Abstract
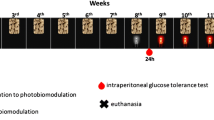
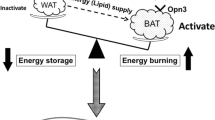
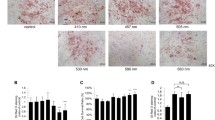
Low-level laser therapy (LLLT) is known for its ability to induce a photochemical process, primarily targeting mitochondria, a process referred to as photobiomodulation (PBM). Recently, its use has been attributed as an adjunct in obesity treatment, to stimulate lipolysis and apoptosis. However, the pathway of stimulation remains uncertain. Thus, the objective of this study was to understand whether mitochondrial stimulation occurs in adipose tissue cells after PBM therapy, which could lead to the processes of lipolysis and apoptosis. A non-randomized clinical trial was conducted using a split abdomen design in obese women who received red and infrared LED photobiomodulation therapy (PBMT). The patients underwent bariatric surgery, and adipose tissue samples were collected for immunohistochemical analysis with primary mitochondrial antibodies. Adipose tissue samples subjected to LED intervention exhibited positivity in mitochondrial antibodies for cAMP, DRP1, FAS, FIS1, MFN2, and OPA1 (p<0.001) compared to the control group. In conclusion, we observed that PBMT was capable of generating mitochondrial stimulation in adipose tissue cells, as evidenced by the positive antibody signals. This finding suggests that mitochondrial stimulation could be the mechanism and action underlying adipose tissue lipolysis and apoptosis.




Similar content being viewed by others
References
Mansano BSDM, da Rocha VP, Antonio EL, Peron DF, do Nascimento de Lima R, Tucci PJF, Serra AJ. (2021) Enhancing the therapeutic potential of mesenchymal stem cells with light-emitting diode: implications and molecular mechanisms. Oxidative Med Cell Longev 3:6663539
Huang YY, Sharma SK, Carroll J, Hamblin MR (2011) Biphasic dose response in low level light therapy - an update. Dose-Response 9(4):602–618
Huang A, Nguyen JK, Jagdeo J (2020) Light-emitting diode-based photodynamic therapy for photoaging, scars, and dyspigmentation: a systematic review. Dermatol Surg 46(11):1388–1394
Jagdeo J, Austin E, Mamalis A, Wong C, Ho D, Siegel DM (2018) Light-emitting diodes in dermatology: a systematic review of randomized controlled trials. Lasers Surg Med 50(6):613–628
Nassab R (2015) The evidence behind noninvasive body contouring devices. Aesthet Surg J 35(3):279–293
Campos RMS, Dâmaso AR, Masquio DCL et al (2018) The effects of exercise training associated with low-level laser therapy on biomarkers of adipose tissue transdifferentiation in obese women. Lasers Med Sci 33(6):1245–1254
Liu PF, Hu YC, Kang BH, Tseng YK, Wu PC, Liang CC, Hou YY, Fu TY, Liou HH, Hsieh IC, Ger LP, Shu CW (2017) Expression levels of cleaved caspase-3 and caspase-3 in tumorigenesis and prognosis of oral tongue squamous cell carcinoma. PLoS One 12(7):e0180620
Modena DAO, Soares CD, Martignago CCS, Almeida S, Cazzo E, Chaim EA (2022) Effects of LED photobiomodulation therapy on the subcutaneous fatty tissue of obese individuals - histological and immunohistochemical analysis. s 8:1–7
Xie D, Li YL, Wang GF, Jiang J, Sun LR (2021) Ultraviolet light-emitting diode irradiation induces reactive oxygen species production and mitochondrial membrane potential reduction in HL-60 cells. J Int Med Res 49(5):3000605211016623
Chaim EA, Pareja JC, Gestic MA et al (2017) Programa multidisciplinar pré-operatório de cirurgia bariátrica: uma proposta para o Sistema Único de Saúde. Arq Gastroenterol 54(1):70–74
National Institutes of Health (1992) Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference statement. Am J Clin Nutr 55(l):615S–619S
Mester E, Mester AE, Mester A (1985) The biomedical effect of laser application. Lasers Surg Med 5(1):31–39
Nicholls DG (2002) Mitochondrial function and dysfunction in the cell: its relevance to aging and aging-related disease. Int J Biochem Cell Biol 34(11):1372–1381
Karu TI (2010) Multiple roles of cytochrome c oxidase in mammalian cells under action of red and IR-A radiation. IUBMB Life 62(8):607–610
de Freitas LF, Hamblin MR (2016) Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J Sel Top Quantum Electron 22(3):348–364
Wu S, Zhou F, Zhang Z, Xing D (2011a) Mitochondrial oxidative stress causes mitochondrial fragmentation via differential modulation of mitochondrial fission–fusion proteins. FEBS J 278(6):941–954
Gwinn DM, Shackelford DB, Egan DF et al (2008) AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 30(2):214–226
Suen DF, Norris KL, Youle RJ (2008) Mitochondrial dynamics and apoptosis. Genes Dev 22(12):1577–1590
Chang CR, Blackstone C (2010) Dynamic regulation of mitochondrial fission through modification of the dynamin-related protein Drp1. Ann N Y Acad Sci 1201:34–39
Nomura J, Matsumoto K, Iguchi-Ariga SMM et al (2005) Positive regulation of Fas gene expression by MSSP and abrogation of Fas-mediated apoptosis induction in MSSP-deficient mice. Exp Cell Res 305(2):324–332
Olichon L, Baricault N, Gas E et al (2003) Loss of OPA1 perturbates the mitochondrial inner membrane structure and integrity, leading to cytochrome-c release and apoptosis. J Biol Chem 278:7743–7746
John GB, Shang Y, Li L et al (2005) The mitochondrial inner membrane protein mitofilin controls cristae morphology. Mol Biol Cell 16:1543–1554
Bach D, Pich S, Soriano FX et al (2003) Mitofusin-2 determines mitochondrial network architecture and mitochondrial metabolism. A novel regulatory mechanism altered in obesity. J Biol Chem 278:17190–17197
Pich S, Bach D, Briones P et al (2005) The Charcot-Marie-Tooth type 2A gene product, Mfn2, up-regulates fuel oxidation through the expression of OXPHOS system. Hum Mol Genet 14:1405–1415
Author information
Authors and Affiliations
Contributions
PhD. D. A. O. M., data collection and analysis, writing of the article. MSc. A. P. F., data collection and analysis, writing of the article. PhD. E. C. de O. G., contributions in the writing of the article. PhD. E. C., data collection, joint supervision of the research and contributions in the writing of the article. PhD. E. A. C., data collection, supervision of the research and contributions in the writing of the article.
Corresponding author
Ethics declarations
Ethical approval
The research was not submitted to an ethics committee, as it was a review study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Modena, D.A.O., Ferro, A.P., de Oliveira Guirro, E.C. et al. Photobiomodulation therapy with light-emitting diode in stimulating adipose tissue mitochondria. Lasers Med Sci 38, 238 (2023). https://doi.org/10.1007/s10103-023-03906-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-023-03906-y