Abstract
Aims
To evaluate whether a second insulin bolus, calculated with a new approach, could improve postprandial glucose (PPG) after the intake of real-life high-fat (HF) and high-protein (HP) mixed meals.
Methods
Fifteen adolescents with T1D treated with non-automated insulin pumps and CGM were enrolled. Patients received standard, HF and HP mixed meals treated with one pre-meal insulin bolus; based on differences in PPG between standard, HF and HP meals, correction boluses were calculated (30% and 60% of pre-meal bolus for HF and HP meals, respectively). Then patients received the same HF or HP meal treated with pre-meal bolus plus second insulin bolus after 3 h. Differences between postprandial variables after HF and HP meals treated with one or two insulin boluses were assessed by paired Student’s t-test.
Results
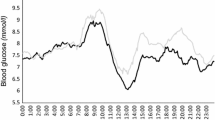
Treating HF and HP meals with two insulin boluses significantly reduced the postprandial BG-AUC (21% and 26% respectively, p < 0.05), increased %TIR (from 52.5 to 78.3% for HF meal; from 32.7 to 57.1% for HP meal; p < 0.01), and reduced mean BG and %TAR (p < 0.01), with no differences in %TBR.
Conclusions
The new way to calculate and administer correction boluses 3 h after HF and HP meals is effective and safe in reducing PPG and the hypoglycemia risk.


Similar content being viewed by others
Change history
02 March 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00592-023-02053-8
References
Diabetes Control and Complications Trial Research Group (1994) Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: diabetes control and complications trial. J Pediatr 125(2):177–188. https://doi.org/10.1016/s0022-3476(94)70190-3
Danne T, Phillip M, Buckingham BA et al. (2018) ISPAD clinical practice consensus guidelines 2018: insulin treatment in children and adolescents with diabetes. Pediatr Diabetes 19(Suppl 27):115–135. https://doi.org/10.1111/pedi.12718
Danne T, Cariou B, Buse JB et al. (2019) Improved time in range and glycemic variability with Sotagliflozin in combination with insulin in adults with type 1 diabetes: a pooled analysis of 24-week continuous glucose monitoring data from the inTandem program. Diabetes Care 42(5):919–930. https://doi.org/10.2337/dc18-2149
Owens DR, Bolli GB (2020) The continuing quest for better subcutaneously administered prandial insulins: a review of recent developments and potential clinical implications. Diabetes Obes Metab 22(5):743–754. https://doi.org/10.1111/dom.13963
Gordin D, Rönnback M, Forsblom C, Heikkilä O, Saraheimo M, Groop PH (2007) Acute hyperglycaemia rapidly increases arterial stiffness in young patients with type 1 diabetes. Diabetologia 50(9):1808–1814. https://doi.org/10.1007/s00125-007-0730-0
Madsbad S (2016) Impact of post-prandial glucose control on diabetes-related complications: how is the evidence evolving? J Diabetes Complicat 30(2):374–385. https://doi.org/10.1016/j.jdiacomp.2015.09.019
Smart CE, Annan F, Higgins LA, Jelleryd E, Lopez M, Acerini CL (2018) ISPAD clinical practice consensus guidelines 2018: nutritional management in children and adolescents with diabetes. Pediatr Diabetes 19(Suppl 27):136–154. https://doi.org/10.1111/pedi.12738
Cardona-Hernandez R, Schwandt A, Alkandari H et al. (2021) Glycemic outcome associated with insulin pump and glucose sensor use in children and adolescents with type 1 diabetes. Data from the international pediatric registry SWEET. Diabetes Care 44(5):1176–1184. https://doi.org/10.2337/dc20-1674
Marigliano M, Eckert AJ, Guness PK, et al. (2021) Association of the use of diabetes technology with HbA1c and BMI-SDS in an international cohort of children and adolescents with type 1 diabetes: the SWEET project experience. Pediatr Diabetes 22(8):1120–1128. https://doi.org/10.1111/pedi.13274
Prigge R, McKnight JA, Wild SH, Scottish Diabetes Research Network Epidemiology Group (2021) International comparison of glycaemic control in people with type 1 diabetes: an update and extension. Diabet Med. https://doi.org/10.1111/dme.14766
Sawyer A, Sobczak M, Forlenza GP, Alonso GT (2022) Glycemic control in relation to technology use in a single center cohort of children with type 1 diabetes. Diabetes Technol Ther. https://doi.org/10.1089/dia.2021.0471
Bell KJ, Barclay AW, Petocz P, Colagiuri S, Brand-Miller JC (2014) Efficacy of carbohydrate counting in type 1 diabetes: a systematic review and meta-analysis. Lancet Diabetes Endocrinol 2(2):133–140. https://doi.org/10.1016/S2213-8587(13)70144-X
Marigliano M, Morandi A, Maschio M, Sabbion A, Contreas G, Tomasselli F, Tommasi M, Maffeis C (2013) Nutritional education and carbohydrate counting in children with type 1 diabetes treated with continuous subcutaneous insulin infusion: the effects on dietary habits, body composition and glycometabolic control. Acta Diabetol 50(6):959–964. https://doi.org/10.1007/s00592-013-0491-9
Smart CE, Ross K, Edge JA, King BR, McElduff P, Collins CE (2010) Can chil- dren with type 1 diabetes and their caregivers estimate the carbohy- drate content of meals and snacks? Diabet Med 27(3):348–353
Smart CE, Ross K, Edge JA, Collins CE, Colyvas K, King BR (2009) Children and adolescents on intensive insulin therapy maintain postprandial glycaemic control without precise carbohydrate counting. Diabet Med 26:279–285
Marran KJ, Davey B, Lang A, Segal DG (2013) Exponential increase in post-prandial blood-glucose exposure with increasing carbohydrate loads using a linear carbohydrate-to-insulin ratio. S Afr Med J 103(7):461–463. https://doi.org/10.7196/samj.6382
Davison KA, Negrato CA, Cobas R et al. (2014) Relationship between adherence to diet, glycemic control and cardiovascular risk factors in patients with type 1 diabetes: a nationwide survey in Brazil. Nutr J 13:19. https://doi.org/10.1186/1475-2891-13-19
Nansel TR, Haynie DL, Lipsky LM, Laffel LM, Mehta SN (2012) Multiple indicators of poor diet quality in children and adolescents with type 1 diabetes are associated with higher body mass index percentile but not glycemic control. J Acad Nutr Diet 112(11):1728–1735. https://doi.org/10.1016/j.jand.2012.08.029
Evert AB, Dennison M, Gardner CD et al. (2019) Nutrition therapy for adults with diabetes or prediabetes: a consensus report. Diabetes Care 42(5):731–754. https://doi.org/10.2337/dci19-0014
Bell KJ, Smart CE, Steil GM, Brand-Miller JC, King B, Wolpert HA (2015) Impact of fat, protein, and glycemic index on post-prandial glucose control in type 1 diabetes: implications for intensive diabetes management in the continuous glucose monitoring era. Diabetes Care 38(6):1008–1015. https://doi.org/10.2337/dc15-0100
Wolpert HA, Atakov-Castillo A, Smith SA, Steil GM (2013) Dietary fat acutely increases glucose concentrations and insulin requirements in patients with type 1 diabetes: implications for carbohydrate-based bolus dose calculation and intensive diabetes management. Diabetes Care 36(4):810–816. https://doi.org/10.2337/dc12-0092
Pańkowska E, Błazik M, Groele L (2012) Does the fat-protein meal increase post-prandial glucose level in type 1 diabetes patients on insulin pump: the conclusion of a randomized study. Diabetes Technol Ther 14(1):16–22. https://doi.org/10.1089/dia.2011.0083
Smart CE, Evans M, O’Connell SM et al. (2013) Both dietary protein and fat increase post-prandial glucose excursions in children with type 1 diabetes, and the effect is additive. Diabetes Care 36(12):3897–3902. https://doi.org/10.2337/dc13-1195
Paterson M, Bell KJ, O’Connell SM, Smart CE, Shafat A, King B (2015) The role of dietary protein and fat in glycaemic control in type 1 diabetes: implications for intensive diabetes management. Curr Diab Rep 15(9):61. https://doi.org/10.1007/s11892-015-0630-5
Paterson MA, Smart CE, Lopez PE et al. (2016) Influence of dietary protein on post-prandial blood glucose levels in individuals with type 1 diabetes mellitus using intensive insulin therapy. Diabet Med 33(5):592–598. https://doi.org/10.1111/dme.13011
Wolever TM, Mullan YM (2011) Sugars and fat have different effects on postprandial glucose responses in normal and type 1 diabetic subjects. Nutr Metab Cardiovasc Dis 21(9):719–725. https://doi.org/10.1016/j.numecd.2010.12.005
Neu A, Behret F, Braun R et al. (2015) Higher glucose concentrations following protein: and fat-rich meals—the Tuebingen grill study: a pilot study in adolescents with type 1 diabetes. Pediatr Diabetes 16(8):587–591. https://doi.org/10.1111/pedi.12224
Paterson MA, King BR, Smart CEM, Smith T, Rafferty J, Lopez PE (2019) Impact of dietary protein on post-prandial glycaemic control and insulin requirements in type 1 diabetes: a systematic review. Diabet Med 36(12):1585–1599. https://doi.org/10.1111/dme.14119
Kordonouri O, Hartmann R, Remus K, Bläsig S, Sadeghian E, Danne T (2012) Benefit of supplementary fat plus protein counting as compared with conventional carbohydrate counting for insulin bolus calculation in children with pump therapy. Pediatr Diabetes 13(7):540–544. https://doi.org/10.1111/j.1399-5448.2012.00880.x
Pańkowska E, Szypowska A, Lipka M, Szpotańska M, Błazik M, Groele L (2009) Application of novel dual wave meal bolus and its impact on glycated hemoglobin A1c level in children with type 1 diabetes. Pediatr Diabetes 10(5):298–303. https://doi.org/10.1111/j.1399-5448.2008.00471.x
Metwally M, Cheung TO, Smith R, Bell KJ (2021) Insulin pump dosing strategies for meals varying in fat, protein or glycaemic index or grazing-style meals in type 1 diabetes: a systematic review. Diabetes Res Clin Pract 172:108516. https://doi.org/10.1016/j.diabres.2020.108516
Lopez PE, Smart CE, McElduff P et al. (2017) Optimizing the combination insulin bolus split for a high-fat, high-protein meal in children and adolescents using insulin pump therapy. Diabet Med 34(10):1380–1384. https://doi.org/10.1111/dme.13392
Bell KJ, Toschi E, Steil GM, Wolpert HA (2016) Optimized mealtime insulin dosing for fat and protein in type 1 diabetes: application of a model-based approach to derive insulin doses for open-loop diabetes management. Diabetes Care 39(9):1631–1634. https://doi.org/10.2337/dc15-2855
Lopez PE, Evans M, King BR et al. (2018) A randomized comparison of three prandial insulin dosing algorithms for children and adolescents with type 1 diabetes. Diabet Med 35(10):1440–1447. https://doi.org/10.1111/dme.13703
Frohock AM, Oke J, Yaliwal C, Edge J, Besser REJ (2022) Additional insulin dosing for fat and protein in children with type 1 diabetes using multiple daily injections. Pediatr Diabetes 23(6):742–748. https://doi.org/10.1111/pedi.13372
Walsh J, Roberts R, Heinemann L (2014) Confusion regarding duration of insulin action: a potential source for major insulin dose errors by bolus calculators. J Diabetes Sci Technol 8(1):170–178. https://doi.org/10.1177/1932296813514319
Piechowiak K, Dżygało K, Szypowska A (2017) The additional dose of insulin for high-protein mixed meal provides better glycemic control in children with type 1 diabetes on insulin pumps: randomized cross-over study. Pediatr Diabetes 18(8):861–868. https://doi.org/10.1111/pedi.12500
Campbell MD, Walker M, King D et al. (2016) Carbohydrate counting at meal time followed by a small secondary post-prandial bolus injection at 3 h prevents late hyperglycemia, without hypoglycemia, after a high-carbohydrate, high-fat meal in type 1 diabetes. Diabetes Care 39(9):e141–e142. https://doi.org/10.2337/dc16-0709
Cacciari E, Milani S, Balsamo A et al. (2006) Italian cross-sectional growth charts for height, weight and BMI (2–20 year). J Endocrinol Invest 29(7):581–593. https://doi.org/10.1007/BF03344156
Tanner JM, Whitehouse RH (1976) Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch Dis Child 51(3):170–179. https://doi.org/10.1136/adc.51.3.170
Scavone G, Manto A, Pitocco D et al. (2010) Effect of carbohydrate counting and medical nutritional therapy on glycaemic control in type 1 diabetic subjects: a pilot study. Diabet Med 27(4):477–479. https://doi.org/10.1111/j.1464-5491.2010.02963.x
Evans M, Smart CEM, Paramalingam N et al. (2019) Dietary protein affects both the dose and pattern of insulin delivery required to achieve post-prandial euglycaemia in type 1 diabetes: a randomized trial. Diabet Med 36(4):499–504. https://doi.org/10.1111/dme.13875
Paterson MA, Smart CEM, Howley P, Price DA, Foskett DC, King BR (2020) High-protein meals require 30% additional insulin to prevent delayed post-prandial hyperglycaemia. Diabet Med 37(7):1185–1191. https://doi.org/10.1111/dme.14308
Bell KJ, Fio CZ, Twigg S et al. (2020) Amount and type of dietary fat, post-prandial glycemia, and insulin requirements in type 1 diabetes: a randomized within-subject trial. Diabetes Care 43(1):59–66. https://doi.org/10.2337/dc19-0687
Smith TA, Smart CE, Fuery MEJ et al. (2021) In children and young people with type 1 diabetes using pump therapy, an additional 40% of the insulin dose for a high-fat, high-protein breakfast improves post-prandial glycaemic excursions: a cross-over trial. Diabet Med 6:e14511. https://doi.org/10.1111/dme.14511
Smith TA, Smart CE, Howley PP, Lopez PE, King BR (2021) For a high fat, high protein breakfast, preprandial administration of 125% of the insulin dose improves post-prandial glycaemic excursions in people with type 1 diabetes using multiple daily injections: a cross-over trial. Diabet Med 9:e14512. https://doi.org/10.1111/dme.14512
Keating B, Smart CEM, Harray AJ et al. (2021) Additional insulin is required in both the early and late post-prandial periods for meals high in protein and fat: a randomised trial. J Clin Endocrinol Metab 6:dgab318. https://doi.org/10.1210/clinem/dgab318
Acknowledgements
M.M. wrote the manuscript and researched data; C.P. reviewed the manuscript; F.T., Al. M. and A. M. researched data; and C.M. co-wrote the manuscript. All authors have revised and approved the submitted manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of Institutional and national research committee and with the 1975 Declaration of Helsinki, as revised in 2008. The study was approved (987CESC) by the Institutional Ethics Committee of Verona (Italy).
Informed consent
Informed consent was obtained from the adolescents and their parents.
Additional information
Managed by Massimo Federici.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The second affiliation of author “Claudia Piona” deleted.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marigliano, M., Piona, C., Tommaselli, F. et al. A new proposal for a second insulin bolus to optimize postprandial glucose profile in adolescents with type 1 diabetes. Acta Diabetol 60, 609–618 (2023). https://doi.org/10.1007/s00592-022-02019-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-022-02019-2