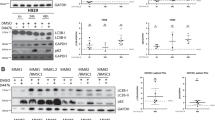
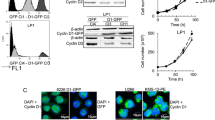
Abstract
The unfolded protein response (UPR) is an essential pathway for both normal and malignant plasma cells to maintain endoplasmic reticulum (ER) homeostasis in response to the large amount of immunoglobulin (Ig) output. The inositol-requiring enzyme 1-X-box binding protein-1 (IRE1-XBP-1) arm of the UPR pathway has been shown to play crucial roles not only in relieving the ER stress by up-regulating a series of genes favoring ER-associated protein degradation and protein folding, but in mediating terminal plasmacytic differentiation and maturation. Myeloma cells comprise various subsets arrested in diverse differentiated phases, and the immaturity of myeloma cells has been taken as a marker for poor prognosis, suggesting that differentiation induction would be a promising therapeutic strategy for myeloma. Herein, we used low-dose pharmacological UPR inducers such as tunicamycin (TM) and dithiothreitol (DTT) to efficiently activate the IRE1-XBP-1 pathway in myeloma cells characterized by transcriptional expression increase in spliced XBP-1 and molecular chaperons, accompanied by significant differentiation and maturation of these myeloma cells, without concomitant cytotoxicity. These differentiated myeloma cells exhibited a more mature appearance with well-developed cytoplasm and a reduced nucleocytoplasmic ratio, and a further differentiated phenotype with markedly increased expression of CD49e together with significantly elevated cellular secretion of Ig light chain as shown by flow cytometry and ELISA, in contrast to the control myeloma cells without exposed to TM or DTT. Moreover, siRNA knockdown of XBP-1 disrupted TM- or DTT-induced myeloma cell differentiation and maturation. Our study, for the first time, validated that the modest activation of the UPR pathway enables myeloma cells to further differentiate, and identified that XBP-1 plays an indispensable role in UPR-mediated myeloma cell differentiation and maturation. Thus, we provided the rationale and feasibility for the exploration of the novel therapeutic strategy of differentiation induction for plasmacytic malignancies.






Similar content being viewed by others
Abbreviations
- ER:
-
Endoplasmic reticulum
- UPR:
-
Unfolded protein response
- XBP-1:
-
X-box binding protein-1
- GRP78:
-
Glucose-regulated protein 78
- GRP94:
-
Glucose-regulated protein 94
- MM:
-
Multiple myeloma
- PI:
-
Proteasome inhibitor
References
Laubach J, Richardson P, Anderson K (2011) Multiple myeloma. Annu Rev Med 62:249–264
Stewart AK (2012) Novel therapeutics in multiple myeloma. Hematology 17(Suppl 1):S105–S108
Ferrarini M, Ferrero E (2011) Proteasome inhibitors and modulators of angiogenesis in multiple myeloma. Curr Med Chem 18:5185–5195
Mi J (2011) Current treatment strategy of acute promyelocytic leukemia. Front Med 5:341–347
Kamimura T, Miyamoto T, Harada M, Akashi K (2011) Advances in therapies for acute promyelocytic leukemia. Cancer Sci 102:1929–1937
Al-Sahmani M, Trnavska I, Sevcikova S, Antosova M, Antosova L, Kissova J, Adam Z, Pour L, Nemec P, Greslikova H, Krejci M, Zahradova L, Bulikova A, Penka M, Hajek R (2011) Prognostic significance of morphological assessment of plasma cells in multiple myeloma. Neoplasma 58:554–560
San Miguel JF, Garcia-Sanz R, Gonzalez M, Orfao A (1995) Immunophenotype and DNA cell content in multiple myeloma. Baillieres Clin Haematol 8:735–759
Takishita M, Kosaka M, Goto T, Saito S (1994) Cellular origin and extent of clonal involvement in multiple myeloma: genetic and phenotypic studies. Br J Haematol 87:735–742
Hou J, Xiong H, Gao W, Jiang H (2005) 2-Methoxyestradiol at low dose induces differentiation of myeloma cells. Leuk Res 29:1059–1067
Zhou X, Jiang H, Hou J (2007) Coordination of upregulated XBP-1 and downregulated c-myc during myeloma cell differentiation induced by 2-methoxyestradiol. Leuk Res 31:1259–1265
Jiang H, Gao W, Sze DM, Xiong H, Hou J (2007) Transcription factors Xbp-1, Blimp-1, and BSAP are involved in the regulation of plasmacytic differentiation induced by 2-methoxyestradiol in myeloma cell lines. Int J Hematol 86:429–437
Kallies A, Hasbold J, Fairfax K, Pridans C, Emslie D, McKenzie BS, Lew AM, Corcoran LM, Hodgkin PD, Tarlinton DM, Nutt SL (2007) Initiation of plasma-cell differentiation is independent of the transcription factor Blimp-1. Immunity 26:555–566
Nutt SL, Taubenheim N, Hasbold J, Corcoran LM, Hodgkin PD (2011) The genetic network controlling plasma cell differentiation. Semin Immunol 23:341–349
Ali MM, Bagratuni T, Davenport EL, Nowak PR, Silva-Santisteban MC, Hardcastle A, McAndrews C, Rowlands MG, Morgan GJ, Aherne W, Collins I, Davies FE, Pearl LH (2011) Structure of the Ire1 autophosphorylation complex and implications for the unfolded protein response. EMBO J 30:894–905
Michallet AS, Mondiere P, Taillardet M, Leverrier Y, Genestier L, Defrance T (2011) Compromising the unfolded protein response induces autophagy-mediated cell death in multiple myeloma cells. PLoS ONE 6:e25820
Baumann P, Mandl-Weber S, Völkl A, Adam C, Bumeder I, Oduncu F, Schmidmaier R (2009) Dihydroorotate dehydrogenase inhibitor A771726 (leflunomide) induces apoptosis and diminishes proliferation of multiple myeloma cells. Mol Cancer Ther 8:366–375
Greipp PR, Raymond NM, Kyle RA, O’Fallon WM (1985) Multiple myeloma: significance of plasmablastic subtype in morphological classification. Blood 65:305–310
Stommel JM, Wahl GM (2004) Accelerated MDM2 auto-degradation induced by DNA-damage kinases is required for p53 activation. EMBO J 23:1547–1556
Schild-Poulter C, Shih A, Yarymowich NC, Haché RJ (2003) Down-regulation of histone H2B by DNA-dependent protein kinase in response to DNA damage through modulation of octamer transcription factor 1. Cancer Res 63:7197–7205
Cirstea D, Hideshima T, Rodig S, Santo L, Pozzi S, Vallet S, Ikeda H, Perrone G, Gorgun G, Patel K, Desai N, Sportelli P, Kapoor S, Vali S, Mukherjee S, Munshi NC, Anderson KC, Raje N (2010) Dual inhibition of akt/mammalian target of rapamycin pathway by nanoparticle albumin-bound-rapamycin and perifosine induces antitumor activity in multiple myeloma. Mol Cancer Ther 9:963–975
Kawano MM, Huang N, Harada H, Harada Y, Sakai A, Tanaka H, Iwato K, Kuramoto A (1993) Identification of immature and mature myeloma cells in the bone marrow of human myelomas. Blood 82:564–570
Obeng EA, Carlson LM, Gutman DM, Harrington WJ Jr, Lee KP, Boise LH (2006) Proteasome inhibitors induce a terminal unfolded protein response in multiple myeloma cells. Blood 107:4907–4916
Fujii R, Ishikawa H, Mahmoud MS, Asaoku H, Kawano MM (1999) MPC-1-CD49e- immature myeloma cells include CD45+ subpopulations that can proliferate in response to IL-6 in human myelomas. Br J Haematol 105:131–140
Otsuyama K, Asaoku H, Kawano MM (2006) An increase in MPC-1- and MPC-1-CD45+ immature myeloma cells in the progressive states of bone marrow plasmacytosis: the revised phenotypic classification of monoclonal marrow plasmacytosis (MOMP-2005). Int J Hematol 83:39–43
Grignani G, Gobbi PG, Formisano R, Pieresca C, Ucci G, Brugnatelli S, Riccardi A, Ascari E (1996) A prognostic index for multiple myeloma. Br J Cancer 73:1101–1107
Kurabayashi H, Kubota K, Tsuchiya J, Murakami H, Tamura J, Naruse T (1999) Prognostic value of morphological classifications and clinical variables in elderly and young patients with multiple myeloma. Ann Hematol 78:19–23
Greipp PR (1992) Advances in the diagnosis and management of myeloma. Semin Hematol 29(suppl 2):24–45
Pasqualetti P, Casale R, Collacciani A, Abruzzo BP, Colantonio D (1990) Multiple myeloma: relationship between survival and cellular morphology. Am J Hematol 33:145–147
Ma Y, Shimizu Y, Mann MJ, Jin Y, Hendershot LM (2010) Plasma cell differentiation initiates a limited ER stress response by specifically suppressing the PERK-dependent branch of the unfolded protein response. Cell Stress Chaperones 15:281–293
Drori A, Tirosh B (2011) Regulation of immunoglobulin synthesis, modification, and trafficking by the unfolded protein response a quantitative approach. Methods Enzymol 491:309–325
Aragon IV, Barrington RA, Jackowski S, Mori K, Brewer JW (2012) The specialized unfolded protein response of B lymphocytes: ATF6α-independent development of antibody-secreting B cells. Mol Immunol 51:347–355
Gass JN, Gifford NM, Brewer JW (2002) Activation of an unfolded protein response during differentiation of antibody-secreting B cells. J Biol Chem 277:49047–49054
Gass JN, Jiang HY, Wek RC, Brewer JW (2008) The unfolded protein response of B-lymphocytes: PERK- independent development of antibody- secreting cells. Mol Immunol 45:1035–1043
Hu CC, Dougan SK, McGehee AM, Love JC, Ploegh HL (2009) XBP-1 regulates signal transduction, transcription factors and bone marrow colonization in B cells. EMBO J 28:1624–1636
Todd DJ, Lee AH, Glimcher LH (2008) The endoplasmic reticulum stress response in immunity and autoimmunity. Nat Rev Immunol 8:663–674
Davenport EL, Moore HE, Dunlop AS, Sharp SY, Workman P, Morgan GJ, Davies FE (2007) Heat shock protein inhibition is associated with activation of the unfolded protein response pathway in myeloma plasma cells. Blood 110:2641–2649
Marcu MG, Doyle M, Bertolotti A, Ron D, Hendershot L, Neckers L (2002) Heat shock protein 90 modulates the unfolded protein response by stabilizing IRE1alpha. Mol Cell Biol 22:8506–8513
Meister S, Schubert U, Neubert K, Herrmann K, Burger R, Gramatzki M, Hahn S, Schreiber S, Wilhelm S, Herrmann M, Jäck HM, Voll RE (2007) Extensive immunoglobulin production sensitizes myeloma cells for proteasome inhibition. Cancer Res 67:1783–1792
Ling SC, Lau EK, Al-Shabeeb A, Nikolic A, Catalano A, Iland H, Horvath N, Ho PJ, Harrison S, Fleming S, Joshua DE, Allen JD (2012) Response of myeloma to the proteasome inhibitor bortezomib is correlated with the unfolded protein response regulator XBP-1. Haematologica 97:64–72
Leung-Hagesteijn C, Erdmann N, Cheung G, Keats JJ, Stewart AK, Reece DE, Chung KC, Tiedemann RE (2013) XBP-1 s-negative tumor B cells and pre-plasmablasts mediate therapeutic proteasome inhibitor resistance in multiple myeloma. Cancer Cell 24:289–304
Acknowledgments
This study was supported in part by funds from National Natural Science Foundation of China (NSFC, Grant Numbers: 30770924 and 30800489). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hua Jiang and Jianfeng Zou have contributed equally to this work and are considered co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10238_2013_269_MOESM1_ESM.tif
Supplementary material 1 (TIFF 9264 kb): siRNA knockdown of XBP-1 aborted TM- and DTT-induced increase of CD49e expression proportion and Ig light chain production in primary myeloma cells. The primary myeloma cells from 3 newly diagnosed patients were transfected by XBP-1 siRNA or mock (non-silencing siRNA), followed by incubation with 0.4 μM TM or 0.5 mM DTT for 72 h. The CD49e expression levels (A) and the concentration of light chain protein in the supernatant (B) were determined by flow cytometry or ELISA
Rights and permissions
About this article
Cite this article
Jiang, H., Zou, J., Zhang, H. et al. Unfolded protein response inducers tunicamycin and dithiothreitol promote myeloma cell differentiation mediated by XBP-1. Clin Exp Med 15, 85–96 (2015). https://doi.org/10.1007/s10238-013-0269-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-013-0269-y