Abstract
Background
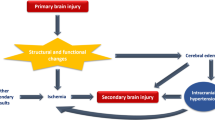
Aneurysmal subarachnoid haemorrhage is associated with significant morbidity and mortality due to the myriad of complications contributing to early brain injury and delayed cerebral ischaemia. There is increasing interest in the exploration of the association between blood-brain barrier integrity and risks of delayed cerebral ischaemia and poor outcomes. Despite recent advances in cerebral imaging, radiographic imaging of blood-brain barrier disruption, as a biomarker for outcome prediction, has not been adopted in clinical practice.
Methods
We performed a narrative review by searching for articles describing molecular changes or radiological identification of changes in BBB permeability following subarachnoid haemorrhage (SAH) on MEDLINE. Preclinical studies were analysed if reported structural changes and clinical studies were included if they investigated for radiological markers of BBB disruption and its correlation with delayed cerebral ischaemia.
Results
There is ample preclinical evidence to suggest that there are structural changes in BBB permeability following SAH. The available clinical literature has demonstrated correlations between permeability imaging and outcomes following aneurysmal subarachnoid haemorrhage (aSAH).
Conclusion
Radiological biomarkers offer a potential non-invasive prognostication tool and may also allow early identifications of patients who may be at risk of DCI.




Similar content being viewed by others
References
Alluri H, Wiggins-Dohlvik K, Davis ML, Huang JH, Tharakan B (2015) Blood-brain barrier dysfunction following traumatic brain injury. Metab Brain Dis 30:1093–1104. https://doi.org/10.1007/s11011-015-9651-7
Barzo P, Marmarou A, Fatouros P, Corwin F, Dunbar J (1996) Magnetic resonance imaging-monitored acute blood-brain barrier changes in experimental traumatic brain injury. J Neurosurg 85:1113–1121. https://doi.org/10.3171/jns.1996.85.6.1113
Bederson JB, Connolly ES Jr, Batjer HH, Dacey RG, Dion JE, Diringer MN, Duldner JE Jr, Harbaugh RE, Patel AB, Rosenwasser RH (2009) Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke 40:994–1025. https://doi.org/10.1161/strokeaha.108.191395
Blecharz-Lang KG, Wagner J, Fries A, Nieminen-Kelha M, Rosner J, Schneider UC, Vajkoczy P (2018) Interleukin 6-mediated endothelial barrier disturbances can be attenuated by blockade of the IL6 receptor expressed in brain microvascular endothelial cells. Transl Stroke Res 9:631–642. https://doi.org/10.1007/s12975-018-0614-2
Cao S, Zhu P, Yu X, Chen J, Li J, Yan F, Wang L, Yu J, Chen G (2016) Hydrogen sulfide attenuates brain edema in early brain injury after subarachnoid hemorrhage in rats: Possible involvement of MMP-9 induced blood-brain barrier disruption and AQP4 expression. Neurosci Lett 621:88–97. https://doi.org/10.1016/j.neulet.2016.04.018
Chassidim Y, Vazana U, Prager O, Veksler R, Bar-Klein G, Schoknecht K, Fassler M, Lublinsky S, Shelef I (2015) Analyzing the blood-brain barrier: the benefits of medical imaging in research and clinical practice. Semin Cell Dev Biol 38:43–52. https://doi.org/10.1016/j.semcdb.2014.11.007
Chassidim Y, Veksler R, Lublinsky S, Pell GS, Friedman A, Shelef I (2013) Quantitative imaging assessment of blood-brain barrier permeability in humans. Fluids Barriers CNS 10:9. https://doi.org/10.1186/2045-8118-10-9
Chen D, Wei XT, Guan JH, Yuan JW, Peng YT, Song L, Liu YH (2012) Inhibition of c-Jun N-terminal kinase prevents blood-brain barrier disruption and normalizes the expression of tight junction proteins clautin-5 and ZO-1 in a rat model of subarachnoid hemorrhage. Acta Neurochir (Wien) 154:1469–1476; discussion 1476. https://doi.org/10.1007/s00701-012-1328-y
Chen F, Hori T, Ohashi N, Baine AM, Eckman CB, Nguyen JH (2011) Occludin is regulated by epidermal growth factor receptor activation in brain endothelial cells and brains of mice with acute liver failure. Hepatology 53:1294–1305. https://doi.org/10.1002/hep.24161
Chen J, Chen G, Li J, Qian C, Mo H, Gu C, Yan F, Yan W, Wang L (2014) Melatonin attenuates inflammatory response-induced brain edema in early brain injury following a subarachnoid hemorrhage: a possible role for the regulation of pro-inflammatory cytokines. J Pineal Res 57:340–347. https://doi.org/10.1111/jpi.12173
Chen J, Jin H, Xu H, Peng Y, Jie L, Xu D, Chen L, Li T, Fan L, He P, Ying G, Gu C, Wang C, Wang L, Chen G (2019) The Neuroprotective effects of necrostatin-1 on subarachnoid hemorrhage in rats are possibly mediated by preventing blood-brain barrier disruption and RIP3-mediated necroptosis. Cell Transplant 28:1358–1372. https://doi.org/10.1177/0963689719867285
Chen J, Qian C, Duan H, Cao S, Yu X, Li J, Gu C, Yan F, Wang L, Chen G (2015) Melatonin attenuates neurogenic pulmonary edema via the regulation of inflammation and apoptosis after subarachnoid hemorrhage in rats. J Pineal Res 59:469–477. https://doi.org/10.1111/jpi.12278
Chen T, Wang W, Li JR, Xu HZ, Peng YC, Fan LF, Yan F, Gu C, Wang L, Chen G (2016) PARP inhibition attenuates early brain injury through NF-kappaB/MMP-9 pathway in a rat model of subarachnoid hemorrhage. Brain Res 1644:32–38. https://doi.org/10.1016/j.brainres.2016.05.005
Chiba H, Osanai M, Murata M, Kojima T, Sawada N (2008) Transmembrane proteins of tight junctions. Biochim Biophys Acta 1778:588–600. https://doi.org/10.1016/j.bbamem.2007.08.017
Claassen J, Bernardini GL, Kreiter K, Bates J, Du YE, Copeland D, Connolly ES, Mayer SA (2001) Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid hemorrhage: the Fisher scale revisited. Stroke 32:2012–2020. https://doi.org/10.1161/hs0901.095677
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA (2002) Global cerebral edema after subarachnoid hemorrhage: frequency, predictors, and impact on outcome. Stroke 33:1225–1232. https://doi.org/10.1161/01.str.0000015624.29071.1f
Daneman R, Zhou L, Kebede AA, Barres BA (2010) Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature 468:562–566. https://doi.org/10.1038/nature09513
Dankbaar JW, Rijsdijk M, van der Schaaf IC, Velthuis BK, Wermer MJ, Rinkel GJ (2009) Relationship between vasospasm, cerebral perfusion, and delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Neuroradiology 51:813–819. https://doi.org/10.1007/s00234-009-0575-y
Doczi T (1985) The pathogenetic and prognostic significance of blood-brain barrier damage at the acute stage of aneurysmal subarachnoid haemorrhage. Clinical and experimental studies. Acta Neurochir (Wien) 77:110–132. https://doi.org/10.1007/bf01476215
Doczi T, Ambrose J, O'Laoire S (1984) Significance of contrast enhancement in cranial computerized tomography after subarachnoid hemorrhage. J Neurosurg 60:335–342. https://doi.org/10.3171/jns.1984.60.2.0335
Doherty CP, O'Keefe E, Wallace E, Loftus T, Keaney J, Kealy J, Humphries MM, Molloy MG, Meaney JF, Farrell M, Campbell M (2016) Blood-brain barrier dysfunction as a hallmark pathology in chronic traumatic encephalopathy. J Neuropathol Exp Neurol 75:656–662. https://doi.org/10.1093/jnen/nlw036
Dorhout Mees SM, Rinkel GJ, Feigin VL, Algra A, van den Bergh WM, Vermeulen M, van Gijn J (2007) Calcium antagonists for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev:Cd000277. https://doi.org/10.1002/14651858.CD000277.pub3
Dreier JP (2011) The role of spreading depression, spreading depolarization and spreading ischemia in neurological disease. Nat Med 17:439–447. https://doi.org/10.1038/nm.2333
Fan LF, He PY, Peng YC, Du QH, Ma YJ, Jin JX, Xu HZ, Li JR, Wang ZJ, Cao SL, Li T, Yan F, Gu C, Wang L, Chen G (2017) Mdivi-1 ameliorates early brain injury after subarachnoid hemorrhage via the suppression of inflammation-related blood-brain barrier disruption and endoplasmic reticulum stress-based apoptosis. Free Radic Biol Med 112:336–349. https://doi.org/10.1016/j.freeradbiomed.2017.08.003
Fang R, Zheng X, Zhang M (2016) Ethyl pyruvate alleviates early brain injury following subarachnoid hemorrhage in rats. Acta Neurochir (Wien) 158:1069–1076. https://doi.org/10.1007/s00701-016-2795-3
Friedrich V, Flores R, Muller A, Sehba FA (2010) Escape of intraluminal platelets into brain parenchyma after subarachnoid hemorrhage. Neuroscience 165:968–975. https://doi.org/10.1016/j.neuroscience.2009.10.038
Fujii M, Duris K, Altay O, Soejima Y, Sherchan P, Zhang JH (2012) Inhibition of Rho kinase by hydroxyfasudil attenuates brain edema after subarachnoid hemorrhage in rats. Neurochem Int 60:327–333. https://doi.org/10.1016/j.neuint.2011.12.014
Fujii M, Sherchan P, Soejima Y, Doycheva D, Zhao D, Zhang JH (2016) Cannabinoid receptor type 2 agonist attenuates acute neurogenic pulmonary edema by preventing neutrophil migration after subarachnoid hemorrhage in rats. Acta Neurochir (Wien) Suppl 121:135–139. https://doi.org/10.1007/978-3-319-18497-5_24
Han YW, Liu XJ, Zhao Y, Li XM (2018) Role of Oleanolic acid in maintaining BBB integrity by targeting p38MAPK/VEGF/Src signaling pathway in rat model of subarachnoid hemorrhage. Eur J Pharmacol 839:12–20. https://doi.org/10.1016/j.ejphar.2018.09.018
Hasegawa Y, Suzuki H, Altay O, Chen H, Zhang JH (2012) Treatment with sodium orthovanadate reduces blood-brain barrier disruption via phosphatase and tensin homolog deleted on chromosome 10 (PTEN) phosphorylation in experimental subarachnoid hemorrhage. J Neurosci Res 90:691–697. https://doi.org/10.1002/jnr.22801
Heye AK, Culling RD, Valdes Hernandez Mdel C, Thrippleton MJ, Wardlaw JM (2014) Assessment of blood-brain barrier disruption using dynamic contrast-enhanced MRI. A systematic review. Neuroimage Clin 6:262–274. https://doi.org/10.1016/j.nicl.2014.09.002
Hu HM, Li B, Wang XD, Guo YS, Hui H, Zhang HP, Wang B, Huang DG, Hao DJ (2018) Fluoxetine is neuroprotective in early brain injury via its anti-inflammatory and anti-apoptotic effects in a rat experimental subarachnoid hemorrhage model. Neurosci Bull 34:951–962. https://doi.org/10.1007/s12264-018-0232-8
Ishiguro M, Mishiro K, Fujiwara Y, Chen H, Izuta H, Tsuruma K, Shimazawa M, Yoshimura S, Satoh M, Iwama T, Hara H (2010) Phosphodiesterase-III inhibitor prevents hemorrhagic transformation induced by focal cerebral ischemia in mice treated with tPA. PLoS One 5:e15178. https://doi.org/10.1371/journal.pone.0015178
Ivanidze J, Ferraro RA, Giambrone AE, Segal AZ, Gupta A, Sanelli PC (2018) Blood-brain barrier permeability in aneurysmal subarachnoid hemorrhage: correlation with clinical outcomes. AJR Am J Roentgenol 211:891–895. https://doi.org/10.2214/ajr.17.18237
Ivanidze J, Kallas ON, Gupta A, Weidman E, Baradaran H, Mir D, Giambrone A, Segal AZ, Claassen J, Sanelli PC (2016) Application of Blood-brain barrier permeability imaging in global cerebral edema. AJNR Am J Neuroradiol 37:1599–1603. https://doi.org/10.3174/ajnr.A4784
Ivanidze J, Kesavabhotla K, Kallas ON, Mir D, Baradaran H, Gupta A, Segal AZ, Claassen J, Sanelli PC (2015) Evaluating blood-brain barrier permeability in delayed cerebral infarction after aneurysmal subarachnoid hemorrhage. AJNR Am J Neuroradiol 36:850–854. https://doi.org/10.3174/ajnr.A4207
Kebir H, Kreymborg K, Ifergan I, Dodelet-Devillers A, Cayrol R, Bernard M, Giuliani F, Arbour N, Becher B, Prat A (2007) Human TH17 lymphocytes promote blood-brain barrier disruption and central nervous system inflammation. Nat Med 13:1173–1175. https://doi.org/10.1038/nm1651
Kishore S, Ko N, Soares BP, Higashida RT, Tong E, Bhogal S, Bredno J, Cheng SC, Wintermark M (2012) Perfusion-CT assessment of blood-brain barrier permeability in patients with aneurysmal subarachnoid hemorrhage. J Neuroradiol 39:317–325. https://doi.org/10.1016/j.neurad.2011.11.004
Kreiter KT, Copeland D, Bernardini GL, Bates JE, Peery S, Claassen J, Du YE, Stern Y, Connolly ES, Mayer SA (2002) Predictors of cognitive dysfunction after subarachnoid hemorrhage. Stroke 33:200–208. https://doi.org/10.1161/hs0102.101080
Li Z, Liang G, Ma T, Li J, Wang P, Liu L, Yu B, Liu Y, Xue Y (2015) Blood-brain barrier permeability change and regulation mechanism after subarachnoid hemorrhage. Metab Brain Dis 30:597–603. https://doi.org/10.1007/s11011-014-9609-1
Lin Z, Li Y, Su P, Mao D, Wei Z, Pillai JJ, Moghekar A, van Osch M, Ge Y, Lu H (2018) Non-contrast MR imaging of blood-brain barrier permeability to water. Magn Reson Med 80:1507–1520. https://doi.org/10.1002/mrm.27141
Liu X, Zhang X, Ma K, Zhang R, Hou P, Sun B, Yuan S, Wang Z, Liu Z (2016) Matrine alleviates early brain injury after experimental subarachnoid hemorrhage in rats: possible involvement of PI3K/Akt-mediated NF-kappaB inhibition and Keap1/Nrf2-dependent HO-1 inductionn. Cell Mol Biol (Noisy-le-grand) 62:38–44
Lublinsky S, Major S, Kola V, Horst V, Santos E, Platz J, Sakowitz O, Scheel M, Dohmen C, Graf R, Vatter H, Wolf S, Vajkoczy P, Shelef I, Woitzik J, Martus P, Dreier JP, Friedman A (2019) Early blood-brain barrier dysfunction predicts neurological outcome following aneurysmal subarachnoid hemorrhage. EBioMedicine 43:460–472. https://doi.org/10.1016/j.ebiom.2019.04.054
Luh C, Feiler S, Frauenknecht K, Meyer S, Lubomirov LT, Neulen A, Thal SC (2019) The contractile apparatus is essential for the integrity of the blood-brain barrier after experimental subarachnoid hemorrhage. Transl Stroke Res 10:534–545. https://doi.org/10.1007/s12975-018-0677-0
Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, Vajkoczy P, Wanke I, Bach D, Frey A, Marr A, Roux S, Kassell N (2011) Clazosentan, an endothelin receptor antagonist, in patients with aneurysmal subarachnoid haemorrhage undergoing surgical clipping: a randomised, double-blind, placebo-controlled phase 3 trial (CONSCIOUS-2). Lancet Neurol 10:618–625. https://doi.org/10.1016/s1474-4422(11)70108-9
Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, Vajkoczy P, Wanke I, Bach D, Frey A, Nowbakht P, Roux S, Kassell N (2012) Randomized trial of clazosentan in patients with aneurysmal subarachnoid hemorrhage undergoing endovascular coiling. Stroke 43:1463–1469. https://doi.org/10.1161/strokeaha.111.648980
Macdonald RL, Kassell NF, Mayer S, Ruefenacht D, Schmiedek P, Weidauer S, Frey A, Roux S, Pasqualin A (2008) Clazosentan to overcome neurological ischemia and infarction occurring after subarachnoid hemorrhage (CONSCIOUS-1): randomized, double-blind, placebo-controlled phase 2 dose-finding trial. Stroke 39:3015–3021. https://doi.org/10.1161/strokeaha.108.519942
Murphy A, Manoel AL, Burgers K, Kouzmina E, Lee T, Macdonald RL, Bharatha A (2015) Early CT perfusion changes and blood-brain barrier permeability after aneurysmal subarachnoid hemorrhage. Neuroradiology 57:767–773. https://doi.org/10.1007/s00234-015-1529-1
Na DG, Kim EY, Ryoo JW, Lee KH, Roh HG, Kim SS, Song IC, Chang KH (2005) CT sign of brain swelling without concomitant parenchymal hypoattenuation: comparison with diffusion- and perfusion-weighted MR imaging. Radiology 235:992–948. https://doi.org/10.1148/radiol.2353040571
Nicholson P, O'Hare A, Power S, Looby S, Javadpour M, Thornton J, Brennan P (2019) Decreasing incidence of subarachnoid hemorrhage. J Neurointerv Surg 11:320–322. https://doi.org/10.1136/neurintsurg-2018-014038
Nitta T, Hata M, Gotoh S, Seo Y, Sasaki H, Hashimoto N, Furuse M, Tsukita S (2003) Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J Cell Biol 161:653–660. https://doi.org/10.1083/jcb.200302070
O'Keeffe E, Kelly E, Liu Y, Giordano C, Wallace E, Hynes M, Tiernan S, Meagher A, Greene C, Hughes S, Burke T, Kealy J, Doyle N, Hay A, Farrell M, Grant GA, Friedman A, Veksler R, Molloy MG, Meaney JF, Pender N, Camarillo D, Doherty CP, Campbell M (2020) Dynamic blood-brain barrier regulation in mild traumatic brain injury. J Neurotrauma 37:347–356. https://doi.org/10.1089/neu.2019.6483
Oldendorf WH, Cornford ME, Brown WJ (1977) The large apparent work capability of the blood-brain barrier: a study of the mitochondrial content of capillary endothelial cells in brain and other tissues of the rat. Ann Neurol 1:409–417. https://doi.org/10.1002/ana.410010502
Pan P, Zhang X, Li Q, Zhao H, Qu J, Zhang JH, Liu X, Feng H, Chen Y (2017) Cyclosporine A alleviated matrix metalloproteinase 9 associated blood-brain barrier disruption after subarachnoid hemorrhage in mice. Neurosci Lett 649:7–13. https://doi.org/10.1016/j.neulet.2017.03.050
Pan P, Zhao H, Zhang X, Li Q, Qu J, Zuo S, Yang F, Liang G, Zhang JH, Liu X, He H, Feng H, Chen Y (2020) Cyclophilin a signaling induces pericyte-associated blood-brain barrier disruption after subarachnoid hemorrhage. J Neuroinflammation 17:16. https://doi.org/10.1186/s12974-020-1699-6
Pang J, Chen Y, Kuai L, Yang P, Peng J, Wu Y, Chen Y, Vitek MP, Chen L, Sun X, Jiang Y (2017) Inhibition of Blood-brain barrier disruption by an apolipoprotein E-mimetic peptide ameliorates early brain injury in experimental subarachnoid hemorrhage. Transl Stroke Res 8:257–272. https://doi.org/10.1007/s12975-016-0507-1
Pickard JD, Murray GD, Illingworth R, Shaw MD, Teasdale GM, Foy PM, Humphrey PR, Lang DA, Nelson R, Richards P et al (1989) Effect of oral nimodipine on cerebral infarction and outcome after subarachnoid haemorrhage: British aneurysm nimodipine trial. Bmj 298:636–642. https://doi.org/10.1136/bmj.298.6674.636
Pradilla G, Chaichana KL, Hoang S, Huang J, Tamargo RJ (2010) Inflammation and cerebral vasospasm after subarachnoid hemorrhage. Neurosurg Clin N Am 21:365–379. https://doi.org/10.1016/j.nec.2009.10.008
Qian C, Jin J, Chen J, Li J, Yu X, Mo H, Chen G (2017) SIRT1 activation by resveratrol reduces brain edema and neuronal apoptosis in an experimental rat subarachnoid hemorrhage model. Mol Med Rep 16:9627–9635. https://doi.org/10.3892/mmr.2017.7773
Qian H, Dou Z, Ruan W, He P, Zhang JH, Yan F (2018) ErbB4 preserves blood-brain barrier integrity via the YAP/PIK3CB pathway after subarachnoid hemorrhage in rats. Front Neurosci 12:492. https://doi.org/10.3389/fnins.2018.00492
Rieth KG, Fujiwara K, Di Chiro G, Klatzo I, Brooks RA, Johnston GS, O'Connor CM, Mitchell LG (1980) Serial measurements of CT attenuation and specific gravity in experimental cerebral edema. Radiology 135:343–348. https://doi.org/10.1148/radiology.135.2.6768102
Russin JJ, Montagne A, D'Amore F, He S, Shiroishi MS, Rennert RC, Depetris J, Zlokovic BV, Mack WJ (2018) Permeability imaging as a predictor of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. J Cereb Blood Flow Metab 38:973–979. https://doi.org/10.1177/0271678x18768670
Sage MR (1982) Blood-brain barrier: phenomenon of increasing importance to the imaging clinician. AJR Am J Roentgenol 138:887–898. https://doi.org/10.2214/ajr.138.5.887
Saitou M, Fujimoto K, Doi Y, Itoh M, Fujimoto T, Furuse M, Takano H, Noda T, Tsukita S (1998) Occludin-deficient embryonic stem cells can differentiate into polarized epithelial cells bearing tight junctions. J Cell Biol 141:397–408. https://doi.org/10.1083/jcb.141.2.397
Sanelli PC, Ugorec I, Johnson CE, Tan J, Segal AZ, Fink M, Heier LA, Tsiouris AJ, Comunale JP, John M, Stieg PE, Zimmerman RD, Mushlin AI (2011) Using quantitative CT perfusion for evaluation of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. AJNR Am J Neuroradiol 32:2047–2053. https://doi.org/10.3174/ajnr.A2693
Sasaki T, Kassell NF, Yamashita M, Fujiwara S, Zuccarello M (1985) Barrier disruption in the major cerebral arteries following experimental subarachnoid hemorrhage. J Neurosurg 63:433–440. https://doi.org/10.3171/jns.1985.63.3.0433
Scholler K, Trinkl A, Klopotowski M, Thal SC, Plesnila N, Trabold R, Hamann GF, Schmid-Elsaesser R, Zausinger S (2007) Characterization of microvascular basal lamina damage and blood-brain barrier dysfunction following subarachnoid hemorrhage in rats. Brain Res 1142:237–246. https://doi.org/10.1016/j.brainres.2007.01.034
Sehba FA, Mostafa G, Knopman J, Friedrich V Jr, Bederson JB (2004) Acute alterations in microvascular basal lamina after subarachnoid hemorrhage. J Neurosurg 101:633–640. https://doi.org/10.3171/jns.2004.101.4.0633
Serlin Y, Ofer J, Ben-Arie G, Veksler R, Ifergane G, Shelef I, Minuk J, Horev A, Friedman A (2019) Blood-Brain Barrier Leakage: A New Biomarker in Transient Ischemic Attacks. Stroke 50:1266–1269. https://doi.org/10.1161/strokeaha.119.025247
Skriver EB, Olsen TS (1981) Transient disappearance of cerebral infarcts on CT scan, the so-called fogging effect. Neuroradiology 22:61–65. https://doi.org/10.1007/bf00344775
Skriver EB, Olsen TS (1982) Contrast enhancement of cerebral infarcts. Incidence and clinical value in different states of cerebral infarction. Neuroradiology 23:259–265. https://doi.org/10.1007/bf00339392
Sourbron SP, Buckley DL (2013) Classic models for dynamic contrast-enhanced MRI. NMR Biomed 26:1004–1027. https://doi.org/10.1002/nbm.2940
Suzuki H, Ayer R, Sugawara T, Chen W, Sozen T, Hasegawa Y, Kanamaru K, Zhang JH (2010) Protective effects of recombinant osteopontin on early brain injury after subarachnoid hemorrhage in rats. Crit Care Med 38:612–618. https://doi.org/10.1097/CCM.0b013e3181c027ae
Sweeney K, Silver N, Javadpour M (2016) Subarachnoid haemorrhage (spontaneous aneurysmal). BMJ Clin Evid 2016:1213
Teo M, Guilfoyle MR, Turner C, Kirkpatrick PJ (2017) What factors determine treatment outcome in aneurysmal subarachnoid hemorrhage in the modern era? A Post Hoc STASH Analysis. World Neurosurg 105:270–281. https://doi.org/10.1016/j.wneu.2017.05.005
Thal SC, Sporer S, Plesnila N, Schoeller K, Schmid-Elsaesser R, Zausinger S (2005) Evolution of brain edema after subarachnoid hemorrhage (SAH) in rats. J Cereb Blood Flow Metab 25:S271–S271
Wang Z, Meng CJ, Shen XM, Shu Z, Ma C, Zhu GQ, Liu HX, He WC, Sun XB, Huo L, Zhang J, Chen G (2012) Potential contribution of hypoxia-inducible factor-1alpha, aquaporin-4, and matrix metalloproteinase-9 to blood-brain barrier disruption and brain edema after experimental subarachnoid hemorrhage. J Mol Neurosci 48:273–280. https://doi.org/10.1007/s12031-012-9769-6
Winkler L, Blasig R, Breitkreuz-Korff O, Berndt P, Dithmer S, Helms HC, Puchkov D, Devraj K, Kaya M, Qin Z, Liebner S, Wolburg H, Andjelkovic AV, Rex A, Blasig IE, Haseloff RF (2020) Tight junctions in the blood-brain barrier promote edema formation and infarct size in stroke - Ambivalent effects of sealing proteins. J Cereb Blood Flow Metab:271678x20904687. https://doi.org/10.1177/0271678x20904687
Won S, Lee JH, Wali B, Stein DG, Sayeed I (2014) Progesterone attenuates hemorrhagic transformation after delayed tPA treatment in an experimental model of stroke in rats: involvement of the VEGF-MMP pathway. J Cereb Blood Flow Metab 34:72–80. https://doi.org/10.1038/jcbfm.2013.163
Xie Z, Enkhjargal B, Reis C, Huang L, Wan W, Tang J, Cheng Y, Zhang JH (2017) Netrin-1 preserves blood-brain barrier integrity through deleted in colorectal cancer/focal adhesion kinase/RhoA signaling pathway following subarachnoid hemorrhage in rats. J Am Heart Assoc 6. https://doi.org/10.1161/jaha.116.005198
Yan J, Chen C, Hu Q, Yang X, Lei J, Yang L, Wang K, Qin L, Huang H, Zhou C (2008) The role of p53 in brain edema after 24 h of experimental subarachnoid hemorrhage in a rat model. Exp Neurol 214:37–46. https://doi.org/10.1016/j.expneurol.2008.07.006
Yan J, Li L, Khatibi NH, Yang L, Wang K, Zhang W, Martin RD, Han J, Zhang J, Zhou C (2011) Blood-brain barrier disruption following subarchnoid hemorrhage may be faciliated through PUMA induction of endothelial cell apoptosis from the endoplasmic reticulum. Exp Neurol 230:240–247. https://doi.org/10.1016/j.expneurol.2011.04.022
Yi R, Xiao-Ping G, Hui L (2015) Atorvastatin prevents angiotensin II-induced high permeability of human arterial endothelial cell monolayers via ROCK signaling pathway. Biochem Biophys Res Commun 459:94–99. https://doi.org/10.1016/j.bbrc.2015.02.076
Yin D, Zhou S, Xu X, Gao W, Li F, Ma Y, Sun D, Wu Y, Guo Q, Liu H, Han L, Wang Z, Wang Y, Zhang J (2018) Dexmedetomidine attenuated early brain injury in rats with subarachnoid haemorrhage by suppressing the inflammatory response: The TLR4/NF-kappaB pathway and the NLRP3 inflammasome may be involved in the mechanism. Brain Res 1698:1–10. https://doi.org/10.1016/j.brainres.2018.05.040
Yuan J, Liu W, Zhu H, Zhang X, Feng Y, Chen Y, Feng H, Lin J (2017) Curcumin attenuates blood-brain barrier disruption after subarachnoid hemorrhage in mice. J Surg Res 207:85–91. https://doi.org/10.1016/j.jss.2016.08.090
Zhang H, Zhang B, Li S, Liang C, Xu K, Li S (2013) Whole brain CT perfusion combined with CT angiography in patients with subarachnoid hemorrhage and cerebral vasospasm. Clin Neurol Neurosurg 115:2496–2501. https://doi.org/10.1016/j.clineuro.2013.10.004
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
For this type of study, formal consent is not required.
Informed consent
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Comments
The authors claim that the manuscript is a narrative review on the hypothesis that clinical imaging of the blood-brain barrier permeability may serve as a predictor for delayed cerebral ischaemia following subarachnoid haemorrhage.
They state that there has been limited use of BBB imaging in clinical practice for the prediction and prognostication of DCI following aSAH. Accordingly, they discuss some of the mechanisms involved in BBB disruption following aSAH and the potential role of BBB integrity imaging in prediction of DCI and outcome prognostication following aSAH.
The concept that detection of focal BBB damage following aneurysmal subarachnoid haemorrhage in the acute phase of the disease as shown by abnormal contrast enhancement may be an early indicator/biomarker of development of delayed vasospasm/focal DCI is rather old. The authors rightly point out that DCI, however, has now been shown to occur independent of cerebral vasospasm, i.e. the pathogenesis of DCI may not be fully a result of pathological vasoconstriction.
The authors performed an anecdotical search in the English literature looking for papers dealing with disruptions to the blood-brain barrier in SAH; BBB permeability imaging as a predictor of DCI. Based on their critical reading experiences, they conclude that there is sufficient evidence from preclinical and clinical studies to suggest that alterations to the permeability of the BBB play a crucial role in the complex pathophysiology of DCI following SAH.
Radiological biomarkers offer a potential non-invasive prognostication tool and may also allow early identifications of patients who may be at risk of DCI.
With the recent advances in permeability imaging, it is imperative that potential associations between BBB disruption and early and delayed brain injuries are investigated in patients with aSAH.
Tamas Peter Doczi
Pecs, Hungary
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Vascular Neurosurgery – Aneurysm
Rights and permissions
About this article
Cite this article
Amoo, M., Henry, J., Pender, N. et al. Blood-brain barrier permeability imaging as a predictor for delayed cerebral ischaemia following subarachnoid haemorrhage. A narrative review. Acta Neurochir 163, 1457–1467 (2021). https://doi.org/10.1007/s00701-020-04670-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04670-6