Abstract
Human–wildlife conflicts are a growing problem in Northern Hemisphere where wild ungulates are one of the taxonomic groups most frequently involved. To mitigate these conflicts, it is essential to develop preventive actions able to avoid encounters between wildlife and human (activities). We here employed photo-trapping to evaluate the behaviour of red deer (Cervus elaphus) when confronted with dissuasive portable deterrents that function on the basis of changing patterns of light and ultrasound. This was done by following a before/after experimental design, with two phases: (i) a test phase, with active deterrents, and (ii) a control phase, without deterrents. When deterrents were activated they achieved a 48.96% reduction in the frequency of use by red deer (up to 66.64% when it was assessed on a thinner Sect. 10 m wide from the line of deterrents) and produced a reduction of 67.71% in the frequency of deterrent-line crossings. However, a habituation effect was detected since the use by red deer of the treatment area increased as time since treatment. These results indicate that these portable devices are effective as regards dissuading deer, mainly on short time scales. The deterrents tested here could be suitable for use at focal points for short periods or in combination with other methods to improve their effectiveness in vulnerability points. This device could potentially be used to mitigate conflicts caused by wildlife species and in response to relevant and timely situations, such as vehicle collisions and damage to crops, among others.
Similar content being viewed by others
Introduction
Human-wildlife conflicts (hereafter, HWCs) occur whenever wildlife poses a direct threat to activities taken by humans (Conover 2001); in essence, they are conflicts between human interests and are currently widespread (Redpath et al. 2015). A number of global trends have contributed to HWCs, such as human population growth, land use transformation, species’ habitat loss, among others (see Distefano 2005; Venter et al. 2016). These trends generally force a greater overlap between wildlife and human activities, thus generating conflicts, including damage to crops and forests, traffic accidents and risk for pathogen transmission to livestock and even humans (Ruiz-Fons 2017; Gemeda and Meles 2018; Valente et al. 2020). Wild ungulates are one of the taxonomic groups most frequently involved in such conflicts owing to their widespread distribution, unprecedented population growth and high population densities in certain places (e.g. Linnell et al. 2020; Carpio et al. 2021).
Several preventive and mitigation measures can be used to minimize HWCs (e.g. Snyder and Rentsch 2020; König et al. 2020). In the case of wild ungulates, the most frequently used tools are aimed to reduce population size and/or constraint habitat use and include harvesting by recreational hunters, trapping, snaring and poisoning (Geisser and Reyer 2004; Christie et al. 2014; Fischer et al. 2016). However, the effectiveness of these lethal control methods is often controversial, and their social acceptance is an issue (Martínez-Jauregui et al. 2020). With regard to non-lethal methods, permanent exclusion fences are commonly used and are often the most effective approach by which to reduce HWCs involving ungulates (VerCauteren et al. 2006a, 2010; Lavelle et al. 2011; Negus et al. 2019). Fences for wildlife are permanent and costly measures, create a strong fragmentation effect and cannot, therefore, be implemented in all situations (VerCauteren et al. 2006a; Lindsey et al. 2012). Furthermore, certain vulnerable points may compromise their effectiveness, such as intersections with rivers, roads or steep terrain (see Mysterud and Rolandsen 2019; Honda 2019; Honda et al. 2020). Traditional non-lethal dissuasive methods include the guarding of fields (by farmers, managers or dogs) and the use of odour repellents (in order to reduce the desirability of resources) (Beringer et al. 1994; Thapa 2010; Monteith et al. 2019; Guerisoli and Pereira 2020). The main problem with odour repellents is, however, that some species rapidly become habituated to olfactory stimuli (Elmeros et al. 2011), whereas guarding fields may result, for instance, in farmers becoming exhausted (Hua et al. 2016). The most widely implemented non-lethal and portable measures are optical (e.g. scarecrows, lights, lasers, reflective devices) and sound deterrents (e.g. gas exploders, harassment shooting, sirens, ultrasonic and bioacoustics devices) (Koehler 1990; Gilsdorf et al. 2004; Brieger et al. 2017), although animals may eventually become habituated to them (Benten et al. 2019; Honda 2019). In this respect, several functionalities are incorporated in the devices to reduce the loss of efficacy owing to animal habituation; for instance, devices activated by an animal’s presence are more effective than permanent or routine displays (Gilsdorf et al. 2002).
Limitations on the use of certain systems include high costs and maintenance (VerCauteren et al. 2006a; Lindsey et al. 2012), the loss of effectiveness owing to habituation (Ujvári et al. 1998; Honda 2019), or low effectiveness as regards altering the animals’ behaviour (e.g. Elmeros et al. 2011; Benten et al. 2019). Measures can be combined in order to increase their efficacy (e.g. Nolte 1999; Honda 2019; Honda et al. 2020), because there is no single and 100% effective solution with which to protect from wildlife damage (in a broad sense) (Koehler 1990; Gilsdorf et al. 2002). There is consequently a need to identify adequate mitigation measures with which to mitigate HWCs (Redick and Jacobs 2020; Candaele et al. 2021). Here, we have assessed the effectiveness of a portable device that was developed by VISEVER S.L. company to deter wildlife with some improvements to reduce habituation (see below). In this context, the general objective of this study was to assess the behaviour of wildlife when confronted with a deterrent based on random light signals and ultrasound. The specific objectives were to (i) assess the effectiveness of the devices as regards preventing animals from using the treatment area and (ii) quantify and characterize the response of the animals to the deterrent activation.
Materials and methods
Study area
The field work was carried out on a perimeter fenced hunting estate called Los Quintos de Mora (6864 ha), located in the Montes de Toledo Mountain range (39° 26′ N, 4° 2′ W), in central Spain. It is a hunting estate, perimetrally fenced and managed by Organismo Autónomo de Parques Nacionales (https://www.miteco.gob.es/es/parques-nacionales-oapn/). The climate is continental Mediterranean, characterised by dry summers and a high inter-annual variability in rainfall. The study area is a hilly area dominated by Mediterranean vegetation. Three native wild ungulate species are present in our study area: red deer (Cervus elaphus, 34.87 ind·km−2 ± 7.37 (standard error, SE)), wild boar (Sus scrofa, 6.19 ind·km−2 ± 1.92 (SE)) and roe deer (Capreolus, 0.44 ind·km−2 ± 0.24 (SE)—males only); estimates were obtained from camera trap data and random encounter models (for further details, see Palencia et al. 2021; 2022). This estate is suitable as regards evaluating the effectiveness of the deterrents owing to the great probability of the number of encounters between wildlife and devices.
Deterrents
The devices tested in this study are portable posts designed to emit signals and deter wildlife (Supplementary Material Fig. S1). The deterrents are composed of two signals: ultrasound (from 20 to 60 kHz) and flashing LED lights (700 nm, 3 W). The problem of habituation was minimized by the following: (i) autonomous activation by passive infrared tigger (PIR) resulting from the proximity of wildlife, and (ii) the randomisation of the light signal and the incorporation of an ultrasound to dissuade fauna. The deterrents are activated and emit a signal in the presence of an animal within the field of detection; according to manufacturing manual, they detect animals in an area of 160° and 13–17 m of effective activation radius. These characteristics result that a 20 m separation between deterrents is sufficient to cover the location in which the animals can access the experimental area.
The experimental design
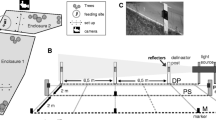
From November 2019 to February 2020, a before/after experimental design was followed. Two sections of 100 m (pseudo replicates) were selected in an area of wooded land on the hunting estate (see Fig. 1). These sections were separated by a 60 m wide vegetated stream, signifying that the animals that accessed one section could not directly see the signals from the deterrents in the other section. However, the proximity between sections does not allow to guarantee the total independence between sections since the same animal could potentially be exposed to both in consecutive days. This potential lack of independence between sections is expected to dilute the effectiveness of the deterrents and, therefore, the results reported here should be interpreted as a conservative estimation of the deterrents performance.
Scheme of the experimental design, in which the two monitored sections are shown along with their two zones; one zone would record the movements of the animals from the resting place to the feeding area (zones A and C, for Sects. 1 and 2, respectively), and the other the movements going back to the resting areas (zones B and D, for Sects. 1 and 2, respectively). Each zone was monitored with two lines of cameras: in the internal line cameras were located on the deterrents and in the external line cameras mounted on iron posts at 10 m from the internal line
Two zones were monitored in each section in order to verify the daily movements of the animals (see Boitani et al. 1994; Allen et al. 2014): their departure from the resting place (in this case to the north of the devices) to the feeding area (in this case to the south of the devices) and vice versa. In this respect, a zone is expected to record the movements of the animals when move to feeding area and the other when back to resting area. Two lines of cameras were placed in each zone. The first line (internal line hereafter) consisted of five cameras located on the deterrents (one camera per device). The second line (external line hereafter) consisted of four cameras mounted on iron posts 40–50 cm above the ground and 10 m from the aforementioned line (see Fig. 1).
The field work was carried out in two phases: (i) the test phase, in which the deterrents were activated, and (ii) the control phase, without deterrents. The experiment, test and control phases were alternated in both sections. In October 1, deterrents and cameras were installed in the field. Devices autonomy-related problems delayed the starting of the experiment 1 month. During this month, deterrents were installed in Sect. 1 but remained inactivates. From November 11 to December 9, deterrents were activated in Sect. 1, whereas from December 9 to January 29, they were activated in Sect. 2. Hunting was not carried out in the area during study period. The cameras were checked every 2 weeks in order to ensure their operability. The cameras were set up to be operative 24 h per day and shot a burst of four photos at each activation and without any delay between consecutive activations. Four Reconyx cameras (Hyperfire 2 Convert) were used on the internal lines, while four Browning cameras (Command Ops Pro) were employed on the external lines, and an additional one was used to complete the internal lines.
Statistical analysis
With regard to the information obtained with the camera traps, we considered that successive captures (individual or group of individuals) of the same species separated by more than ten minutes were independent events (Meek et al. 2014). At random points (i.e. far for any attracting resource) the probability to capture two different groups of animals in a single and narrow point (as that monitored with camera traps) in less of 10 min can be considered negligible (see also Jimenez et al. 2017). The capture rate, estimated as the number of events divided by the number of days that the camera was operative (camera-days), was estimated at the camera trap and week levels and is expressed as events·day−1.
A general linear mixed model (Gaussian distribution and logarithmic link function; Zuur et al. 2016) was parameterized to identify the factors explaining the variation in the capture rate (the response variable). The capture rate at weekly level was used to measure the frequency of use of a certain area. In this context, we expected the capture rate to decrease after the activation of the deterrents (test phase) as a consequence of the animals’ response. Date (number of weeks from 2019–1-1), time since starting each experimental phase (weeks; see the experimental design), treatment line (internal vs. external) and phase (control vs. test) were considered as fixed effect factors. Date and time since starting each experimental phase were correlated (Pearson R = 0.4), and they were included to specify the experimental design. The statistical interactions between time since starting and experimental phase and line and experimental phase were also tested. Date was considered to account for potential differences in the use of the area by the species along the experimental period. Time since treatment is aimed to assess the evolution of the species response along the experiment. Camera ID nested in zone were used as random effect factors. A backward stepwise procedure based on AIC was performed to exclude redundant terms (full model: Response ~ Date + Time + Phase + Line + Time:Phase + Line:Phase + (1|Zone:CamID). This model was first parameterized using all events and subsequently using only those recorded in the internal line; in that case excluding the terms involving the factor experimental line.
Another mixed model was parameterized to assess the factors affecting the crossing success of the deterrent line. The response variable was the number of events that produced crossings in relation to the total number of events that a camera recorded at weekly level; in this case, a generalized linear mixed model (binomial distribution and logit link function). A cross is defined when a capture produces events in different zones within the same section. Fixed factors, including interactions, random effects factors and stepwise procedure were the same previously described for the other model.
Analyses were carried out using the stats R package (R Core Team 2013). Mixed models were performed with lme4 R package (Bates et al. 2015). The r.squareGLMM function of the MuMIn R package (Barton 2022) was used to calculate conditional and marginal coefficient of determination for generalized mixed-effect models. A protocol for the exploration of data and residuals to assess normality and variance homogeneity was applied by following the recommendations of Zuur et al. (2010). The statistical significance of the p value was set at 0.05.
Results
The total monitoring effort consisted of 2827 camera-days. In the experimental phase, Sect. 1 had 488 camera-days in the control phase (without deterrents) and 918 camera-days in the test phase (with active deterrents). In Sect. 2, 918 camera-days were registered in the control phase and 503 in the test phase. We obtained 72,894 captures of animals during the experiment, 71,202 of which were of red deer. These were reduced to 6543 events by using a time lag of ten minutes between captures in order to define the events, and the vast majority were of red deer (6186). The analyses were then limited to the events involving red deer, since only this species provided sufficient information with which to evaluate the performance of the deterrents.
Effectiveness of the deterrents
The statistical parameters of the model carried out for assessing deterrents effectiveness and animals’ habituation are reported in Table 1. The marginal and conditional R2 were 0.37 and 0.51, respectively. Independently of the other factors, (i) the activation of the devices significantly reduced the capture rate by 48.96% and (ii) the capture rate was 41.87% lower on the internal than on the external line (Fig. 2). The model showed a positive and significant association between the trapping rate and the time since starting each experimental phase (Fig. 2). Given the notable effect of the camera line in the model, we parameterized another model just on data for the internal line (Table 2). In this case, the only significant predictor was the experimental phase and the marginal and conditional R2 were 0.35 and 0.48, respectively. Just on the internal line, the activation of the devices reduced the capture rate by 66.64% (Fig. 3). In this case, the time since starting each experimental phase was marginally related to capture rate.
Effectiveness of the deterrents in relation to significant factors, after controlling for camera trap nested in zone as random effect factors. Capture rate was used as the frequency of use of a certain area and was related to A experimental phase (control vs. test), B line of cameras (internal vs. external) and C time since starting each experimental phase
Crossing success of the deterrents’ lines
The statistical parameters of the model for assessing variability in the crossing success were reported in Table 3. The marginal and conditional R2 were 0.74 and 0.82, respectively. The crossing success was statistically related to the experimental phase (lower success during test than in control phase), and negatively associated to date and the time since starting each experimental phase (Fig. 4). The activation of the devices reduced the crossing success by 67.71%
Discussion
Our experiment showed that the activation of the deterrents tested here: (i) reduced the use of the treatment area by red deer by 48.96% (up to 66.64% when it was assessed on the internal lines); (ii) achieved a reduction of 67.71% in the number of deterrent-line crossings; and (iii) lost their deterrent effect at 6–7 weeks. The low-cost autonomous and portable deterrent device tested, therefore, dissuaded the red deer for a few weeks and has the potential to be used both as a single measure to protect target points and in combination with other invasive and/or non-invasive measures, such as intensive harvesting by recreational hunters or fences, among others.
Devices producing artificial light and sound are generally more promising as regards deterring crepuscular and nocturnal species than either alone (Koehler et al. 1990). Moreover, those activated by an animal’s presence are more effective than routine displays (Gilsdorf et al. 2002). The device tested in this study fulfils all these requirements and was moderately effective in the medium term, although the animals became habituated to it after 6–7 weeks.
Previous studies using a wide variety of frightening devices have reported a wide range of effectiveness (Beringer et al. 2003; Gilsdorf et al. 2004; Benten et al. 2018). However, greater effectiveness has been reported when physical barriers are complemented with other devices (Peterson et al. 2003; Allen et al. 2013; Honda 2019). For example, Honda et al. (2020) designed an ungulate-exclusion grate that was 98.5% effective in keeping deer from accessing the road, although its installation incurred high costs. We recorded a moderate reduction in the use of the treatment area with some variability between zones; a lower reduction was observed when the animals were approaching from the scrubland area (which typically occurred at sunset and dusk) than when arriving from the grassland (which typically occurred at night or sunrise when they were seeking shelter). This can be partially explained by the fact that deer approaching from the grassland alerted other individuals or groups as the deterrents triggered, since deer are social animals that learn spatial pattern of risk avoidance (Stankowich 2008; Coppes et al. 2017). In this respect, Jayakody et al. (2008) reported that the percentage of deer that were vigilant in grassland was higher than in scrubland. Another explanation could be a lower effect of the activation of the devices when the animals left the resting place (sunset) and there was still sunlight. When using a flashlight to deter deer, Honda (2019) activated this device only from 6:00 P.M. to 6:00 A.M. because it was expected to be effective only at night. Furthermore, the external line captured more animals than the internal one simply because it was further away from the negative stimulus of the device (see below).
After deterrents activation, the crossing success was significantly reduced. This evidence the effectiveness of the devices as regards deterring red deer from crosses through the line of deterrents and entering the grassland protected by them. In this study, the devices were located on wooded land with a large proportion of grassland, which is the main natural resource selected by deer (Allen et al. 2014). In this respect, the deterrents were effective as regards protecting the grassland surface (2000 m2), although this could be due to the exposure to the novel stimulus in the first days. More studies in this line are required to evaluate their effectiveness with regard to protecting a specifically demanded/limited resource.
Red deer tended to become habituated to the deterrents, signifying that their effectiveness in reducing the use of the treatment area by red deer decreased at 6–7 weeks after activation. However, the deterrent-line crossings decreased with time since activation. This is similar to that which has occurred previous studies, in which animals repeatedly exposed to a stimulus, without reward or punishment, became habituated to that stimulus (Manning 2012). The same pattern was previously reported by Beringer et al. (2003), who employed an animal-activated scarecrow that was effective for 6 weeks in summer when deer browsed soybean fields. Moreover, previous studies using lighting techniques found that they were effective only between a few days and a couple of weeks (Koehler et al. 1990; Belant et al. 1998), whereas lasers did not prevent deer from entering farm fields (VerCauteren et al. 2003; 2006b). With regard to sound devices, Ujvári et al. (1998) observed that fallow deer (Dama dama) became habituated from the second week, although Belant et al. (1996) had previously observed that gas exploders activated by deer movement were effective as regards reducing deer incursions into feeding stations for up to 6 weeks, while periodically firing exploders (every 8–10 min) were effective for only 2 days. Recently, Honda (2019) demonstrated the effectiveness of a sonic device as regards reducing the intrusions of deer into a fenced area, although for a period no longer than 1 month. Devices that are activated by animals with changing patterns of multiple stimuli (like the deterrents tested here) were more effective than permanent or routine displays (Nolte 2003). The familiarity of red deer with the devices, including randomised signals (Linhart et al. 1992), could be minimized by installing or operating them at specific moments, only when resources are most susceptible to damage (Nolte 1999), or in other circumstances when an increased number of passes are expected (e.g. during hunting activities). However, the high density of the red deer in the study area could be related to a higher level of exposure (passive) to the deterrents and faster habituations. The habituation patterns reported here could, therefore, differ substantially when working with populations/species at lower densities, since the degree of habituation depends on the repeated exposure of an individual to the stimulus. In this respect, the effects of the high density of the red deer on faster habituations requires further research. On the other hand, the experimental design is not able to assure total independence among sections and, therefore, results of the performance of the deterrents reported here should be interpreted as conservative estimations. Finally, the reduction in crossing success with time could be suggesting that animals finally avoid crossing the treatment area. This fact could be motivated by our experimental design since deers just moving 100 m right or left can avoid the exposure to deterrents. Further studies to disentangle this behaviour are still needed.
Applicability
From a practical point of view, deterrents tested here are portable effective automatic devices that do not require an external power source (Supplementary Material Fig. S1). The cost of the device in relation to other systems ($300) is also an advantage (Putman et al. 2004). For example, Gilsdorf et al. (2004) developed a deer-activated bioacustic frightening devices that cost about $600, whereas Peterson et al. (2003) invested $26,040 in each bridge-grate employed to reduce deer-collisions on urban highways. Deterrents tested here can be used: (i) to protect small perimeters (e.g. small crops, reforestations, orchards (Gilsdorf et al. 2004)), (ii) at vulnerable points where other methods require a high investment (e.g. intersection rivers and fences (Peterson et al. 2003; Honda 2020)), or (iii) to mitigate wildlife-vehicle collisions in accident black spots (Benten et al. 2018), among others. They could also be used together with other temporary portable devices as an integrated approach. Diversifying frightening stimuli by changing the placement and type of frightening devices increases their effectiveness (Nolte 1999; Koehler et al. 1990). However, it would be necessary to evaluate their effectiveness in other scenarios and for other species and ranges of population densities in order to attain a precise estimation of their practical value as regards reducing HWCs.
Change history
04 October 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10344-022-01619-1
References
Allen TD, Huijser MP, Willey DW (2013) Effectiveness of wildlife guards at access roads. Wildl Soc Bull 37:402–408. https://doi.org/10.1002/wsb.253
Allen AM, Månsson J, Jarnemo A, Bunnefeld N (2014) The impacts of landscape structure on the winter movements and habitat selection of female red deer. Eur J Wildl Res 60:411–421. https://doi.org/10.1007/s10344-014-0797-0
Barton K (2022) MuMIn: Multi-Model Inference. R package version 1.46.0. https://CRAN.R-project.org/package=MuMIn
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67: 1–48. https://doi.org/10.48550/arXiv.1406.5823
Belant JL, Seamans TW, Dwyer CP (1996) Evaluation of propane exploders as white-tailed deer deterrents. Crop Prot 15:575–578. https://doi.org/10.1016/0261-2194(96)00027-0
Belant JL, Seamans TW, Tyson LA (1998) Evaluation of electronic frightening devices as white-tailed deer deterrents. In ‘Proceedings of the 18th Vertebrate Pest Conference’. (Eds. R. O. Baker, and A. C. Crabb) pp.107–110. (University of California: Davis, USA).
Benten A, Annighöfer P, Vor T (2018) Wildlife warning reflectors’ potential to mitigate wildlife-vehicle collisions—a review on the evaluation methods. Front Ecol Evol 6:37. https://doi.org/10.3389/fevo.2018.00037
Benten A, Balkenhol N, Vor T, Ammer C (2019) Wildlife warning reflectors do not alter the behavior of ungulates to reduce the risk of wildlife-vehicle collisions. Eur J Wildl Res 65:1–12. https://doi.org/10.1007/s10344-019-1312-4
Beringer J, Hansen LP, Heinen RA, Giessman NF (1994) Use of dogs to reduce damage by deer to a white pine plantation. Wildl Soc Bull 22:627–632
Beringer J, VerCauteren KC, Millspaugh JJ (2003) Evaluation of an animal-activated scarecrow and a monofilament fence for reducing deer use of soybean fields. Wildl Soc Bull 31:492–498
Boitani L, Mattei L, Nonis D, Corsi F (1994) Spatial and activity patterns of wild boars in Tuscany, Italy. J Mammal 75:600–612. https://doi.org/10.2307/1382507
Brieger F, Hagen R, Kröschel M, Hartig F, Petersen I, Ortmann S, Suchant R (2017) Do roe deer react to wildlife warning reflectors? A test combining a controlled experiment with field observations. Eur J Wildl Res 63:1–11. https://doi.org/10.1007/s10344-017-1130-5
Candaele R, Lejeune P, Licoppe A, Malengreaux C, Brostaux Y, Morelle K, Latte N (2021) Mitigation of bark stripping on spruce: the need for red deer population control. Eur J for Res 140:227–240. https://doi.org/10.1007/s10342-020-01326-z
Carpio AJ, Apollonio M, Acevedo P (2021) Wild ungulate overabundance in Europe: Contexts, causes, monitoring and management recommendations. Mammal Rev 51:95–108. https://doi.org/10.1111/mam.12221
Christie J, DeMarco E, Hiroyasu E, Kreger A, Ludington M (2014) Wild pig management at Tejon Ranch. Bren School Group Project.
Conover MR (2001) Resolving human-wildlife conflicts: the science of wildlife damage management. CRC Press, Boca Raton, USA
Coppes J, Burghardt F, Hagen R, Suchant R, Braunisch V (2017) Human recreation affects spatio-temporal habitat use patterns in red deer (Cervus elaphus). PLoS ONE 12:e0175134. https://doi.org/10.1371/journal.pone.0175134
Distefano E (2001) Human-Wildlife Conflict worldwide: collection of case studies, analysis of management strategies and good practices. Food and Agricultural Organization of the United Nations (FAO), Sustainable Agriculture and Rural Development Initiative (SARDI), Rome, Italy.
Elmeros M, Winbladh JK, Andersen PN, Madsen AB, Christensen JT (2011) Effectiveness of odour repellents on red deer (Cervus elaphus) and roe deer (Capreolus capreolus): a field test. Eur J Wildl Res 57:1223–1226. https://doi.org/10.1007/s10344-011-0517-y
Fischer JW, McMurtry D, Blass CR, Walter WD, Beringer J, VerCauteren KC (2016) Effects of simulated removal activities on movements and space use of feral swine. Eur J Wildl Res 62:285–292. https://doi.org/10.1007/s10344-016-1000-6
Geisser H, Reyer HU (2004) Efficacy of hunting, feeding, and fencing to reduce crop damage by wild boars. J Wildl Manag 68:939–946. https://doi.org/10.2193/0022-541X(2004)068[0939:EOHFAF]2.0.CO;2
Gemeda DO, Meles SK (2018) Impacts of human-wildlife conflict in developing countries. JASEM 22:1233–1238. https://doi.org/10.4314/jasem.v22i8.14
Gilsdorf JM, Hygnstrom SE, VerCauteren KC (2002) Use of frightening devices in wildlife damage management. Integr Pest Manag Rev 7:29–45. https://doi.org/10.1023/A:1025760032566
Gilsdorf JM, Hygnstrom SE, VerCauteren KC, Clements GM, Blankenship EE, Engeman RM (2004) Evaluation of a deer-activated bio-acoustic frightening device for reducing deer damage in cornfields. Wildl Soc Bull 32:515–523. https://doi.org/10.2193/0091-7648(2004)32[515:EOADBF]2.0.CO;2
Guerisoli MM, Pereira JA (2020) Deer damage: a review of repellents to reduce impacts worldwide. J Environ Manage 271:110977. https://doi.org/10.1016/j.jenvman.2020.110977
Honda T (2019) A sound deterrent prevented deer intrusions at the intersection of a river and fence. Mammal Study 44:269–274. https://doi.org/10.3106/ms2019-0034
Honda T, Kubota Y, Ishizawa Y (2020) Ungulates-exclusion grates as an adjoining facility to crop damage prevention fences. Eur J Wildl Res 66:1–7. https://doi.org/10.1007/s10344-020-1362-7
Hua X, Yan J, Li H, He W, Li X (2016) Wildlife damage and cultivated land abandonment: Findings from the mountainous areas of Chongqing, China. Crop Prot 84:141–149. https://doi.org/10.1016/j.cropro.2016.03.005
Jayakody S, Sibbald AM, Gordon IJ, Lambin X (2008) Red deer Cervus elephus vigilance behaviour differs with habitat and type of human disturbance. Wildlife Biol 14:81–91. https://doi.org/10.2981/0909-6396(2008)14[81:RDCEVB]2.0.CO;2
Jiménez J, Higuero R, Charre Medellín JF, Acevedo P (2017) Spatial mark-resight models to estimate feral pig population density. Hystrix 28: 208–213. https://doi.org/10.4404/hystrix-28.2-12141
Koehler AE, Marsh RE, Salmon TP (1990) Frightening methods and devices/stimuli to prevent mammal damage. A review. In ‘Proceedings of the 14th Vertebrate Pest Conference’. (Eds. L. R. Davis, and R. E. Marsh) pp. 168–173. (University of California: Davis, USA).
König HJ, Kiffner C, Kramer-Schadt S, Fürst C, Keuling O, Ford AT (2020) Human–wildlife coexistence in a changing world. Conserv Biol 34:786–794. https://doi.org/10.1111/cobi.13513
Lavelle MJ, VerCauteren KC, Hefley TJ, Phillips GE, Hygnstrom SE, Long DB, Fischer JW, Swafford SR, Campbell TA (2011) Evaluation of fences for containing feral swine under simulated depopulation conditions. J Wildl Manag 75:1200–1208. https://doi.org/10.1002/jwmg.134
Lindsey PA, Masterson CL, Beck AL, Romañach S (2012) Ecological, social and financial issues related to fencing as a conservation tool in Africa. In: Somers MJ, Hayward MH (eds) Fencing for conservation. Springer, New York, USA pp 215–234
Linhart SB, Dasch GJ, Johnson RR, Roberts JD (1992) Electronic frightening devices for reducing coyote predation on domestic sheep: efficacy under range conditions and operational use. In ‘Proceedings of the 15th Vertebrate Pest Conference’. (Eds. J. E. Borrecco, and R. E. Marsh) pp. 386–392. (University of California: Davis, USA)
Linnell JD, Cretois B, Nilsen EB, Rolandsen CM, Solberg EJ, Veiberg V, Kaczensky P, Van Moorter B, Panzacchi M, Rauset GR, Kaltenborn B (2020) The challenges and opportunities of coexisting with wild ungulates in the human-dominated landscapes of Europe’s Anthropocene. Biol Conserv 244:108500. https://doi.org/10.1016/j.biocon.2020.108500
Manning A, Dawkins MS (2012) An introduction to animal behaviour. Cambridge University Press, New York, USA
Martínez-Jauregui M, Delibes-Mateos M, Arroyo B, Soliño M (2020) Addressing social attitudes toward lethal control of wildlife in national parks. Conserv Biol 34:868–878. https://doi.org/10.1111/cobi.13468
Meek PD, Ballard G, Claridge A, Kays R, Moseby K, O’Brien T, O’Connell A, Sanderson J, Swann DE, Tobler M, Townsend S (2014) Recommended guiding principles for reporting on camera trapping research. Biodivers Conserv 23:2321–2343. https://doi.org/10.1007/s10531-014-0712-8
Monteith KB, Monteith KL, Jenks JA (2019) Condensed tannins as a deterrent to crop depredation by white-tailed deer: Effects of concentration and learning. Wildl Soc Bull 43:693–700. https://doi.org/10.1002/wsb.1028
Mysterud A, Rolandsen CM (2019) Fencing for wildlife disease control. J Appl Ecol 56:519–525. https://doi.org/10.1111/1365-2664.13301
Negus PM, Marshall JC, Clifford SE, Blessing JJ, Steward AL (2019) No sitting on the fence: protecting wetlands from feral pig damage by exclusion fences requires effective fence maintenance. Wetl Ecol Manag 27:581–585. https://doi.org/10.1007/s11273-019-09670-7
Nolte DL (1999) Behavioral approaches for limiting depredation by wild ungulates. Grazing behavior of livestock and wildlife 60–69
Nolte DL (2003) Managing ungulates to protect trees. USDA National Wildlife Research Center-Staff Publications 261
Palencia P, Rowcliffe JM, Vicente J, Acevedo P (2021) Assessing the camera trap methodologies used to estimate density of unmarked populations. J Appl Ecol 58:1583–1592. https://doi.org/10.1111/1365-2664.13913
Palencia P, Barroso P, Vicente J, Hofmeester TR, Ferreres J, Acevedo P (2022) Random encounter model is a reliable method for estimating population density of multiple species using camera traps. Remote Sensing in Ecology and Conservation. https://doi.org/10.1002/rse2.269
Peterson MN, Lopez RR, Silvy NJ, Owen CB, Frank PA, Braden AW (2003) Evaluation of deer-exclusion grates in urban areas. Wildl Soc Bull 31:1198–1204
Putman R, Langbein J, Staines BW (eds) (2004) Deer and Road Traffic Accidents, a Review of Mitigation Measures: Costs and Cost-effectiveness. Scottish Natural Heritage, Scotland
R Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Redick CH, Jacobs DF (2020) Mitigation of Deer Herbivory in Temperate Hardwood Forest Regeneration: A Meta-Analysis of Research Literature. Forests 11:1220. https://doi.org/10.3390/f11111220
Redpath SM, Bhatia S, Young J (2015) Tilting at wildlife: reconsidering human–wildlife conflict. Oryx 49:222–225. https://doi.org/10.1017/S0030605314000799
Ruiz-Fons F (2017) A review of the current status of relevant zoonotic pathogens in wild swine (Sus scrofa) populations: changes modulating the risk of transmission to humans. Transbound Emerg Dis 64:68–88. https://doi.org/10.1111/tbed.12369
Snyder KD, Rentsch D (2020) Rethinking assessment of success of mitigation strategies for elephant-induced crop damage. Conserv Biol 34:829–842. https://doi.org/10.1111/cobi.13433
Stankowich T (2008) Ungulate flight responses to human disturbance: a review and meta-analysis. Biol Conserv 141:2159–2173. https://doi.org/10.1016/j.biocon.2008.06.026
Thapa S (2010) Effectiveness of crop protection methods against wildlife damage: a case study of two villages at Bardia National Park. Nepal Crop Prot 29:1297–1304. https://doi.org/10.1016/j.cropro.2010.06.015
Ujvári M, Baagøe HJ, Madsen AB (1998) Effectiveness of wildlife warning reflectors in reducing deer-vehicle collisions: a behavioral study. J Wildl Manag 62:1094–1099. https://doi.org/10.2307/3802562
Valente AM, Acevedo P, Figueiredo AM, Fonseca C, Torres RT (2020) Overabundant wild ungulate populations in Europe: management with consideration of socio-ecological consequences. Mammal Rev 50:353–366. https://doi.org/10.1111/mam.12202
Venter O, Sanderson EW, Magrach A, Allan JR, Beher J, Jones KR, Possingham HP, Laurence WF, Wood P, Fekete BM, Levy MA, Watson JEM (2016) Sixteen years of change in the global terrestrial human footprint and implications for biodiversity conservation. Nat Commun 7:1–11. https://doi.org/10.1038/ncomms12558
VerCauteren KC, Hygnstrom SE, Pipas MJ, Fioranelli PB, Werner SJ, Blackwell BF (2003) Red lasers are ineffective for dispersing deer at night. Wildl Soc Bull 31:247–252
VerCauteren KC, Lavelle MJ, Hygnstrom S (2006a) From the field: fences and deer-damage management: a review of designs and efficacy. Wildl Soc Bull 34:191–200. https://doi.org/10.2193/0091-7648(2006)34[191:FADMAR]2.0.CO;2
VerCauteren KC, Gilsdorf JM, Hygnstrom SE, Fioranelli PB, Wilson JA, Barras S (2006b) Green and blue lasers are ineffective for dispersing deer at night. Wildl Soc Bull 34:371–374. https://doi.org/10.2193/0091-7648(2006)34[371:GABLAI]2.0.CO;2
VerCauteren KC, Vandeelen TR, Lavelle MJ, Hall WH (2010) Assessment of abilities of white-tailed deer to jump fences. J Wildl Manag 74:1378–1381. https://doi.org/10.1111/j.1937-2817.2010.tb01260.x
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Zuur AF, Ieno EN (2016) A protocol for conducting and presenting results of regression-type analyses. Methods Ecol Evol 7:636–645. https://doi.org/10.1111/2041-210X.12577
Acknowledgements
The authors would like to thank Sally Newton for reviewing the English. They are also very grateful to Los Quintos de Mora estate for allowing them to carry out the field work; many thanks to Carlos Rodríguez, Ángel Moreno, Amanda García del Rincón, and the other workers for providing their support and facilities.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This research was funded by VISEVER S.L. E. Laguna is supported by a predoctoral grant (Programa Talento Formación) funded by the European Social Fund (ESF) and the Castilla-La Mancha regional government (JCCM) (ref: SBPLY/16/180501/000269). P. Palencia is supported by MINECO (FPU16/00039). AJ. Carpio is supported by a ‘Juan de la Cierva’ contract (FJCI-2017–33114) from MINECO-UCLM. C. Herraiz is supported by ESF and JCCM (ref: SUPLY/19/180501/000487).
Author information
Authors and Affiliations
Contributions
Conceptualization, P. Acevedo, and P. Palencia; field work and data curation, P. Palencia, C. Herraiz and J. Mateos-Aparicio; statistical analyses, P. Acevedo; C. Notario funding; writing—original draft preparation, E. Laguna, AJ. Carpio, and P. Acevedo; writing—review and editing, all authors. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Monitoring procedures were conducted according to the specifications of the protocol approved by the Animal Experiment Committee of Castilla-La Mancha University and by the Spanish Ethics Committee (PR-2016-10-16). This protocol was designed by specifically trained and certified scientists (B and C animal experimentation categories) according to EC Directive 86/609/EEC for animal handling and experiments.
Conflict of interest
The authors declare that they have conflict of interest. Carlos Notario, CEO of VISAVER, not contributed to the experimental design nor data analyses and interpretation. The authors have no further conflicts of interest to declare.
Data availability
The data that support this study will be shared upon reasonable request to the corresponding author.
Consent for publication
All parties involved consent to the publication of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: This article was originally published without an ethics approval.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laguna, E., Palencia, P., Carpio, A.J. et al. Evaluation of a combined and portable light-ultrasound device with which to deter red deer. Eur J Wildl Res 68, 50 (2022). https://doi.org/10.1007/s10344-022-01599-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-022-01599-2