Abstract
Objective
This study aimed to evaluate the effects of chronic consumption of cachaça, a Brazilian beverage containing alcohol, on submandibular glands (SM) of rats by using histomorphometric and biochemical parameters.
Materials and methods
Twenty-four male rats (40 days of age) were assigned into the following four groups (n = 6): two control groups for 75 days (C75) and 105 days (C105), and two experimental groups of cachaça ingestion with ascending concentrations for consecutive 75 days (CA75) and 105 days (CA105). On the right SM glands, the striated, granular and acini ducts were processed for histomorphometric analysis. The left SM glands were weighed and stored at − 80 °C, to evaluate through biochemical tests carried out by spectrophotometric methods, the functional activity of total acid phosphatase (TAP), tartrate-resistant acid phosphatase (TRAP), and alkaline phosphatase (ALP) and to determine the mucin levels.
Results
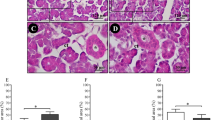
The absolute and relative weights of the SM glands in both experimental groups were reduced in relation to the controls (p < 0.05). The histomorphometric analysis showed a significant reduction of the acini area (p < 0.05) and non-relevant reduction of striated ducts (p > 0.05). The granular ducts did not show a significant increase of the area (p > 0.05). The TAP and TRAP activities were significantly decreased in the experimental groups (p < 0.05), while the ALP functional activity decreased moderately (p > 0.05). Mucin levels also had a significant reduction when compared with the control groups (p < 0.05).
Conclusions
Chronic consumption of cachaça can cause morphological changes associated with glandular atrophy, loss of biochemical functionality of phosphatases, and the reduction of mucin synthesis.
Clinical relevance
The consumption of cachaça can compromise the functions of the submandibular glands by altering their morphology and enzymatic activity.




Similar content being viewed by others
References
Mehta AJ (2016) Alcoholism and critical illness: a review. World J Crit Care Med 5:27–35
World Health Organization (2014) Global status report on alcohol and health. Geneva: WHO Library Cataloguing-in-Publication Data 2014:http://wwwwhoint/substance_abuse/publications/global_alcohol_report/en/indexhtml/ Accessed 29 November 2018
Riedel F, Goessler UR, Hormann K (2005) Alcohol-related diseases of the mouth and throat. Dig Dis 23:195–203
Enberg N, Alho H, Loimaranta V, Lenander-Lumikari M (2001) Saliva flow rate, amylase activity, and protein and electrolyte concentrations in saliva after acute alcohol consumption. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 92:292–298
Waszkiewicz N, Zalewska-Szajda B, Zalewska A, Waszkiewicz M, Szajda SD, Repka B, Szulc A, Kępka A, Minarowska A, Chojnowska S, Konarzewska B, Ładny JR, Kowzan U, Zwierz K (2012) Decrease in salivary lactoferrin output in chronically intoxicated alcohol-dependent patients. Folia Histochem Cytobiol 50:248–254
Waszkiewicz N, Zalewska-Szajda B, Zalewska A, Waszkiewicz M, Szajda SD, Repka B, Szulc A, Kepka A, Minarowska A, Ladny JR, Zwierz K (2012) Salivary lysozyme in smoking alcohol dependent persons. Folia Histochem Cytobiol 50:609–612
Waszkiewicz N, Zalewska A, Szajda SD, Waszkiewicz M, Szulc A, Kepka A, Konarzewska B, Minarowska A, Zalewska-Szajda B, Wilamowska D, Waszkiel D, Ladny JR, Zwierz K (2012) The effect of chronic alcohol intoxication and smoking on the output of salivary immunoglobulin a. Folia Histochem Cytobiol 50:605–608
Waszkiewicz N, Zalewska A, Szulc A, Kepka A, Konarzewska B, Zalewska-Szajda B et al (2011) The influence of alcohol on the oral cavity, salivary glands and saliva. Pol Merkur Lekarski 30:69–74
Waszkiewicz N, Chojnowska S, Zalewska A, Zwierz K, Szulc A, Szajda SD (2013) Salivary hexosaminidase in smoking alcoholics with bad periodontal and dental states. Drug Alcohol Depend 129:33–40
Slomiany BL, Liau YH, Zalesna G, Slomiany A (1988) Effect of ethanol on the in vitro sulfation of salivary mucin. Alcohol Clin Exp Res 12:774–779
Busch L, Sterin-Borda L, Borda E (2008) Beta-adrenoceptor alterations coupled with secretory response and experimental periodontitis in rat submandibular glands. Arch Oral Biol 53:509–516
Song JY, Tigchelaar W, Schellens JP, Van Marle J, Van Noorden CJ, Frederiks WM (1996) Ultrastructural localization of activity of phosphatases by low temperature incubation of unfixed cryostat sections. Histochem Cell Biol 106:351–355
Chaves Neto AH, Sassaki KT, Nakamune ACdMS (2011) Protein phosphatase activities in the serum and saliva of healthy children. RPG Rev Pós Grad 18:90–95
Isacsson GA (1986) Histochemical study of rat salivary gland acid phosphatase. Arch Oral Biol 31:95–99
Bastos MF, Gaag GL, Romero JR, Gabrili JJ, Marques MR, Duarte PM (2014) Effects of Cachaça, a typical Brazilian alcoholic beverage, on alveolar bone loss and density: a study in peripubertal rats. Arch Oral Biol 59:82–91
Ministério da Agricultura Pecuária e Abastecimento do Brasil (2009) Decreto n°. 6871/2009, de 5 de junho de 2009. Regulamenta a Lei 8918, de 14 de julho de 1994, que fornece diretrizes para a inspeção do registro da classificação de padronização e o monitoramento da produção de bebidas, vol. 1. Brasília: Diário oficial da União. p. 20. (in Portuguese)
Instituto Brasileiro de Cachaça (2019) Serviços, estatísticas, mercado externo, http://www.ibrac.net/index.php/servicos/estatisticas/mercado-externo/ Acessed 10 november 2019 (in Portuguese)
Silva ARS, Ribeiro ACP, Salzedas LM, Soubhia AMP, Sundefeld MLMM (2007) Análise da densidade óssea radiográfica de ratos submetidos ao alcoolismo crônico utilizando imagem digital. Revista Odonto Ciência 22:77–81
Fernandes LM, Teixeira FB, Alves-Junior SM, Pinheiro JJ, Maia CS, Lima RR (2015) Immunohistochemical changes and atrophy after chronic ethanol intoxication in rat salivary glands. Histol Histopathol 30:1069–1078
Hartree EF (1972) Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal Biochem 48:422–427
Janckila AJ, Parthasarathy RN, Parthasarathy LK, Seelan RS, Hsueh YC, Rissanen J, Alatalo SL, Halleen JM, Yam LT (2005) Properties and expression of human tartrate-resistant acid phosphatase isoform 5a by monocyte-derived cells. J Leukoc Biol 77:209–218
Hall RL, Miller RJ, Peatfield AC, Richardson PS, Williams I, Lampert I (1980) A colorimetric assay for mucous glycoproteins using Alcian blue [proceedings]. Biochem Soc Trans 8:72
Sarosiek J, Rourk RM, Piascik R, Namiot Z, Hetzel DP, McCallum RW (1994) The effect of esophageal mechanical and chemical stimuli on salivary mucin secretion in healthy individuals. Am J Med Sci 308:23–31
Lieber CS (1991) Hepatic, metabolic and toxic effects of ethanol 1991 uptead. Alcohol Clin Exp Res 15:573–592
Scott J, Burns J, Flower EA (1988) Histological analysis of parotid and submandibular glands in chronic alcohol abuse: a necropsy study. J Clin Pathol 41:837–840
Dutta SK, Orestes M, Vengulekur S, Kwo P (1992) Ethanol and human saliva: effect of chronic alcoholism on flow rate, composition, and epidermal growth factor. Am J Gastroenterol 87:350–354
Tirapelli L, Tirapelli C, Tirapelli D, Cassel F, Petroni S, Tamega O (2001) Morphometric analysis of seromucous acini and granular ducts of submandibular glands from rats (Rattus norvectus) submitted to experimental chronic alcoholism. Rev Chil Anat 3:263–269
Proctor GB, Carpenter GH (2014) Salivary secretion: mechanism and neural regulation. Monogr Oral Sci 24:14–29
Amano O, Mizobe K, Bando Y, Sakiyama K (2012) Anatomy and histology of rodent and human major salivary glands: overview of the Japan salivary gland society-sponsored workshop. Acta Histochem Cytochem 45:241–250
Faustino SE, Stipp AC (2003) Effects of chronic alcoholism and alcoholic detoxication on rat submandibular glands: morphometric study. J Appl Oral Sci 11:21–26
Fagundes NC, Fernandes LM, Paraense RS, de Farias-Junior PM, Teixeira FB, Alves-Junior SM et al (2016) Binge drinking of ethanol during adolescence induces oxidative damage and morphological changes in salivary glands of female rats. Oxidative Med Cell Longev 2016:1–11
Banderas JA, Gaitan LA, Portilla J, Aguirre A (1992) Effects of chronic ethanol consumption on the rat parotid gland. Arch Oral Biol 37:69–72
Carranza M, Ferraris ME, Galizzi M (2005) Structural and morphometrical study in glandular parenchyma from alcoholic sialosis. J Oral Pathol Med 34:374–379
Maciejewski R, Hermanowicz-Dryka T, Czerny K, Wójtowicz Z, Dryka T (1999) Changes of the ATPase, acid phosphatase and alkaline phosphatase reaction intensity in the parotid and submandibular glands of rabbits in experimental diabetes. Folia Histochem Cytobiol 37:99–100
Basseto A, Neves F, Toledo S, Silva T, Buzalaf M, Faria F et al (2001) Effect of cigarette smoke on sublingual gland and liver phosphotyrosine protein phosphatase and acid phosphatase activities. Rev FOB 9:49–54
Campos SC, Moreira DA, Nunes TD, Colepicolo P, Brigagão MR (2005) Oxidative stress in alcohol-induced rat parotid sialadenosis. Arch Oral Biol 50:661–668
Purushotham KR, Wang P, Humphreys-Beher MG (1995) Effect of vanadate on amylase secretion and protein tyrosine phosphatase activity in the rat parotid gland. Mol Cell Biochem 152:87–94
Slomiany BL, Slomiany A (2010) Ghrelin protection against cytotoxic effect of ethanol on rat salivary Mucin synthesis involves cytosolic phospholipase A2 activation through S-Nitrosylation. Int J Biomed Sci 6:37–44
Renato Romero J, Krause Neto W, Sabbag da Silva A, Luiz Dos Santos E, Aurélio Added M, Pianca E et al (2018) Chronic cachaça consumption affects the structure of tibial bone by decreasing bone density and density of mature collagen fibers in middle-aged Wistar rats. Aging Male:1–6
Moreira RN, Marinho SA, Verli FD, Douglas-de-Oliveira DW, Mesquita AT, Lima NL (2018) Computer-assisted analysis of tongue thickness of golden hamster (Mesocricetus auratus) following topical chronic exposure to distilled alcoholic beverages. Anat Histol Embryol 47:428–434
Funding
The project was developed without funding.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and the design of the study. Ketelin Juliane Dal Prá, Aneliza de Fatima Moraes da Silva, Antonio Hernades Chaves Neto, and Ana Maria Pires Soubhia acquired the data. Ketelin Juliane Dal Prá, Aneliza de Fatima Moraes da Silva, and Antonio Hernades Chaves Neto analyzed and interpreted the data. Ketelin Juliane Dal Prá, Antonio Hernades Chaves Neto, and Ana Maria Pires Soubhia drafted the article, and all authors revised it for important intellectual content, and read and approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
Author Ketelin Juliane Dal Prá declares that he has no conflict of interest. Author Aneliza de Fátima Moraes da Silva declares that he has no conflict of interest. Author Antonio Hernandes Chaves Neto declares that he has no conflict of interest. Author Ana Maria Pires Soubhia declares that he has no conflict of interest.
Ethical approval
The research protocol was approved by the Ethics Committee on Animal Use of the Aracatuba Dental School—UNESP and all applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dal Prá, K.J., de Fatima Moraes da Silva, A., Chaves-Neto, A.H. et al. Effects of cachaça, a typical Brazilian alcoholic beverage, on submandibular glands of rats: a histomorphometric and biochemical study. Clin Oral Invest 24, 4283–4290 (2020). https://doi.org/10.1007/s00784-020-03290-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03290-9