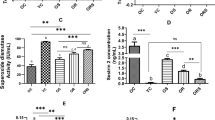
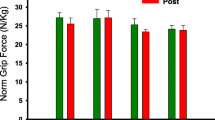
Abstract
Taurine (2-aminoethanesulfonic acid) is a free amino acid found abundantly in mammalian tissues. Increasing evidence suggests that taurine plays a role in the maintenance of skeletal muscle function and increase of exercise capacity. Most energy drinks contain this amino acid; however, there is insufficient research on the effects of long-term, low-dose supplementation of taurine. In this study, we investigated the effects of long-term administration of taurine at low doses on aging in rodents. In Experiment 1, we examined age-related changes in aging Sprague–Dawley (SD) rats (32–92 weeks old) that O2 consumption and spontaneous activity decreased significantly with aging. In Experiment 2, we examined the effects of long-term (21-week) administration of taurine on healthy aging SD rats. SD rats were stabilized for 32–34 weeks and divided into three groups, administrated water (control), 0.5% taurine (25 mg/kg body weight (BW)/day), or 1% taurine (50 mg/kg BW/day) from age 34 to 56 weeks (5 days/week, 5 mL/kg BW). Our findings suggest that long-term administration of taurine at relatively low dose could attenuate the age-related decline in O2 consumption and spontaneous locomotor activity. Upon intestinal absorption, taurine might modulate age-related changes in respiratory metabolism and skeletal muscle function via peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), succinate dehydrogenase (SDH), cytochrome c (Cycs), myocyte enhancer factor 2A (MEF2A), glucose transporter 4 (GLUT4), and myoglobin, which are regulated by the activation of AMP-activated protein kinase (AMPK). This article examines the mechanism underlying the effects of taurine on age-related changes, which may have potential clinical implications.









Similar content being viewed by others
Code availability
Not applicable.
References
Anderson R, Prolla T (2009) PGC-1alpha in aging and anti-aging interventions. Biochem Biophys Acta 1790:1059–1066. https://doi.org/10.1016/j.bbagen.2009.04.005
Azuma J, Sawamura A, Awata N, Ohta H, Hamaguchi T, Harada H, Takihara K et al (1985) Therapeutic effect of taurine in congestive heart failure: a double-blind crossover trial. Clin Cardiol 8:276–282. https://doi.org/10.1002/clc.4960080507
Batista TM, Ribeiro RA, da Silva PM, Camargo RL, Lollo PC, Boschero AC, Carneiro EM (2013) Taurine supplementation improves liver glucose control in normal protein and malnourished mice fed a high-fat diet. Mol Nutr Food Res 57:423–434. https://doi.org/10.1002/mnfr.201200345
Buskirk ER, Hodgson JL (1987) Age and aerobic power: the rate of change in men and women. Fed Proc 46:1824–1829
Cai J, Yang J, Jones DP (1998) Mitochondrial control of apoptosis: the role of cytochrome c. Biochem Biophys Acta 1366:139–149. https://doi.org/10.1016/s0005-2728(98)00109-1
Carling D (2004) The AMP-activated protein kinase cascade—a unifying system for energy control. Trends Biochem Sci 29:18–24. https://doi.org/10.1016/j.tibs.2003.11.005
Cheong SH, Chang KJ (2013) Antidiabetic effect of taurine in cultured rat skeletal l6 myotubes. Adv Exp Med Biol 775:311–320. https://doi.org/10.1007/978-1-4614-6130-2_26
Cooper MP, Uldry M, Kajimura S, Arany Z, Spiegelman BM (2008) Modulation of PGC-1 coactivator pathways in brown fat differentiation through LRP130. J Biol Chem 283:31960–31967. https://doi.org/10.1074/jbc.M805431200
Dawson R Jr, Liu S, Eppler B, Patterson T (1999) Effects of dietary taurine supplementation or deprivation in aged male Fischer 344 rats. Mech Ageing Dev 107:73–91. https://doi.org/10.1016/s0047-6374(98)00138-9
Dawson R Jr, Biasetti M, Messina S, Dominy J (2002) The cytoprotective role of taurine in exercise-induced muscle injury. Amino Acids 22:309–324. https://doi.org/10.1007/s007260200017
De Luca A, Pierno S, Camerino DC (2015) Taurine: the appeal of a safe amino acid for skeletal muscle disorders. J Transl Med 13:243. https://doi.org/10.1186/s12967-015-0610-1
Dunn-Lewis C, Kraemer WJ, Kupchak BR, Kelly NA, Creighton BA, Luk HY, Ballard KD et al (2011) A multi-nutrient supplement reduced markers of inflammation and improved physical performance in active individuals of middle to older age: a randomized, double-blind, placebo-controlled study. Nutr J 10:90. https://doi.org/10.1186/1475-2891-10-90
Ermini M, Szelényi I, Moser P, Verzár F (1971) The ageing of skeletal (striated) muscle by changes of recovery metabolism. Gerontology 17:300–311. https://doi.org/10.1159/000211836
Esterbauer H, Oberkofler H, Krempler F, Patsch W (1999) Human peroxisome proliferator activated receptor gamma coactivator 1 (PPARGC1) gene: cDNA sequence, genomic organization, chromosomal localization, and tissue expression. Genomics 62:98–102. https://doi.org/10.1006/geno.1999.5977
Faitg J, Reynaud O, Leduc-Gaudet JP, Gouspillou G (2017) Skeletal muscle aging and mitochondrial dysfunction: an update. Med Sci M/S 33:955–962. https://doi.org/10.1051/medsci/20173311012
Ferreira IMPLVO, Nunes MV, Mendes E, Remião F, Ferreira MA (1997) Development of an HPLC-UV method for determination of taurine in infant formulae and breast milk. J Liq Chromatogr Relat Technol 20:1269–1278. https://doi.org/10.1080/10826079708010975
Finck BN, Kelly DP (2006) PGC-1 coactivators: inducible regulators of energy metabolism in health and disease. J Clin Investig 116:615–622. https://doi.org/10.1172/jci27794
Fontana L (2009) Modulating human aging and age-associated diseases. Biochem Biophys Acta 1790:1133–1138. https://doi.org/10.1016/j.bbagen.2009.02.002
Franconi F, Bennardini F, Mattana A, Miceli M, Ciuti M, Mian M, Gironi A et al (1995) Plasma and platelet taurine are reduced in subjects with insulin-dependent diabetes mellitus: effects of taurine supplementation. Am J Clin Nutr 61:1115–1119. https://doi.org/10.1093/ajcn/61.4.1115
Gastaldelli A, Kozakova M, Højlund K, Flyvbjerg A, Favuzzi A, Mitrakou A, Balkau B (2009) Fatty liver is associated with insulin resistance, risk of coronary heart disease, and early atherosclerosis in a large European population. Hepatology (Baltimore, MD) 49:1537–1544. https://doi.org/10.1002/hep.22845
Giles GE, Mahoney CR, Brunyé TT, Gardony AL, Taylor HA, Kanarek RB (2012) Differential cognitive effects of energy drink ingredients: caffeine, taurine, and glucose. Pharmacol Biochem Behav 102:569–577. https://doi.org/10.1016/j.pbb.2012.07.004
Handschin C, Spiegelman BM (2006) Peroxisome proliferator-activated receptor gamma coactivator 1 coactivators, energy homeostasis, and metabolism. Endocr Rev 27:728–735. https://doi.org/10.1210/er.2006-0037
Handschin C, Rhee J, Lin J, Tarr PT, Spiegelman BM (2003) An autoregulatory loop controls peroxisome proliferator-activated receptor gamma coactivator 1alpha expression in muscle. Proc Natl Acad Sci USA 100:7111–7116. https://doi.org/10.1073/pnas.1232352100
Hardie DG (2004) The AMP-activated protein kinase pathway—new players upstream and downstream. J Cell Sci 117:5479–5487. https://doi.org/10.1242/jcs.01540
Heath GW, Hagberg JM, Ehsani AA, Holloszy JO (1981) A physiological comparison of young and older endurance athletes. J Appl Physiol Respir Environ Exerc Physiol 51:634–640. https://doi.org/10.1152/jappl.1981.51.3.634
Hepple RT (2016) Impact of aging on mitochondrial function in cardiac and skeletal muscle. Free Radic Biol Med 98:177–186. https://doi.org/10.1016/j.freeradbiomed.2016.03.017
Holmes BF, Sparling DP, Olson AL, Winder WW, Dohm GL (2005) Regulation of muscle GLUT4 enhancer factor and myocyte enhancer factor 2 by AMP-activated protein kinase. Am J Physiol Endocrinol Metab 289:E1071-1076. https://doi.org/10.1152/ajpendo.00606.2004
Huxtable RJ (1992) Physiological actions of taurine. Physiol Rev 72:101–163. https://doi.org/10.1152/physrev.1992.72.1.101
Irrcher I, Ljubicic V, Kirwan AF, Hood DA (2008) AMP-activated protein kinase-regulated activation of the PGC-1alpha promoter in skeletal muscle cells. PLoS ONE 3:e3614. https://doi.org/10.1371/journal.pone.0003614
Ito T, Schaffer SW, Azuma J (2012) The potential usefulness of taurine on diabetes mellitus and its complications. Amino Acids 42:1529–1539. https://doi.org/10.1007/s00726-011-0883-5
Ito T, Yoshikawa N, Inui T, Miyazaki N, Schaffer SW, Azuma J (2014) Tissue depletion of taurine accelerates skeletal muscle senescence and leads to early death in mice. PLoS ONE 9:e107409. https://doi.org/10.1371/journal.pone.0107409
Kang C, Chung E, Diffee G, Ji LL (2013) Exercise training attenuates aging-associated mitochondrial dysfunction in rat skeletal muscle: role of PGC-1α. Exp Gerontol 48:1343–1350. https://doi.org/10.1016/j.exger.2013.08.004
Kaplan B, Karabay G, Zağyapan RD, Ozer C, Sayan H, Duyar I (2004) Effects of taurine in glucose and taurine administration. Amino Acids 27:327–333. https://doi.org/10.1007/s00726-003-0063-3
Kemp BE, Stapleton D, Campbell DJ, Chen ZP, Murthy S, Walter M, Gupta A et al (2003) AMP-activated protein kinase, super metabolic regulator. Biochem Soc Trans 31:162–168. https://doi.org/10.1042/bst0310162
Kirkendall DT, Garrett WE (1998) The effects of aging and training on skeletal muscle. Am J Sports Med 26:598–602. https://doi.org/10.1177/03635465980260042401
Lantier L, Fentz J, Mounier R, Leclerc J, Treebak JT, Pehmøller C, Sanz N et al (2014) AMPK controls exercise endurance, mitochondrial oxidative capacity, and skeletal muscle integrity. FASEB J 28:3211–3224. https://doi.org/10.1096/fj.14-250449
Liu HH, Li JJ (2015) Aging and dyslipidemia: a review of potential mechanisms. Ageing Research Rev 19:43–52. https://doi.org/10.1016/j.arr.2014.12.001
Long YC, Zierath JR (2006) AMP-activated protein kinase signaling in metabolic regulation. J Clin Investig 116:1776–1783. https://doi.org/10.1172/jci29044
Lubec B, Ya-hua Z, Pertti S, Pentti T, Kitzmüller E, Lubec G (1997) Distribution and disappearance of the radiolabeled carbon derived from l-arginine and taurine in the mouse. Life Sci 60:2373–2381. https://doi.org/10.1016/s0024-3205(97)00297-x
Mangum JE, Hardee JP, Fix DK, Puppa MJ, Elkes J, Altomare D, Bykhovskaya Y et al (2016) Pseudouridine synthase 1 deficient mice, a model for mitochondrial myopathy with sideroblastic anemia, exhibit muscle morphology and physiology alterations. Sci Rep 6:26202. https://doi.org/10.1038/srep26202
Maruta H, Yamashita H (2020) Acetic acid stimulates G-protein-coupled receptor GPR43 and induces intracellular calcium influx in L6 myotube cells. PLoS ONE 15:e0239428. https://doi.org/10.1371/journal.pone.0239428
Maruta H, Yoshimura Y, Araki A, Kimoto M, Takahashi Y, Yamashita H (2016) Activation of AMP-activated protein kinase and stimulation of energy metabolism by acetic acid in l6 myotube cells. PLoS ONE 11:e0158055. https://doi.org/10.1371/journal.pone.0158055
Masoro EJ (2001) Physiology of aging. Int J Sport Nutr Exerc Metab 11:S218. https://doi.org/10.1123/ijsnem.11.s1.s218
McGee SL, van Denderen BJ, Howlett KF, Mollica J, Schertzer JD, Kemp BE, Hargreaves M (2008) AMP-activated protein kinase regulates GLUT4 transcription by phosphorylating histone deacetylase 5. Diabetes 57:860–867. https://doi.org/10.2337/db07-0843
Mensink M, Hesselink MK, Russell AP, Schaart G, Sels JP, Schrauwen P (2007) Improved skeletal muscle oxidative enzyme activity and restoration of PGC-1 alpha and PPAR beta/delta gene expression upon rosiglitazone treatment in obese patients with type 2 diabetes mellitus. Int J Obes (2005) 31:1302–1310. https://doi.org/10.1038/sj.ijo.0803567
Miyazaki T, Matsuzaki Y, Ikegami T, Miyakawa S, Doy M, Tanaka N, Bouscarel B (2004) Optimal and effective oral dose of taurine to prolong exercise performance in rat. Amino Acids 27:291–298. https://doi.org/10.1007/s00726-004-0133-1
Ogawa T, Spina RJ, Martin WH 3rd, Kohrt WM, Schechtman KB, Holloszy JO, Ehsani AA (1992) Effects of aging, sex, and physical training on cardiovascular responses to exercise. Circulation 86:494–503. https://doi.org/10.1161/01.cir.86.2.494
O’Neill HM (2013) AMPK and exercise: glucose uptake and insulin sensitivity. Diabetes Metab J 37:1–21. https://doi.org/10.4093/dmj.2013.37.1.1
O’Neill HM, Holloway GP, Steinberg GR (2013) AMPK regulation of fatty acid metabolism and mitochondrial biogenesis: implications for obesity. Mol Cell Endocrinol 366:135–151. https://doi.org/10.1016/j.mce.2012.06.019
Pasantes-Morales H, Quesada O, Alcocer L, Olea RS (1989) Taurine content in foods. Nutr Rep Int 40:793–801
Peacock A, Martin FH, Carr A (2013) Energy drink ingredients. Contribution of caffeine and taurine to performance outcomes. Appetite 64:1–4. https://doi.org/10.1016/j.appet.2012.12.021
Pierno S, De Luca A, Camerino C, Huxtable RJ, Camerino DC (1998) Chronic administration of taurine to aged rats improves the electrical and contractile properties of skeletal muscle fibers. J Pharmacol Exp Ther 286:1183–1190
Pilegaard H, Saltin B, Neufer PD (2003) Exercise induces transient transcriptional activation of the PGC-1alpha gene in human skeletal muscle. J Physiol 546:851–858. https://doi.org/10.1113/jphysiol.2002.034850
Richter EA, Hargreaves M (2013) Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol Rev 93:993–1017. https://doi.org/10.1152/physrev.00038.2012
Rivera AM, Pels AE 3rd, Sady SP, Sady MA, Cullinane EM, Thompson PD (1989) Physiological factors associated with the lower maximal oxygen consumption of master runners. J Appl Physiol (Bethesda, Md: 1985) 66:949–954. https://doi.org/10.1152/jappl.1989.66.2.949
Salminen A, Kaarniranta K (2012) AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res Rev 11:230–241. https://doi.org/10.1016/j.arr.2011.12.005
Schaffer SW, Jong CJ, Ramila KC, Azuma J (2010) Physiological roles of taurine in heart and muscle. J Biomed Sci 17(Suppl 1):S2. https://doi.org/10.1186/1423-0127-17-s1-s2
Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS (2005) Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci USA 102:5618–5623. https://doi.org/10.1073/pnas.0501559102
Spinazzi M, Casarin A, Pertegato V, Salviati L, Angelini C (2012) Assessment of mitochondrial respiratory chain enzymatic activities on tissues and cultured cells. Nat Protoc 7:1235–1246. https://doi.org/10.1038/nprot.2012.058
Stacchiotti A, Favero G, Lavazza A, Monsalve M, Rodella LF, Rezzani R (2018) Taurine supplementation alleviates puromycin aminonucleoside damage by modulating endoplasmic reticulum stress and mitochondrial-related apoptosis in rat kidney. Nutrients. https://doi.org/10.3390/nu10060689
Sved DW, Godsey JL, Ledyard SL, Mahoney AP, Stetson PL, Ho S, Myers NR, Resnis P, Renwick G (2007) Absorption, tissue distribution, metabolism and elimination of taurine given orally to rats. Amino Acids 32:459–466
Takahashi Y, Urushibata E, Hatta H (2013) Higher voluntary wheel running activity following endurance exercise due to oral taurine administration in mice. J Phys Fit Sports Med 2:373–379. https://doi.org/10.7600/jpfsm.2.373
Terada S, Goto M, Kato M, Kawanaka K, Shimokawa T, Tabata I (2002) Effects of low-intensity prolonged exercise on PGC-1 mRNA expression in rat epitrochlearis muscle. Biochem Biophys Res Commun 296:350–354. https://doi.org/10.1016/s0006-291x(02)00881-1
Terrill JR, Grounds MD, Arthur PG (2015) Taurine deficiency, synthesis and transport in the mdx mouse model for Duchenne Muscular Dystrophy. Int J Biochem Cell Biol 66:141–148. https://doi.org/10.1016/j.biocel.2015.07.016
Terrlink T, van Leeuwen PA, Houdijk A (1994) Plasma amino acids determined by liquid chromatography within 17 minutes. Clin Chem 40:245–249. https://doi.org/10.1093/clinchem/40.2.245
Trounce I, Byrne E, Marzuki S (1989) Decline in skeletal muscle mitochondrial respiratory chain function: possible factor in ageing. Lancet (London, England) 1:637–639. https://doi.org/10.1016/s0140-6736(89)92143-0
Warskulat U, Flögel U, Jacoby C, Hartwig HG, Thewissen M, Merx MW, Molojavyi A et al (2004) Taurine transporter knockout depletes muscle taurine levels and results in severe skeletal muscle impairment but leaves cardiac function uncompromised. FASEB J 18:577–579. https://doi.org/10.1096/fj.03-0496fje
Waterfield CJ (1994) Determination of taurine in biological samples and isolated hepatocytes by high-performance liquid chromatography with fluorimetric detection. J Chromatogr B Biomed Appl 657:37–45. https://doi.org/10.1016/0378-4347(94)80067-7
White JP, Baltgalvis KA, Puppa MJ, Sato S, Baynes JW, Carson JA (2011) Muscle oxidative capacity during IL-6-dependent cancer cachexia. Am J Physiol Regul Integr Comp Physiol 300:R201-211. https://doi.org/10.1152/ajpregu.00300.2010
Willig F, Palacios A, Monmaur P, M’Harzi M, Laurent J, Delacour J (1987) Short-term memory, exploration and locomotor activity in aged rats. Neurobiol Aging 8:393–402. https://doi.org/10.1016/0197-4580(87)90033-9
Winder WW, Holmes BF, Rubink DS, Jensen EB, Chen M, Holloszy JO (2000) Activation of AMP-activated protein kinase increases mitochondrial enzymes in skeletal muscle. J Appl Physiol (Bethesda, Md: 1985) 88:2219–2226. https://doi.org/10.1152/jappl.2000.88.6.2219
Yatabe Y, Miyakawa S, Miyazaki T, Matsuzaki Y, Ochiai N (2003) Effects of taurine administration in rat skeletal muscles on exercise. J Orthop Sci 8:415–419. https://doi.org/10.1007/s10776-002-0636-1
Yildirim Z, Kiliç N, Ozer C, Babul A, Take G, Erdogan D (2007) Effects of taurine in cellular responses to oxidative stress in young and middle-aged rat liver. Ann N Y Acad Sci 1100:553–561. https://doi.org/10.1196/annals.1395.061
Zhao L, Zou X, Feng Z, Luo C, Liu J, Li H, Chang L et al (2014) Evidence for association of mitochondrial metabolism alteration with lipid accumulation in aging rats. Exp Gerontol 56:3–12. https://doi.org/10.1016/j.exger.2014.02.001
Acknowledgements
We would like to thank all members of the present study group for their ideas, suggestions, participation and support. This work was supported by TOKUBETSU KENKYUHI of Okayama Prefectural University.
Funding
This work was supported by TOKUBETSU KENKYUHI of Okayama Prefectural University.
Author information
Authors and Affiliations
Contributions
YM, BS, HM, CI, and HY conceived, designed, and performed the experiments; YM, BS, HM, and HY analyzed the data; HY organized the funding. YM, HM, and HY wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All animal experiments were conducted in accordance with the guidelines of Okayama Prefectural University and the laws and notifications of the Japanese government. All animal experiments were approved by the Animal Care and Use Committee of Okayama Prefectural University (protocol number 31-5).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Additional information
Handling editor: S. W. Schaffer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ma, Y., Maruta, H., Sun, B. et al. Effects of long-term taurine supplementation on age-related changes in skeletal muscle function of Sprague–Dawley rats. Amino Acids 53, 159–170 (2021). https://doi.org/10.1007/s00726-020-02934-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-020-02934-0