Abstract
Background
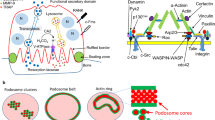
Profilin-1 (Pfn1), an evolutionarily conserved actin-binding protein, is an important regulator of the cytoskeleton. We previously reported the osteoclast-specific Pfn1-conditional knockout (cKO) mice had postnatal osteolytic phenotype with craniofacial and long-bone deformities associated with increased migration of cultured osteoclasts. We hypothesized the increased cellular processes structured with branched actin filaments may underlies the mechanism of increased bone resorption in these mutant mice.
Materials and methods
The morphological structure and cell migration of the cultured osteoclasts were analyzed using fluorescent microscopy and time-lapse image capturing. Fractional migration distances, as well as the index of protrusive structures (%-PB) that evaluates relative border length of the protrusion were compared between the cells from control and Pfn1-cKO mice.
Results
Time-lapse image analysis showed that %-PB was significantly larger in Pfn1-cKO osteoclasts. In addition, the fractional migration distance was positively correlated with the index. When the branched actin filament organization was suppressed by chemical inhibitors, the osteoclast migration was declined. Importantly, the suppression was more extensive in Pfn1-cKO than in control osteoclasts.
Conclusion
Our results indicated the causative involvement of the increased branched actin filament formation at least in part for their excessive migration. Our findings provide a mechanistic rationale for testing novel therapeutic approaches targeting branched actin filaments in osteolytic disorders.





Similar content being viewed by others
Change history
19 August 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00774-023-01462-7
References
Ralston SH, Layfield R (2012) Pathogenesis of paget disease of bone. Calcif Tissue Int 91:97–113
Ralston SH, Albagha OM (2014) Genetics of paget’s disease of bone. Curr Osteoporos Rep 12:263–271
Gennari L, Rendina D, Falchetti A, Merlotti D (2019) Paget’s disease of bone. Calcif Tissue Int 104:483–500
Merlotti D, Materozzi M, Bianciardi S, Guarnieri V, Rendina D, Volterrani L, Bellan C, Mingiano C, Picchioni T, Frosali A, Orfanelli U, Cenci S, Gennari L (2020) Mutation of PFN1 gene in an early onset, polyostotic paget-like disease. J Clin Endocrinol Metab 105:2553–2565
Scotto di Carlo F, Pazzaglia L, Esposito T, Gianfrancesco F (2020) The loss of profilin 1 causes early onset paget’s disease of bone. J Bone Miner Res 35:1387–1398
Böttcher RT, Wiesner S, Braun A, Wimmer R, Berna A, Elad N, Medalia O, Pfeifer A, Aszodi A, Costell M, Fassler R (2009) Profilin 1 is required for abscission during late cytokinesis of chondrocytes. EMBO J 28:1157–1169
Miyajima D, Hayata T, Suzuki T, Hemmi H, Nakamoto T, Notomi T, Amagasa T, Bottcher RT, Costell M, Fassler R, Ezura Y, Noda M (2012) Profilin1 regulates sternum development and endochondral bone formation. J Biol Chem 287:33545–33553
Lin W, Izu Y, Smriti A, Kawasaki M, Pawaputanon C, Bottcher RT, Costell M, Moriyama K, Noda M, Ezura Y (2018) Profilin1 is expressed in osteocytes and regulates cell shape and migration. J Cell Physiol 233:259–268
Shirakawa J, Kajikawa S, Bottcher RT, Costell M, Izu Y, Hayata T, Noda M, Ezura Y (2019) Profilin 1 negatively regulates osteoclast migration in postnatal skeletal growth, remodeling, and homeostasis in mice. JBMR Plus 3:e10130
Schachtner H, Calaminus SD, Thomas SG, Machesky LM (2013) Podosomes in adhesion, migration, mechanosensing and matrix remodeling. Cytoskeleton 70:572–589
Henty-Ridilla JC, Goode BL (2015) Global resource distribution: allocation of actin building blocks by profilin. Dev Cell 32:5–6
Takeshita S, Kaji K, Kudo A (2000) Identification and characterization of the new osteoclast progenitor with macrophage phenotypes being able to differentiate into mature osteoclasts. J Bone Miner Res 15:1477–1488
Kajikawa S, Taguchi Y, Hayata T, Ezura Y, Ueta R, Arimura S, Inoue JI, Noda M, Yamanashi Y (2018) Dok-3 and Dok-1/-2 adaptors play distinctive roles in cell fusion and proliferation during osteoclastogenesis and cooperatively protect mice from osteopenia. Biochem Biophys Res Commun 498:967–974
Hetrick B, Han MS, Helgeson LA, Nolen BJ (2013) Small molecules CK-666 and CK-869 inhibit actin-related protein 2/3 complex by blocking an activating conformational change. Chem Biol 20:701–712
Beckham Y, Vasquez RJ, Stricker J, Sayegh K, Campillo C, Gardel ML (2014) Arp2/3 inhibition induces amoeboid-like protrusions in MCF10A epithelial cells by reduced cytoskeletal-membrane coupling and focal adhesion assembly. PLoS ONE 9:e100943
Skruber K, Warp PV, Shklyarov R, Thomas JD, Swanson MS, Henty-Ridilla JL, Read TA, Vitriol EA (2020) Arp2/3 and Mena/VASP require profilin 1 for actin network assembly at the leading edge. Curr Biol 30:2651–2664 (e2655)
Destaing O, Saltel F, Géminard JC, Jurdic P, Bard F (2003) Podosomes display actin turnover and dynamic self-organization in osteoclasts expressing actin-green fluorescent protein. Mol Biol Cell 14:407–416
van den Dries K, Linder S, Maridonneau-Parini I, Poincloux R (2019) Probing the mechanical landscape - new insights into podosome architecture and mechanics. J Cell Sci 132:jcs236828
Bae YH, Ding Z, Das T, Wells A, Gertler F, Roy P (2010) Profilin1 regulates PI(3,4)P2 and lamellipodin accumulation at the leading edge thus influencing motility of MDA-MB-231 cells. Proc Natl Acad Sci USA 107:21547–21552
Bender M, Stritt S, Nurden P, van Eeuwijk JM, Zieger B, Kentouche K, Schulze H, Morbach H, Stegner D, Heinze KG, Dütting S, Gupta S, Witke W, Falet H, Fischer A, Hartwig JH, Nieswandt B (2014) Megakaryocyte-specific profilin1-deficiency alters microtubule stability and causes a Wiskott-Aldrich syndrome-like platelet defect. Nat Commun 5:4746
Schoppmeyer R, Zhao R, Cheng H, Hamed M, Liu C, Zhou X, Schwarz EC, Zhou Y, Knorck A, Schwar G, Ji S, Liu L, Long J, Helms V, Hoth M, Yu X, Qu B (2017) Human profilin 1 is a negative regulator of CTL mediated cell-killing and migration. Eur J Immunol 47:1562–1572
Acknowledgments
We thank Dr. Reinhard Fässler and Dr. Ralph Böttcher for their support providing the mutant Pfn1-flox mouse line for this research, and Dr. Sunao Takeshita for kindly providing CMG14-12 cell lines. Dr. Koichiro Komatsu and Dr. Hisashi Ideno at Tsurumi University for kind support and discussion.
Funding
This study was funded by grants-in-aid (KAKENHI: 16K10892, 19K09617 and 19K24056), and grant for Joint Usage/Research Program of Medical Research Institute, Tokyo Medical and Dental University, from the Ministry of Education, Culture, Sports, Science and Technology. This study was also supported by the research and education funds from Ehime prefecture and Imabari City, Japan.
Author information
Authors and Affiliations
Contributions
SK was responsible for data collection, data analysis, and manuscript writing. YE was responsible for the conception, designing, data analysis, manuscript writing, and editing. YI and KN was responsible for critical reviewing and discussion. MN supervised this project. AN was responsible for the conception, designing, and reviewing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no competing financial interests. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Ethical approval
All experimental protocols were approved by the animal welfare committee of the Tokyo Medical and Dental University (A2017-082A, A2018-180A, and A2019-222A).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kajikawa, S., Ezura, Y., Izu, Y. et al. Profilin-1 negatively controls osteoclast migration by suppressing the protrusive structures based on branched actin filaments. J Bone Miner Metab 40, 561–570 (2022). https://doi.org/10.1007/s00774-022-01320-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-022-01320-y