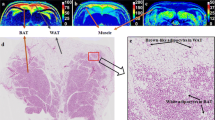
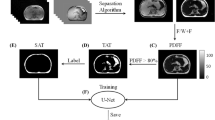
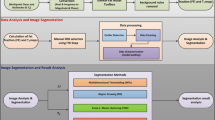
Abstract
Objective
The study aims to propose an accurate labelling method of interscapular BAT (iBAT) in rats using dynamic MR fat fraction (FF) images with noradrenaline (NE) stimulation and then develop an automatic iBAT segmentation method using a U-Net model.
Materials and methods
Thirty-four rats fed different diets or housed at different temperatures underwent successive MR scans before and after NE injection. The iBAT were labelled automatically by identifying the regions with obvious FF change in response to the NE stimulation. Further, these FF images along with the recognized iBAT mask images were used to develop a deep learning network to accomplish the robust segmentation of iBAT in various rat models, even without NE stimulation. The trained model was then validated in rats fed with high-fat diet (HFD) in comparison with normal diet (ND).
Result
A total of 6510 FF images were collected using a clinical 3.0 T MR scanner. The dice similarity coefficient (DSC) between the automatic and manual labelled results was 0.895 ± 0.022. For the network training, the DSC, precision rate, and recall rate were found to be 0.897 ± 0.061, 0.901 ± 0.068 and 0.899 ± 0.086, respectively. The volumes and FF values of iBAT in HFD rats were higher than ND rats, while the FF decrease was larger in ND rats after NE injection.
Conclusion
An automatic iBAT segmentation method for rats was successfully developed using the dynamic labelled FF images of activated BAT and deep learning network.








Similar content being viewed by others
Availability of data and materials
The datasets used in the current study are available from the corresponding author on reasonable request.
Abbreviations
- iBAT:
-
Interscapular brown adipose tissue
- WAT:
-
White adipose tissue
- FF:
-
Fat fraction
- NE:
-
Noradrenaline
- HFD:
-
High-fat diet
- ND:
-
Normal diet
- SD:
-
Sprague Dawley
References
van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, Bouvy ND, Schrauwen P, Teule GJ (2009) Cold-activated brown adipose tissue in healthy men. N Engl J Med 360(15):1500–1508
Virtanen KA, Lidell ME, Orava J, Heglind M, Westergren R, Niemi T, Taittonen M, Laine J, Savisto N-J, Enerbäck S (2009) Functional brown adipose tissue in healthy adults. N Engl J Med 360(15):1518–1525
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng Y-H, Doria A (2009) Identification and importance of brown adipose tissue in adult humans. N Engl J Med 360(15):1509–1517
Crane JD, Palanivel R, Mottillo EP, Bujak AL, Wang H, Ford RJ, Collins A, Blümer RM, Fullerton MD, Yabut JM (2015) Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat Med 21(2):166–172
Giralt M, Villarroya F (2013) White, brown, beige/brite: different adipose cells for different functions? Endocrinology 154(9):2992–3000
Labbé SM, Caron A, Bakan I, Laplante M, Carpentier AC, Lecomte R, Richard D (2015) In vivo measurement of energy substrate contribution to cold-induced brown adipose tissue thermogenesis. FASEB J 29(5):2046–2058
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, Rasbach KA, Boström EA, Choi JH, Long JZ (2012) A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 481(7382):463–468
Mirbolooki MR, Schade KN, Constantinescu CC, Pan ML, Mukherjee J (2015) Enhancement of 18F-fluorodeoxyglucose metabolism in rat brain frontal cortex using a β3 adrenoceptor agonist. Synapse 69(2):96–98
Sampath SC, Sampath SC, Bredella MA, Cypess AM, Torriani M (2016) Imaging of brown adipose tissue: state of the art. Radiology 280(1):4
Wu M, Junker D, Branca RT, Karampinos DC (2020) Magnetic resonance imaging techniques for brown adipose tissue detection. Front Endocrinol 11:421
Hu HH, Smith DL Jr, Nayak KS, Goran MI, Nagy TR (2010) Identification of brown adipose tissue in mice with fat–water IDEAL-MRI. J Magn Reson Imag 31(5):1195–1202
Bhanu Prakash K, Verma SK, Yaligar J, Goggi J, Gopalan V, Lee SS, Tian X, Sugii S, Leow MKS, Bhakoo K (2016) Segmentation and characterization of interscapular brown adipose tissue in rats by multi-parametric magnetic resonance imaging. Magn Reson Mater Phys Biol Med 29(2):277–286
Bhanu Prakash K, Srour H, Velan SS, Chuang K-H (2016) A method for the automatic segmentation of brown adipose tissue. Magn Reson Mater Phys Biol Med 29(2):287–299
Rangel-Azevedo C, Santana-Oliveira DA, Miranda CS, Martins FF, Mandarim-de-Lacerda CA, Souza-Mello V (2022) Progressive brown adipocyte dysfunction: Whitening and impaired nonshivering thermogenesis as long-term obesity complications. J Nutr Biochem 105:109002
Shimizu I, Walsh K (2015) The whitening of brown fat and its implications for weight management in obesity. Curr Obes Rep 4(2):224–229
Cheng C, Zou C, Liang C, Liu X, Zheng H (2017) Fat-water separation using a region-growing algorithm with self-feeding phasor estimation. Magn Reson Med 77(6):2390–2401
Zou C, Cheng C, Qiao Y, Wan Q, Tie C, Pan M, Liang D, Zheng H, Liu X (2019) Hierarchical iterative linear-fitting algorithm (HILA) for phase correction in fat quantification by bipolar multi-echo sequence. Quant Imag Med Surg 9(2):247
Vercauteren T, Pennec X, Perchant A, Ayache N (2009) Diffeomorphic demons: Efficient non-parametric image registration. Neuroimage 45(1):S61–S72
Tan M, Le Q (2019) Efficientnet: rethinking model scaling for convolutional neural networks. PMLR pp. 6105–6114
Aurelio YS, de Almeida GM, de Castro CL, Braga AP (2019) Learning from imbalanced data sets with weighted cross-entropy function. Neural Process Lett 50(2):1937–1949
Maswood MMS, Hussain T, Khan MB, Islam MT, Alharbi AG (2020) Cnn based detection of the severity of diabetic retinopathy from the fundus photography using efficientnet-b5. IEEE. p. 0147–0150
Trinh M-N, Nguyen V-D, Pham V-T, Tran T-T (2023) An EffcientNet-encoder U-Net Joint Residual Refinement Module with Tversky-Kahneman Baroni–Urbani–Buser loss for biomedical image Segmentation. Biomed Signal Process Control 83:104631
Wu B, Cheng C, Qi Y, Zhou H, Peng H, Wan Q, Liu X, Zheng H, Zhang H, Zou C (2022) Automatic segmentation of human supraclavicular adipose tissue using high-resolution T2-weighted magnetic resonance imaging. Magn Reson Mater Phys Biol Med 36:1–9
Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev. https://doi.org/10.1152/physrev.00015.2003
Chen KY, Cypess AM, Laughlin MR, Haft CR, Hu HH, Bredella MA, Enerbäck S, Kinahan PE, van Marken Lichtenbelt W, Lin FI (2016) Brown adipose reporting criteria in imaging Studies (BARCIST 1.0): recommendations for standardized FDG-PET/CT experiments in humans. Cell Metabol 24(2):210–222
Carpentier AC, Blondin DP, Virtanen KA, Richard D, Haman F, Turcotte ÉE (2018) Brown adipose tissue energy metabolism in humans. Front Endocrinol 9:447
Chen YI, Cypess AM, Sass CA, Brownell AL, Jokivarsi KT, Kahn CR, Kwong KK (2012) Anatomical and functional assessment of brown adipose tissue by magnetic resonance imaging. Obesity 20(7):1519–1526
Huo M, Ye J, Dong Z, Cai H, Wang M, Yin G, Qian L, Li Z-P, Zhong B, Feng S-T (2022) Quantification of brown adipose tissue in vivo using synthetic magnetic resonance imaging: an experimental study with mice model. Quant Imag Med Surg 12(1):526
Branca RT, McCallister A, Yuan H, Aghajanian A, Faber JE, Weimer N, Buchanan R, Floyd CS, Antonacci M, Zhang L (2018) Accurate quantification of brown adipose tissue mass by xenon-enhanced computed tomography. Proc Natl Acad Sci 115(1):174–179
Clerte M, Baron DM, Brouckaert P, Ernande L, Raher MJ, Flynn AW, Picard MH, Bloch KD, Buys ES, Scherrer-Crosbie M (2013) Brown adipose tissue blood flow and mass in obesity: a contrast ultrasound study in mice. J Am Soc Echocardiogr 26(12):1465–1473
Schade KN, Baranwal A, Liang C, Mirbolooki MR, Mukherjee J (2015) Preliminary evaluation of β3-adrenoceptor agonist-induced 18F-FDG metabolic activity of brown adipose tissue in obese Zucker rat. Nucl Med Biol 42(8):691–694
Sakamoto T, Nitta T, Maruno K, Yeh Y-S, Kuwata H, Tomita K, Goto T, Takahashi N, Kawada T (2016) Macrophage infiltration into obese adipose tissues suppresses the induction of UCP1 level in mice. Am J Physiol Endocrinol Metabol 310(8):E676–E687
Fernández Vázquez G, Reiter RJ, Agil A (2018) Melatonin increases brown adipose tissue mass and function in Zücker diabetic fatty rats: implications for obesity control. J Pineal Res 64(4):e12472
de Jong JM, Sun W, Pires ND, Frontini A, Balaz M, Jespersen NZ, Feizi A, Petrovic K, Fischer AW, Bokhari MH (2019) Human brown adipose tissue is phenocopied by classical brown adipose tissue in physiologically humanized mice. Nat Metab 1(8):830–843
Karampinos DC, Weidlich D, Wu M, Hu HH, Franz D (2019) Techniques and applications of magnetic resonance imaging for studying brown adipose tissue morphometry and function. Brown Adipose Tissue. https://doi.org/10.1007/164_2018_158
Sardjoe Mishre AS, Martinez-Tellez B, Straat ME, Boon MR, Dzyubachyk O, Webb AG, Rensen PC, Kan HE (2023) Image registration and mutual thresholding enable low interimage variability across dynamic MRI measurements of supraclavicular brown adipose tissue during mild cold exposure. Magn Reson Med 90(4):1316–1327
Cannon B, de Jong JM, Fischer AW, Nedergaard J, Petrovic N (2020) Human brown adipose tissue: classical brown rather than brite/beige? Exp Physiol 105(8):1191–1200
Funding
This research was supported by the National key research and development program of China: 2023YFC2410504, Natural Science Foundation of Guangdong Province: 2022A1515010162, Key Laboratory for Magnetic Resonance and Multimodality Imaging of Guangdong Province: 2023B1212060052, Scientific Instrument Innovation Team of the Chinese Academy of Sciences: GJJSTD20180002.
Author information
Authors and Affiliations
Contributions
CC: conception, design, provision of study materials and writing. BW: provision of study materials, data acquisition and writing. LZ: data analysis and interpretation, and writing—editing. QW: data acquisition and writing—editing. HP: data acquisition and writing—editing. XL: data analysis and interpretation, and writing—editing. HZ: administrative support and writing—editing. HZ: data analysis and interpretation, and writing-editing. CZ: conception, design and manuscript writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
The animals study received care in accordance with the Guidance Suggestions for the Care and Use of Laboratory Animals and were used under protocols approved by Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences Animal Care and Use Committee. (IRB approval number: SIAT-IACUC-190218-YGS-CCL-A0624).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, C., Wu, B., Zhang, L. et al. Automatic segmentation of the interscapular brown adipose tissue in rats based on deep learning using the dynamic magnetic resonance fat fraction images. Magn Reson Mater Phy 37, 215–226 (2024). https://doi.org/10.1007/s10334-023-01133-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-023-01133-8