/s10439-015-1442-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-015-1442-0