Abstract
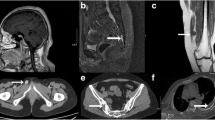
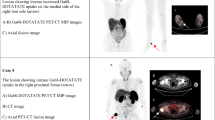
Tumor-induced osteomalacia (TIO) is a rare acquired form of hypophosphatemic osteomalacia, which is usually attributed to the overproduction of fibroblast growth factor 23 (FGF-23) by benign mesenchymal neoplasms. Localization and thereafter surgical resection of tumors lead to a cure. The present study aimed to investigate the clinical data, diagnostic methods, and follow-up after tumor resection at one medical center in Shanghai to characterize the profile of this rare disorder and to share our successful experience in diagnosis and treatment. Twenty-three patients with adult-onset hypophosphatemia osteomalacia seen in Shanghai Sixth People’s Hospital from 2009 to 2014 and 95 normal individuals were enrolled. After taking a medical history and performing a physical examination, we analyzed the laboratory results (including the serum FGF-23 levels) and localized the tumors by 18F-fluorodeoxyglucose positron emission tomography and computed tomography (18F-FDG PET/CT), 99mTc-octreotide (99mTc-OCT) scintigraphy, and magnetic resonance imaging (MRI). On the basis of the results of laboratory tests and imaging findings, tumor resection was conducted in 17 patients with a certain diagnosis of TIO. The results demonstrated that the 17 patients (nine men and eight women, average age 46.6 ± 12.9 years) had TIO. FGF-23 level was elevated in 94.1 % of patients (16 of 17 patients) . Serum phosphorus level decreased in 100 % of patients. 18F-FDG PET/CT revealed five tumors, 99mTc-OCT scintigraphy revealed two tumors, physical examination revealed nine tumors, and MRI revealed one tumor, among which 58.8 % of the causative tumors (10 of 17 tumors) were located in the lower extremities. After tumor resection, serum phosphorus levels normalized in 100 % of patients (all 17 patients) in 4–21 days and FGF-23 levels decreased in 90 % of patients (nine of ten patients). We found 64.7 % of the tumors (11 of 17 tumors) were phosphaturic mesenchymal tumors or a phosphaturic mesenchymal tumor mixed connective tissue variant. Measurement of serum phosphorus and FGF-23 levels in patients with suspected TIO is of paramount importance for diagnosing of TIO. 18F-FDG PET/CT, 99mTc-OCT scintigraphy, and physical examination play a considerable role in revealing TIO-associated tumors. TIO-associated tumors were more frequently located in the lower extremities than in other places; thus, the lower extremities need to be carefully checked. Complete surgical resection results in normalization of parameters in laboratory tests and relief of symptoms of TIO patients.



Similar content being viewed by others
References
Prader A, Illig R, Uehlinger E, Stalder G (1959) Rickets following bone tumor. Helv Paediatr Acta 14:554–565
Jiang Y, Xia WB, Xing XP, Silva BC, Li M, Wang O, Zhang HB, Li F, Jing HL, Zhong DR, Jin J, Gao P, Zhou L, Qi F, Yu W, Bilezikian JP, Meng XW (2012) Tumor-induced osteomalacia: an important cause of adult-onset hypophosphatemic osteomalacia in China: report of 39 cases and review of the literature. J Bone Miner Res 27:1967–1975
Chong WH, Molinolo AA, Chen CC, Collins MT (2011) Tumor-induced osteomalacia. Endocr Relat Cancer 18:R53–R77
Jadhav S, Kasaliwal R, Lele V, Rangarajan V, Chandra P, Shah H, Malhotra G, Jagtap VS, Budyal S, Lila AR, Bandgar T, Shah NS (2014) Functional imaging in primary tumour-induced osteomalacia: relative performance of FDG PET/CT vs somatostatin receptor-based functional scans: a series of nine patients. Clin Endocrinol (Oxf) 81:31–37
Chong WH, Andreopoulou P, Chen CC, Reynolds J, Guthrie L, Kelly M, Gafni RI, Bhattacharyya N, Boyce AM, El-Maouche D, Crespo DO, Sherry R, Chang R, Wodajo FM, Kletter GB, Dwyer A, Collins MT (2013) Tumor localization and biochemical response to cure in tumor-induced osteomalacia. J Bone Miner Res 28:1386–1398
Econs MJ, Drezner MK (1994) Tumor-induced osteomalacia—unveiling a new hormone. N Engl J Med 330:1679–1681
Jan de Beur SM (2005) Tumor-induced osteomalacia. JAMA 294:1260–1267
Fukumoto S, Takeuchi Y, Nagano A, Fujita T (1999) Diagnostic utility of magnetic resonance imaging skeletal survey in a patient with oncogenic osteomalacia. Bone 25:375–377
Dupond JL, Mahammedi H, Prie D, Collin F, Gil H, Blagosklonov O, Ricbourg B, Meaux-Ruault N, Kantelip B (2005) Oncogenic osteomalacia: diagnostic importance of fibroblast growth factor 23 and F-18 fluorodeoxyglucose PET/CT scan for the diagnosis and follow-up in one case. Bone 36:375–378
Hodgson SF, Clarke BL, Tebben PJ, Mullan BP, Cooney WP 3rd, Shives TC (2006) Oncogenic osteomalacia: localization of underlying peripheral mesenchymal tumors with use of Tc 99m sestamibi scintigraphy. Endocr Pract 12:35–42
Jan de Beur SM, Streeten EA, Civelek AC, McCarthy EF, Uribe L, Marx SJ, Onobrakpeya O, Raisz LG, Watts NB, Sharon M, Levine MA (2002) Localisation of mesenchymal tumours by somatostatin receptor imaging. Lancet 359:761–763
Walton RJ, Bijvoet OL (1975) Nomogram for derivation of renal threshold phosphate concentration. Lancet 2:309–310
Qiu ZL, Xue YL, Song HJ, Luo QY (2012) Comparison of the diagnostic and prognostic values of 99mTc-MDP-planar bone scintigraphy, 131I-SPECT/CT and 18F-FDG-PET/CT for the detection of bone metastases from differentiated thyroid cancer. Nucl Med Commun 33:1232–1242
White KE, Jonsson KB, Carn G, Hampson G, Spector TD, Mannstadt M, Lorenz-Depiereux B, Miyauchi A, Yang IM, Ljunggren O, Meitinger T, Strom TM, Juppner H, Econs MJ (2001) The autosomal dominant hypophosphatemic rickets (ADHR) gene is a secreted polypeptide overexpressed by tumors that cause phosphate wasting. J Clin Endocrinol Metab 86:497–500
White KE, Carn G, Lorenz-Depiereux B, Benet-Pages A, Strom TM, Econs MJ (2001) Autosomal-dominant hypophosphatemic rickets (ADHR) mutations stabilize FGF-23. Kidney Int 60:2079–2086
Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S, Takeuchi Y, Fujita T, Fukumoto S, Yamashita T (2001) Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia. Proc Natl Acad Sci U S A 98:6500–6505
Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, Fujita T, Nakahara K, Fukumoto S, Yamashita T (2004) FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res 19:429–435
Tenenhouse HS, Sabbagh Y (2002) Novel phosphate-regulating genes in the pathogenesis of renal phosphate wasting disorders. Pflugers Arch 444:317–326
Nelson AE, Bligh RC, Mirams M, Gill A, Au A, Clarkson A, Juppner H, Ruff S, Stalley P, Scolyer RA, Robinson BG, Mason RS, Bligh PC (2003) Clinical case seminar: fibroblast growth factor 23: a new clinical marker for oncogenic osteomalacia. J Clin Endocrinol Metab 88:4088–4094
Jagtap VS, Sarathi V, Lila AR, Malhotra G, Sankhe SS, Bandgar T, Menon P, Shah NS (2011) Tumor-induced osteomalacia: a single center experience. Endocr Pract 17:177–184
Roarke MC, Nguyen BD (2007) PET/CT localization of phosphaturic mesenchymal neoplasm causing tumor-induced osteomalacia. Clin Nucl Med 32:300–301
Malhotra G, Agrawal A, Jambhekar NA, Sarathi V, Jagtap V, Agarwal MG, Kulkarni M, Asopa RV, Shah NS (2010) Interesting image. The search for primary tumor in a patient with oncogenic osteomalacia: F-18 FDG PET resolves the conundrum. Clin Nucl Med 35:896–898
Chua SC, O’Connor SR, Wong WL, Ganatra RH (2008) Case report: solitary plasmacytoma of bone with oncogenic osteomalacia: recurrence of tumour confirmed by PET/CT. A case report with a review of the radiological literature. Br J Radiol 81:e110–e114
Clifton-Bligh RJ, Hofman MS, Duncan E, Sim Ie W, Darnell D, Clarkson A, Wong T, Walsh JP, Gill AJ, Ebeling PR, Hicks RJ (2013) Improving diagnosis of tumor-induced osteomalacia with gallium-68 DOTATATE PET/CT. J Clin Endocrinol Metab 98:687–694
Seufert J, Ebert K, Muller J, Eulert J, Hendrich C, Werner E, Schuuze N, Schulz G, Kenn W, Richtmann H, Palitzsch KD, Jakob F (2001) Octreotide therapy for tumor-induced osteomalacia. N Engl J Med 345:1883–1888
Cescato R, Schulz S, Waser B, Eltschinger V, Rivier JE, Wester HJ, Culler M, Ginj M, Liu Q, Schonbrunn A, Reubi JC (2006) Internalization of sst2, sst3, and sst5 receptors: effects of somatostatin agonists and antagonists. J Nucl Med 47:502–511
Andreopoulou P, Dumitrescu CE, Kelly MH, Brillante BA, Cutler Peck CM, Wodajo FM, Chang R, Collins MT (2011) Selective venous catheterization for the localization of phosphaturic mesenchymal tumors. J Bone Miner Res 26:1295–1302
Breer S, Brunkhorst T, Beil FT, Peldschus K, Heiland M, Klutmann S, Barvencik F, Zustin J, Gratz KF, Amling M (2014) 68Ga DOTA-TATE PET/CT allows tumor localization in patients with tumor-induced osteomalacia but negative 111In-octreotide SPECT/CT. Bone 64:222–227
Antunes P, Ginj M, Zhang H, Waser B, Baum RP, Reubi JC, Maecke H (2007) Are radiogallium-labelled DOTA-conjugated somatostatin analogues superior to those labelled with other radiometals? Eur J Nucl Med Mol Imaging 34:982–993
Naswa N, Sharma P, Kumar R, Malhotra A, Bal C (2013) Successful localization of residual culprit tumor in a case of tumor-induced osteomalacia using 68Ga-DOTANOC PET/CT. Clin Nucl Med 38:639–640
von Falck C, Rodt T, Rosenthal H, Langer F, Goesling T, Knapp WH, Galanski M (2008) 68Ga-DOTANOC PET/CT for the detection of a mesenchymal tumor causing oncogenic osteomalacia. Eur J Nucl Med Mol Imaging 35:1034
Shane E, Parisien M, Henderson JE, Dempster DW, Feldman F, Hardy MA, Tohme JF, Karaplis AC, Clemens TL (1997) Tumor-induced osteomalacia: clinical and basic studies. J Bone Miner Res 12:1502–1511
Quarles LD (2003) Evidence for a bone–kidney axis regulating phosphate homeostasis. J Clin Invest 112:642–646
Carpenter TO, Ellis BK, Insogna KL, Philbrick WM, Sterpka J, Shimkets R (2005) Fibroblast growth factor 7: an inhibitor of phosphate transport derived from oncogenic osteomalacia-causing tumors. J Clin Endocrinol Metab 90:1012–1020
Habra MA, Jimenez C, Huang SC, Cote GJ, Murphy WA Jr, Gagel RF, Hoff AO (2008) Expression analysis of fibroblast growth factor-23, matrix extracellular phosphoglycoprotein, secreted frizzled-related protein-4, and fibroblast growth factor-7: identification of fibroblast growth factor-23 and matrix extracellular phosphoglycoprotein as major factors involved in tumor-induced osteomalacia. Endocr Pract 14:1108–1114
Hesse E, Rosenthal H, Bastian L (2007) Radiofrequency ablation of a tumor causing oncogenic osteomalacia. N Engl J Med 357:422–424
Jadhav S, Kasaliwal R, Shetty NS, Kulkarni S, Rathod K, Popat B, Kakade H, Bukan A, Khare S, Budyal S, Jagtap VS, Lila AR, Bandgar T, Shah NS (2014) Radiofrequency ablation, an effective modality of treatment in tumor-induced osteomalacia: a case series of three patients. J Clin Endocrinol Metab 99:3049–3054
Folpe AL, Fanburg-Smith JC, Billings SD, Bisceglia M, Bertoni F et al (2004) Most osteomalacia-associated mesenchymal tumors are a single histopathologic entity: an analysis of 32 cases and a comprehensive review of the literature. Am J Surg Pathol 28:1–30
Acknowledgments
We thank all of the patients and healthy individuals for participating in this study. The study was supported by the National Basic Research Program of China (2014CB942903), the National Natural Science Foundation of China (81370978 to Zhen-Lin Zhang, 81200646 to J.M. Gu, 8127096 to Chun Wang), the Shanghai Leading Talents Award (051) to Zhen-Lin Zhang, and the Chongqing Municipal Science and Technology Commission (CSTC2013jcyjC00009 to Zhen-Lin Zhang).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
About this article
Cite this article
Yu, WJ., He, JW., Fu, WZ. et al. Reports of 17 Chinese patients with tumor-induced osteomalacia. J Bone Miner Metab 35, 298–307 (2017). https://doi.org/10.1007/s00774-016-0756-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-016-0756-9