Abstract
Background
Marmesin, an important coumarin isolated from Broussonetia kazinoki, has been proposed to possess many pharmacological activities including anti-tumor activity. However, the anti-cancer effect of marmesin on esophageal cancer (EC) has not been characterized. The study aimed to explore the anti-cancer role of marmesin using EC cell lines in vitro.
Methods and results
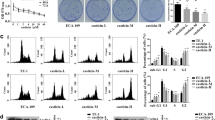
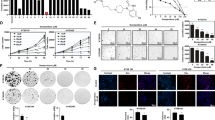
Cell proliferation was evaluated by CCK-8 and Edu cell proliferation assays and apoptosis was detected by TUNEL assay. Western blot analysis was used to determine the expression of Ki67, proliferating cell nuclear antigen (PCNA), Bcl-2, Bax, phosphatidylinositol 3-kinase (PI3K), phosphoryrated-PI3K (p-PI3K), protein kinase B (Akt), and phosphoryrated-Akt (p-Akt). The mechanism of action of marmesin was analyzed using network pharmacology approach. Marmesin exhibited anti-proliferative effect against EC cells, which was further confirmed by the reduced expression of Ki67 and PCNA. Marmesin exerted pro-apoptotic activity on EC cells by downregulating Bcl-2 and upregulating Bax. According to the results from network pharmacology approach, we speculated that PI3K/Akt pathway may participate in the effect of marmesin on EC cells. Additionally, the PI3K/Akt pathway was suppressed by marmesin in EC cells. Moreover, forced expression of Akt reversed the inhibition of cell proliferation and induction of apoptosis induced by marmesin in EC cells.
Conclusions
Marmesin exerted anti-cancer activity in EC cells by inhibiting the PI3K/Akt pathway.






Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Sardana RK, Chhikara N, Tanwar B, Panghal A. Dietary impact on esophageal cancer in humans: a review. Food Funct. 2018;9(4):1967–77.
Smyth EC, Lagergren J, Fitzgerald RC, et al. Oesophageal cancer. Nat Rev Dis Primers. 2017;3:17048.
Shah MA. Update on metastatic gastric and esophageal cancers. J Clin Oncol. 2015;33(16):1760–9.
Ohashi S, Miyamoto S, Kikuchi O, Goto T, Amanuma Y, Muto M. Recent advances from basic and clinical studies of esophageal squamous cell carcinoma. Gastroenterology. 2015;149(7):1700–15.
Bai Y, Lin H, Fang Z, et al. Plasma microRNA-19a as a potential biomarker for esophageal squamous cell carcinoma diagnosis and prognosis. Biomark Med. 2017;11(5):431–41.
Domper Arnal MJ, Ferrández Arenas Á, Lanas AÁ. Esophageal cancer: risk factors, screening and endoscopic treatment in Western and Eastern countries. World J Gastroenterol. 2015;21(26):7933–43.
Kang X, Chen K, Li Y, Li J, D’Amico TA, Chen X. Personalized targeted therapy for esophageal squamous cell carcinoma. World J Gastroenterol. 2015;21(25):7648–58.
Thakur A, Singla R, Jaitak V. Coumarins as anticancer agents: a review on synthetic strategies, mechanism of action and SAR studies. Eur J Med Chem. 2015;101:476–95.
Cha JY, Kim YT, Kim HS, Cho YS. Antihyperglycemic effect of stem bark powder from paper mulberry (Broussonetia kazinoki Sieb.) in type 2 diabetic Otsuka Long-Evans Tokushima fatty rats. J Med Food. 2008;11(3):499–505.
Bae UJ, Jang HY, Lim JM, Hua L, Ryu JH, Park BH. Polyphenols isolated from Broussonetia kazinoki prevent cytokine-induced β-cell damage and the development of type 1 diabetes. Exp Mol Med. 2015;47(4):e160.
Ryu JH, Ahn H, Jin LH. Inhibition of nitric oxide production on LPS-activated macrophages by kazinol B from Broussonetia kazinoki. Fitoterapia. 2003;74(4):350–4.
Kim HS, Lim J, Lee DY, Ryu JH, Lim JS. Kazinol C from Broussonetia kazinoki activates AMP-activated protein kinase to induce antitumorigenic effects in HT-29 colon cancer cells. Oncol Rep. 2015;33(1):223–9.
Kim JS, Kim JC, Shim SH, et al. Chemical constituents of the root of Dystaenia takeshimana and their anti-inflammatory activity. Arch Pharm Res. 2006;29(8):617–23.
Kim JH, Kim JK, Ahn EK, et al. Marmesin is a novel angiogenesis inhibitor: regulatory effect and molecular mechanism on endothelial cell fate and angiogenesis. Cancer Lett. 2015;369(2):323–30.
Chen IS, Chang CT, Sheen WS, et al. Coumarins and antiplatelet aggregation constituents from Formosan Peucedanum japonicum. Phytochemistry. 1996;41(2):525–30.
Dong L, Xu WW, Li H, Bi KH. In vitro and in vivo anticancer effects of marmesin in U937 human leukemia cells are mediated via mitochondrial-mediated apoptosis, cell cycle arrest, and inhibition of cancer cell migration. Oncol Rep. 2018;39(2):597–602.
Wang X, Shen Y, Wang S, et al. PharmMapper 2017 update: a web server for potential drug target identification with a comprehensive target pharmacophore database. Nucleic Acids Res. 2017;45(W1):W356–60.
Safran M, Chalifa-Caspi V, Shmueli O, et al. Human gene-centric databases at the Weizmann institute of science: GeneCards, UDB, CroW 21 and HORDE. Nucleic Acids Res. 2003;31(1):142–6.
Szklarczyk D, Gable AL, Nastou KC, et al. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021;49(D1):D605–12.
Pink RC, Bailey TA, Iputo JE, Sammon AM, Woodman AC, Carter DR. Molecular basis for maize as a risk factor for esophageal cancer in a South African population via a prostaglandin E2 positive feedback mechanism. Nutr Cancer. 2011;63(5):714–21.
Znati M, Ben Jannet H, Cazaux S, Souchard JP, Harzallah Skhiri F, Bouajila J. Antioxidant, 5-lipoxygenase inhibitory and cytotoxic activities of compounds isolated from the Ferula lutea flowers. Molecules. 2014;19(10):16959–75.
Kim JH, Kim MS, Lee BH, et al. Marmesin-mediated suppression of VEGF/VEGFR and integrin β1 expression: its implication in non-small cell lung cancer cell responses and tumor angiogenesis. Oncol Rep. 2017;37(1):91–7.
Li P, Chen J, Wang J, et al. Systems pharmacology strategies for drug discovery and combination with applications to cardiovascular diseases. J Ethnopharmacol. 2014;151(1):93–107.
Boezio B, Audouze K, Ducrot P, Taboureau O. Network-based approaches in pharmacology. Mol Inform. 2017;36(10):1700048.
Annovazzi L, Mellai M, Caldera V, Valente G, Tessitore L, Schiffer D. mTOR, S6 and AKT expression in relation to proliferation and apoptosis/autophagy in glioma. Anticancer Res. 2009;29(8):3087–94.
Chappell WH, Steelman LS, Long JM, et al. Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR inhibitors: rationale and importance to inhibiting these pathways in human health. Oncotarget. 2011;2(3):135–64.
McCubrey JA, Steelman LS, Chappell WH, et al. Advances in targeting signal transduction pathways. Oncotarget. 2012;3(12):1505–21.
Falasca M. PI3K/Akt signalling pathway specific inhibitors: a novel strategy to sensitize cancer cells to anti-cancer drugs. Curr Pharm Des. 2010;16(12):1410–6.
Li B, Li J, Xu WW, et al. Suppression of esophageal tumor growth and chemoresistance by directly targeting the PI3K/AKT pathway. Oncotarget. 2014;5(22):11576–87.
Yu H, Jiang X, Jiang L, et al. Protocadherin 8 (PCDH8) inhibits proliferation, migration, invasion, and angiogenesis in esophageal squamous cell carcinoma. Med Sci Monit. 2020;26:e920665.
Yokota T, Serizawa M, Hosokawa A, et al. PIK3CA mutation is a favorable prognostic factor in esophageal cancer: molecular profile by next-generation sequencing using surgically resected formalin-fixed, paraffin-embedded tissue. BMC Cancer. 2018;18(1):826.
Mori R, Ishiguro H, Kimura M, et al. PIK3CA mutation status in Japanese esophageal squamous cell carcinoma. J Surg Res. 2008;145(2):320–6.
Wei L, Xu Z. Cross-signaling among phosphinositide-3 kinase, mitogen-activated protein kinase and sonic hedgehog pathways exists in esophageal cancer. Int J Cancer. 2011;129(2):275–84.
Kwak AW, Lee MJ, Lee MH, Yoon G, Cho SS, Chae JI, Shim JH. The 3-deoxysappanchalcone induces ROS-mediated apoptosis and cell cycle arrest via JNK/p38 MAPKs signaling pathway in human esophageal cancer cells. Phytomedicine. 2021;86:153564.
Author information
Authors and Affiliations
Contributions
QW performed the experiments and wrote the manuscript. SZ performed the experiments and analyzed the data. HW analyzed the data and contributed to bioinformatics analysis. QW designed and supervised this study. All authors read the manuscript and approved it for publication.
Corresponding author
Ethics declarations
Ethical statement
Not applicable.
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Zhong, S., Wu, H. et al. In vitro anti-cancer effect of marmesin by suppression of PI3K/Akt pathway in esophagus cancer cells. Esophagus 19, 163–174 (2022). https://doi.org/10.1007/s10388-021-00872-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-021-00872-8