Abstract
Objective
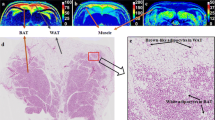
Brown adipose tissue (BAT) plays a key role for thermogenesis in mammals and infants. Recent confirmation of BAT presence in adult humans has aroused great interest for its potential to initiate weight-loss and normalize metabolic disorders in diabetes and obesity. Reliable detection and differentiation of BAT from the surrounding white adipose tissue (WAT) and muscle is critical for assessment/quantification of BAT volume. This study evaluates magnetic resonance (MR) acquisition for BAT and the efficacy of different automated methods for MR features-based BAT segmentation to identify the best suitable method.
Materials and methods
Multi-point Dixon and multi-echo T2 spin-echo images were acquired from 12 mice using an Agilent 9.4T scanner. Four segmentation methods: multidimensional thresholding (MTh); region-growing (RG); fuzzy c-means (FCM) and neural-network (NNet) were evaluated for the interscapular region and validated against manually defined BAT, WAT and muscle.
Results
Statistical analysis of BAT segmentation yielded a median Dice-Statistical-Index, and sensitivity of 89. 92 % for NNet, 82. 86 % for FCM, 72. 74 % for RG, and 72. 70 %, for MTh, respectively.
Conclusion
This study demonstrates that NNet improves the specificity to BAT from surrounding tissue based on 3-point Dixon and T2 MRI. This method facilitates quantification and longitudinal measurement of BAT in preclinical-models and human subjects.








Similar content being viewed by others

References
Cinti S (2011) Between brown and white: novel aspects of adipocyte differentiation. Ann Med 43(2):104–115
Nedergaard J, Bengtsson T, Cannon B (2007) Unexpected evidence for active brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab 293:E444–E452
Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev 84:277–359
Lichtenbelt WDM, Vanhommerig JW, Smulders NM, Drossaerts JMAFL, Kemerink GJ et al (2009) Cold-activated brown adipose tissue in healthy men. N Engl J Med 2009(360):1500–1508
Cypess A, Lehman S, Williams G, Tal I, Rodman D et al (2009) Identification and importance of brown adipose tissue in adult humans. N Engl J Med 360(15):1509–1517
Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T et al (2009) High incidence of metabolically active brown adipose tissue in healthy adult humans: effects of cold exposure and adiposity. Diabetes 58(7):1526–1531
Van der Lans A, Wierts R, Vosselman M, Schrauwen P, Brans B et al (2014) Cold-activated brown adipose tissue in human adults: methodological issues. Am J Physiol Regul Integr Comp Physiol 307(2):R103–R113
Lee P, Greenfield J, Ho K, Fulham M (2010) A critical appraisal of the prevalence and metabolic significance of brown adipose tissue in adult humans. Am J Physiol Endocrinol Metab 299(4):E601–E606
Ouellet V, Labbé S, Blondin D, Phoenix S, Guérin B et al (2012) Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J Clin Investig 122(2):545–552
Brix G, Lechel U, Glatting G, Ziegler S, Münzing W et al (2005) Radiation exposure of patients undergoing whole-body dual-modality 18F-FDG PET/CT examinations. J Nucl Med 46(4):608–613
Huang B, Law M, Khong P (2009) Whole-body PET/CT scanning: estimation of radiation dose and cancer risk. Radiology 251(1):166–174
Henkelman R (1992) New imaging technologies: prospects for target definition. Int J Radiat Oncol Biol Phys 22(2):251–257
Hu H, Nayak K (2010) Change in the proton T(1) of fat and water in mixture. Magn Reson Med 63(2):494
Strobel K, van den Hoff J, Pietzsch J (2008) Localized proton magnetic resonance spectroscopy of lipids in adipose tissue at high spatial resolution in mice in vivo. J Lipid Res 49(2):473–480
Zingaretti M, Crosta F, Vitali A, Guerrieri M, Frontini A et al (2009) The presence of UCP1 demonstrates that metabolically active adipose tissue in the neck of adult humans truly represents brown adipose tissue. FASEB J 23(9):3113–3120
Hu H, Börnert P, Hernando D, Kellman P, Ma J et al (2012) ISMRM workshop on fat–water separation: insights, applications and progress in MRI. Magn Reson Med 68(2):378–388
Peng X, Ju S, Fang F, Wang Y, Fang K et al (2013) Comparison of brown and white adipose tissue fat fractions in ob, seipin, and Fsp27 gene knockout mice by chemical shift-selective imaging and 1H-MR spectroscopy. Am J Physiol Endocrinol Metab 304(2):E160–E167
Hu H, Perkins TG, Chia JM, Gilsanz V (2013) Characterization of human brown adipose tissue by chemical-shift water–fat MRI. AJR Am J Roentgenol 200(1):177–183
Reeder S, Sirlin C (2010) Quantification of liver fat with magnetic resonance imaging. Magn Reson Imaging Clin N Am 18(3):337–357
Lunati E, Marzola P, Nicolato E, Fedrigo M, Villa M, Sbarbati A (1999) In vivo quantitative lipidic map of brown adipose tissue by chemical shift imaging at 4.7 Tesla. J Lipid Res 40(8):1395–1400
Branca R, Warren W (2011) In vivo brown adipose tissue detection and characterization using water–lipid intermolecular zero quantum coherences. Magn Reson Med 65(2):313–319
Borga M, Virtanen K, Romu T, Leinhard O, Persson A et al (2014) Brown adipose tissue in humans: detection and functional analysis using PET (positron emission tomography), MRI (magnetic resonance imaging), and DECT (dual energy computed tomography). Methods Enzymol 537:141–159
Lee P, Brychta R, Linderman J, Smith S, Chen K, Celi F (2013) Mild cold exposure modulates fibroblast growth factor 21 (FGF21) diurnal rhythm in humans: relationship between FGF21 levels, lipolysis, and cold-induced thermogenesis. J Clin Endocrinol Metab 98(1):E98–E102
Chen Y, Cypess A, Sass C, Brownell A, Jokivarsi K et al (2012) Anatomical and functional assessment of brown adipose tissue by magnetic resonance imaging. Obesity (Silver Spring, Md) 20(7):1519–1526
Positano V, Gastaldelli A, Sironi AM, Santarelli MF, Lombardi M, Landini L (2004) An accurate and robust method for unsupervised assessment of abdominal fat by MRI. J Magn Reson Imaging 20(4):684–689
Kullberg J, Ahlström H, Johansson L, Frimmel H (2007) Automated and reproducible segmentation of visceral and subcutaneous adipose tissue from abdominal MRI. Int J Obes (Lond) 31(12):1806–1817
Liou TH, Chan WP, Pan LC, Lin PW, Chou P, Chen CH (2006) Fully automated large-scale assessment of visceral and subcutaneous abdominal adipose tissue by magnetic resonance imaging. Int J Obes (Lond) 30(5):844–852
Ranefall P, Bidar AW, Hockings PD (2009) Automatic segmentation of intra-abdominal and subcutaneous adipose tissue in 3D whole mouse MRI. J Magn Reson Imaging 30(3):554–560
Tang Y, Sharma P, Nelson MD, Simerly R, Moats RA (2011) Automatic abdominal fat assessment in obese mice using a segmental shape model. J Magn Reson Imaging 34(4):866–873
Rasmussen JM, Entringer S, Nguyen A, van Erp TGM, Guijarro A, Oveisi F et al (2013) Brown adipose tissue quantification in human neonates using water–fat separated MRI. PLoS ONE 8(10):e77907. doi:10.1371/journal.pone.0077907
Sandouk A, Bagci U, Xu Z, Mansoor A, Foster B, Mollura D (2013) Accurate quantification of brown adipose tissue through PET-guided CT image segmentation. J Nucl Med 54(2):318
Hu HH, Wu T-W, Yin L, Kim MS, Chia JM, Perkins TG, Gilsanz V (2014) MRI detection of brown adipose tissue with low fat content in newborns with hypothermia. Magn Reson Imaging 32(2):107–117. doi:10.1016/j.mri.2013.10.003
Hardy PA, Hinks RS, Tkach JA (1995) Seperation of fat and water in fast spin echo MR imaging with the three point Dixon technique. J Magn Reson Imaging 5:181–185
Berglund J, Johansson L, Ahlström H, Kullberg J (2010) Three-point dixon method enables whole-body water and fat imaging of obese subjects. Magn Reson Med 63(6):1659–1668
Hu HH, Wu T-W, Yin L et al (2014) MRI detection of brown adipose tissue with low fat content in newborns with hypothermia. Magn Reson Imaging 32(2):107–117
Tam CS, Lecoultre V, Ravussin E (2012) Brown adipose tissue: mechanisms and potential therapeutic targets. Circulation 125(22):2782–2791. doi:10.1161/CIRCULATIONAHA.111.042929
Symonds ME (2013) Brown adipose tissue growth and development. Scientifica 2013:305763. doi:10.1155/2013/305763
Bartelt A, Heeren J (2014) Adipose tissue browning and metabolic health. Nat Rev Endocrinol 10:24–36
Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S et al (2006) User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. Neuroimage 31(3):1116–1128
Kriegel HP, Kroger P, Zimek A (2010) Outlier detection techniques. In: The 2010 SIAM international conference on data mining, tutorial notes 2010
Kriegel H-P, Kröger P, Schubert E, Zimek A (2009a) LoOP: local outlier probabilities. In: Proceedings ACM conference on information and knowledge management (CIKM), Hong Kong, China
Ben-Gal I (2005) Outlier detection. In: Maimon O, Rockach L (eds) Data mining and knowledge discovery handbook: a complete guide for practitioners and researchers. Kluwer Academic Publishers. ISBN: 0-387-24435-2
Bezdek JC, Hall LO, Clarke LP (1993) Review of MR image segmentation techniques using pattern recognition. Med Phys 20(4):1033–1048
Jain AK, Duin RPW, Mao J (2000) Statistical pattern recognition: a review. IEEE Trans Pattern Anal Mach Intell 22(1):4–37
Du KL (2010) Clustering: a neural network approach. Neural Netw 23:89–107
Stekh Y, Sardieh FME, Lobar M (2009) Neural network based clustering algorithm. In: Proceedings of the 5th international conference, perspective technologies and methods in MEMS design, Zakarpattya, pp 168–169
Pratt WK (2007) Digital image processing: PIKS inside. Wiley, Los Altos
Gonzalez R, Woods R (2002) Digital image processing. Prentice Hall, New Jersey
Dunn JC (1973) A fuzzy relative of the ISODATA process and its use in detecting compact well separated clusters. J Cybern 3:32–57
Bezdek JC (1981) Pattern recognition with fuzzy objective function algorithms. Plenum, NY
Bezdek JC, Ehrlich R, Full W (1984) FCM: the fuzzy c-means clustering algorithm. Comput Geosci 10(2–3):191–203
Egmont-Petersen M, de Ridder D, Handels H (2002) Image processing with neural networks a review. Pattern Recognit 35(10):2279–2301
Zou KH, Warfield SK, Bharatha A, Tempany MC, Kaus MR et al (2004) Statistical validation of image segmentation quality based on a spatial overlap. Acad Radiol 11(2):178–189
Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH et al (2005) Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 58(10):982–990
Srour H, Chuang KH (2015) Functional imaging of brown fat in mouse. In: ISMRM 2015. Abstract—4675
Acknowledgments
The work was supported by the Intramural Research program of the Singapore Bioimaging Consortium, Biomedical Sciences Institutes, Agency for Science, Technology and Research (A*STAR), Singapore.
Authors contribution
Bhanu Prakash KN, Hussein Srour, Kai-Hsiang Chuang: Protocol/project development; Hussein Srour: Data collection or management; Bhanu Prakash KN, S. Sendhil Velan: Data analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All animal experiments were approved by and complied with regulations set forth by the local Institutional Animal Care and Use Committee (A*STAR, Singapore).
Additional information
K. N. Bhanu Prakash and Hussein Srour are to be considered as joint first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhanu Prakash, K.N., Srour, H., Velan, S.S. et al. A method for the automatic segmentation of brown adipose tissue. Magn Reson Mater Phy 29, 287–299 (2016). https://doi.org/10.1007/s10334-015-0517-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-015-0517-0