Abstract
Objectives
The aim of this study was to compare the different responses to hypocrellin B (HB)-mediated sonodynamic treatment between human gastric adenocarcinoma cell line SGC7901 and SGC7901/ADR.
Methods
Tumor cells in culture dishes (35-mm diameter) were exposed to planar ultrasound at an intensity of 0.5 W/cm2 for 60 s combined with/without 2.5 μM HB. Cell viability was determined by MTT and Guava ViaCount assay. Production of reactive oxygen species (ROS) and destabilization of the mitochondrial membrane potential were assessed by flow cytometry. Apoptosis was analyzed using annexin-PE/7-amino-actinomycin D staining. The cell membrane integrity was estimated by isothiocyanate–dextran (FD500) uptake assay. Ultrastructural alterations on the membrane surface were observed by scanning electron microscopy. The membrane fluidity was also compared between the two cell lines using spectrophotometry.
Results
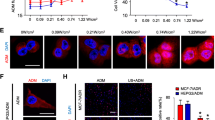
Compared with SGC7901 cells, HB-mediated sonodynamic therapy (HB-SDT) showed higher cytotoxic in SGC7901/ADR cells at the same treatment doses. Abundant intracellular ROS, a decrease in the mitochondrial membrane potential, and an increased rate of apoptosis were detected in the SDT group of both cell lines, wherein SGC7901/ADR cells showed a much more higher rate. Cell membrane permeability was remarkably enhanced after HB-SDT application. In addition, relatively severe cell damage was observed under scanning electron microscopy after HB-SDT treatment in SGC7901/ADR cells compared with SGC7901 cells.
Conclusions
These results suggest that HB-SDT could induce apoptosis in SGC7901 and SGC7901/ADR cells via production of ROS. SGC7901/ADR was found to be more sensitive to HB-SDT than SGC7901 cells under the same experimental condition. Meanwhile, a noteworthy difference in cell membrane injury between SGC7901 and SGC7901/ADR cells was detected. The decreased membrane fluidity in SGC7901/ADR cells may be one of the reasons for its increased membrane damage.










Similar content being viewed by others
References
Hartgrink HH, Jansen EP, van Grieken NC, et al. Gastric cancer. Lancet. 2009;374:477–90.
Mu J, Liu T, Jiang L, et al. The traditional chinese medicine baicalein potently inhibits gastric cancer cells. J Cancer. 2016;7:453–61.
Parkin DM, Pisani P, Ferlay MJ. Global cancer statistics. CA Cancer J Clin. 1999;49:33–64.
Wang P, Li Z, Liu H, et al. MicroRNA-126 increases chemosensitivity in drug-resistant gastric cancer cells by targeting EZH2. Biochem Biophys Res Commun. 2016;479:91–6.
Sun W, Lv C, Zhu T, et al. Ophiobolin-O reverses adriamycin resistance via cell cycle arrest and apoptosis sensitization in adriamycin-resistant human breast carcinoma (MCF-7/ADR) cells. Mar Drugs. 2013;11:4570–84.
Li C, Zhang K, Wang P, et al. Sonodynamic antitumor effect of a novel sonosensitizer on S180 solid tumor. Biopharm Drug Dispos. 2014;35:50–9.
Li Q, Wang X, Wang P, et al. Efficacy of chlorin e6-mediated sono-photodynamic therapy on 4T1 cells. Cancer Biother Radiopharm. 2014;29:42–52.
Li Y, Wang P, Wang X, et al. Involvement of mitochondrial and reactive oxygen species in the sonodynamic toxicity of chlorin e6 in human leukemia K562 cells. Ultrasound Med Biol. 2014;40:990–1000.
Jia Y, Yuan W, Zhang K, et al. Comparison of cell membrane damage induced by the therapeutic ultrasound on human breast cancer MCF-7 and MCF-7/ADR cells. Ultrason Sonochem. 2015;26:128–35.
Su X, Chen Y, Wang X, et al. PpIX induces mitochondria-related apoptosis in murine leukemia L1210 cells. Drug Chem Toxicol. 2014;37:348–56.
Wang H, Wang X, Wang P, et al. Ultrasound enhances the efficacy of chlorin E6-mediated photodynamic therapy in MDA-MB-231 cells. Ultrasound Med Biol. 2013;39:1713–24.
Shen S, Wu L, Liu J, et al. Core-shell structured Fe3O4@TiO2-doxorubicin nanoparticles for targeted chemo-sonodynamic therapy of cancer. Int J Pharm. 2015;486:380–8.
Wang H, Wang P, Li L, et al. Microbubbles enhance the antitumor effects of sinoporphyrin sodium mediated sonodynamic therapy both in vitro and in vivo. Int J Biol Sci. 2015;11:1401–9.
Yoshida T, Kondo T, Ogawa R, et al. Combination of doxorubicin and low-intensity ultrasound causes a synergistic enhancement in cell killing and an additive enhancement in apoptosis induction in human lymphoma U937 cells. Cancer Chemother Pharmacol. 2008;61:559–67.
Jiang Y, Leung AW, Wang X, et al. Effect of photodynamic therapy with hypocrellin B on apoptosis, adhesion, and migration of cancer cells. Int J Radiat Biol. 2014;90:575–9.
Jiang Y, Xia X, Leung AW, et al. Apoptosis of breast cancer cells induced by hypocrellin B under light-emitting diode irradiation. Photodiagnosis Photodyn Ther. 2012;9:337–43.
Jiang Y, Leung AW, Wang X, et al. Inactivation of Staphylococcus aureusby photodynamic action of hypocrellin B. Photodiagnosis Photodyn Ther. 2013;10:600–6.
Wang P, Xu CS, Xu J, et al. Hypocrellin B enhances ultrasound-induced cell death of nasopharyngeal carcinoma cells. Ultrasound Med Biol. 2010;36:336–42.
Wang X, Leung AW, Jiang Y, et al. Hypocrellin B-mediated sonodynamic action induces apoptosis of hepatocellular carcinoma cells. Ultrasonics. 2012;52:543–6.
Wang X, Ip M, Leung AW, et al. Sonodynamic action of hypocrellin B on methicillin-resistant Staphylococcus aureus. Ultrasonics. 2016;65:137–44.
Bai M, Shen M, Teng Y, et al. Enhanced therapeutic effect of Adriamycin on multidrug resistant breast cancer by the ABCG2-siRNA loaded polymeric nanoparticles assisted with ultrasound. Oncotarget. 2015;6:43779–90.
Wang X, Hu J, Zhang S, et al. Analysis of the in vivo and in vitro effects of photodynamic therapy on breast cancer by using a sensitizer, sinoporphyrin sodium. Theranostics. 2015;7:772–86.
Wang ZB, Liu YQ, Zhang Y, et al. Reactive oxygen species, but not mitochondrial membrane potential, is associated with radiation-induced apoptosis of AHH-1 human lymphoblastoid cells. Cell Biol Int. 2007;31:1353–8.
Wang X, Leung AW, Hua H, et al. Sonodynamic action of hypocrellin B on biofilm-producing Staphylococcus epidermidis in planktonic condition. J Acoust Soc Am. 2015;138:2548–53.
Krishna R, Mayer LD. Multidrug resistance (MDR) in cancer. Mechanisms, reversal using modulators of MDR and the role of MDR modulators in influencing the pharmacokinetics of anticancer drugs. Eur J Pharm Sci. 2000;11:265–83.
Wang H, Wang X, Zhang S, et al. Sinoporphyrin sodium, a novel sensitizer, triggers mitochondrial-dependent apoptosis in ECA-109 cells via production of reactive oxygen species. Int J Nanomed. 2014;2014:3077–90.
Escoffre JM, Piron J, Novell A, et al. Doxorubicin delivery into tumor cells with ultrasound and microbubbles. Mol Pharm. 2011;8:799–806.
Tang W, Fan W, Liu Q, et al. The role of p53 in the response of tumor cells to sonodynamic therapy in vitro. Ultrasonics. 2011;51:777–85.
Costley D, Mc Ewan C, Fowley C, et al. Treating cancer with sonodynamic therapy: a review. Int J Hyperthermia. 2015;31:107–17.
Zheng X, Wu J, Shao Q, et al. Apoptosis of THP-1 macrophages induced by pseudohypericin-mediated sonodynamic therapy through the mitochondria-caspase pathway. Cell Physiol Biochem. 2016;38:545–57.
Yumita N, Iwase Y, Nishi K, et al. Involvement of reactive oxygen species in sonodynamically induced apoptosis using a novel porphyrin derivative. Theranostics. 2012;2:880–8.
Li Q, Liu Q, Wang P, et al. The effects of Ce6-mediated sono-photodynamic therapy on cell migration, apoptosis and autophagy in mouse mammary 4T1 cell line. Ultrasonics. 2014;54:981–9.
Wang X, Luo J, Leung AW, et al. Hypocrellin B in hepatocellular carcinoma cells: subcellular localization and sonodynamic damage. Int J Radiat Biol. 2015;91:1–22.
Miller DL, Dou C. Membrane damage thresholds for pulsed or continuous ultrasound in phagocytic cells loaded with contrast agent gas bodies. Ultrasound Med Biol. 2004;30:405–11.
Hao Q, Liu Q, Wang X, et al. Membrane damage effect of therapeutic ultrasound on Ehrlich ascitic tumor cells. Cancer Biother Radiopharm. 2009;24:41–8.
Yang L, Wei DD, Chen Z, et al. Reversal of multidrug resistance in human breast cancer cells by Curcuma wenyujin and Chrysanthemum indicum. Phytomedicine. 2011;18:710–8.
Boutin C, Roche Y, Millot C, et al. High heterogeneity of plasma membrane microfluidity in multidrug-resistant cancer cells. J Biomed Opt. 2009;14:034030.
Hassan MA, Furusawa Y, Minemura M, et al. Ultrasound-induced new cellular mechanism involved in drug resistance. PLoS One. 2012;7:e48291.
Wang E, Lee MD, Dunn KW. Lysosomal accumulation of drugs in drug-sensitive MES-SA but not multidrug-resistant MES-SA/Dx5 uterine sarcoma cells. J Cell Physiol. 2000;184:263–74.
Tang W, Liu Q, Wang X, et al. Membrane fluidity altering and enzyme inactivating in sarcoma 180 cells post the exposure to sonoactivated hematoporphyrin in vitro. Ultrasonics. 2008;48:66–73.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 81872497), the Fundamental Research Funds for the Central Universities (No. GK201602003, 2016TS056), and the Natural Science Foundation of Shaanxi Province (No. 2017JM8004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical statements
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
About this article
Cite this article
Liu, Y., Bai, H., Wang, H. et al. Comparison of hypocrellin B-mediated sonodynamic responsiveness between sensitive and multidrug-resistant human gastric cancer cell lines. J Med Ultrasonics 46, 15–26 (2019). https://doi.org/10.1007/s10396-018-0899-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10396-018-0899-5