Abstract
Purpose
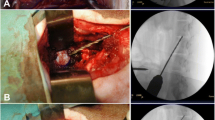
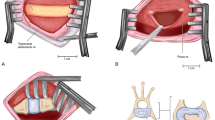
This work describes a minimally invasive damage model for ovine lumbar discs via partial nucleotomy using a posterolateral approach.
Methods
Two cadavers were dissected to analyze the percutaneous corridor. Subsequently, 28 ovine had their annulus fibrosus punctured via awl penetration under fluoroscopic control and nucleus pulposus tissue removed via rongeur. Efficacy was assessed by animal morbidity, ease of access to T12-S1 disc spaces, and production of a mechanical injury as verified by discography, radiography, and histology.
Results
T12-S1 were accessible with minimal nerve damage morbidity. Scar tissue sealed the disc puncture site in all animals within 6 weeks, withstanding 1 MP of intradiscal pressure. Partial nucleotomy led to a significant reduction in intervertebral disk height and an increased histological degeneration score.
Conclusion
Inducing a reproducible injury pattern of disc degeneration required minimal time, effort, and equipment. The posterolateral approach allows operation on several discs within a single surgery and multiple animal surgeries within a single day.







Similar content being viewed by others
References
Saito J, Ohtori S, Kishida S et al (2012) Difficulty of diagnosing the origin of lower leg pain in patients with both lumbar spinal stenosis and hip joint osteoarthritis. Spine 37:2089–2093. doi:10.1097/BRS.0b013e31825d213d
Sakai D, Andersson GBJ (2015) Stem cell therapy for intervertebral disc regeneration: obstacles and solutions. Nat Rev Rheumatol 11:243–256. doi:10.1038/nrrheum.2015.13
Sakai D, Grad S (2015) Advancing the cellular and molecular therapy for intervertebral disc disease. Adv Drug Deliv Rev 84:159–171. doi:10.1016/j.addr.2014.06.009
Reitmaier S, Schmidt H, Ihler R et al (2013) Preliminary investigations on intradiscal pressures during daily activities: an in vivo study using the merino sheep. PLoS One 8:e69610. doi:10.1371/journal.pone.0069610
Wilke HJ, Kettler A, Wenger KH, Claes LE (1997) Anatomy of the sheep spine and its comparison to the human spine. Anat Rec 247:542–555. doi:10.1002/(SICI)1097-0185(199704)247:4<542
Mageed M, Berner D, Julke H et al (2013) Morphometrical dimensions of the sheep thoracolumbar vertebrae as seen on digitised CT images. Lab Anim Res 29:138–147. doi:10.5625/lar.2013.29.3.138
Hoogendoorn RJ, Wuisman PI, Smit TH et al (2007) Experimental intervertebral disc degeneration induced by chondroitinase ABC in the goat. Spine 32:1816–1825. doi:10.1097/BRS.0b013e31811ebac5
Alini M, Eisenstein SM, Ito K et al (2008) Are animal models useful for studying human disc disorders/degeneration? Eur Spine J 17:2–19. doi:10.1007/s00586-007-0414-y
Moss IL, Zhang YJ, Shi P et al (2013) Retroperitoneal approach to the intervertebral disc for the annular puncture model of intervertebral disc degeneration in the rabbit. Spine J 13:229–234. doi:10.1016/j.spinee.2012.02.028
Baramki HG, Papin P, Steffen T (2000) A surgical approach to the ventral aspect of the lumbar vertebrae in the sheep model. Surg Radiol Anat 22:25–27. doi:10.1007/s00276-000-0025-3
Guder E, Hill S, Kandziora F, Schnake KJ (2009) Partial nucleotomy of the ovine disc as an in vivo mode for disc degeneration. Z Orthop Unfallchir 147:52–58. doi:10.1055/s-2008-1039139
Oehme D, Goldschlager T, Rosenfeld J et al (2012) Lateral surgical approach to lumbar intervertebral discs in an ovine model. Sci World J 2012:1–5. doi:10.1100/2012/873726
Vadala G, De Strobel F, Bernardini M et al (2013) The transpedicular approach for the study of intervertebral disc regeneration strategies: in vivo characterization. Eur Spine J 22:S972–S978. doi:10.1007/s00586-013-3007-y
Dabbs VM, Dabbs LG (1990) Correlation between disk height narrowing and low-back-pain. Spine 15:1366–1369. doi:10.1097/00007632-199012000-00026
Mulisch M, Welsch U (2010) Romeis—Mikroskopische Technik. Springer, Berlin
Oehme D, Ghosh P, Shimmon S et al (2014) Mesenchymal progenitor cells combined with pentosan polysulfate mediating disc regeneration at the time of microdiscectomy: a preliminary study in an ovine model. J Neurosurg Spine 20:657–669. doi:10.3171/2014.2.SPINE13760
Oehme D, Goldschlager T, Ghosh P et al (2015) Cell-based therapies used to treat lumbar degenerative disc disease: a systematic review of animal studies and human clinical trials. Stem Cells Int. doi:10.1155/2015/946031
Wilke H-J, Heuer F, Neidlinger-Wilke C, Claes L (2006) Is a collagen scaffold for a tissue engineered nucleus replacement capable of restoring disc height and stability in an animal model? Eur Spine J 15:433–438. doi:10.1007/s00586-006-0177-x
Vadala G, Russo F, Pattappa G et al (2013) The transpedicular approach as an alternative route for intervertebral disc regeneration. Spine 38:E319–E324. doi:10.1097/BRS.0b013e318285bc4a
Reitmaier S, Volkheimer D, Berger-Roscher N et al (2014) Increase or decrease in stability after nucleotomy? Conflicting in vitro and in vivo results in the sheep model. J R Soc Interface 11:UNSP 20140650. doi:10.1098/rsif.2014.0650
Melrose J, Roberts S, Smith S et al (2002) Increased nerve and blood vessel ingrowth associated with proteoglycan depletion in an ovine anular lesion model of experimental disc degeneration. Spine. doi:10.1097/00007632-200206150-00007
van Heeswijk VM, Thambyah A, Robertson PA, Broom ND (2017) Posterolateral disc prolapse in flexion initiated by lateral inner annular failure: an investigation of the herniation pathway. Spine. doi:10.1097/BRS.0000000000002181
Rajasekaran S, Bajaj N, Tubaki V et al (2013) ISSLS prize winner: the anatomy of failure in lumbar disc herniation: an in vivo, multimodal, prospective study of 181 subjects. Spine 38:1491–1500
Berger-Roscher N, Galbusera F, Rasche V, Wilke H-J (2015) Intervertebral disc lesions: visualisation with ultra-high field MRI at 11.7 T. Eur Spine J 24:2488–2495
Wilke H-J, Kienle A, Maile S et al (2016) A new dynamic six degrees of freedom disc-loading simulator allows to provoke disc damage and herniation. Eur Spine J 25:1363–1372
Mengoni M, Jones AC, Wilcox RK (2016) Modelling the failure precursor mechanism of lamellar fibrous tissues, example of the annulus fibrosus. J Mech Behav Biomed Mater 63:265–272. doi:10.1016/j.jmbbm.2016.06.030
Wade KR, Robertson PA, Thambyah A, Broom ND (2015) Surprise loading in flexion increases the risk of disc herniation due to annulus-endplate junction failure: a mechanical and microstructural investigation. Spine 40:891–901
Zak M, Pezowicz C (2013) Spinal sections and regional variations in the mechanical properties of the annulus fibrosus subjected to tensile loading. Acta Bioeng Biomech 15:51–59. doi:10.5277/abb130107
Hadjipavlou AG, Tzermiadianos MN, Bogduk N, Zindrick MR (2008) The pathophysiology of disc degeneration. J Bone Joint Surg Br 90B:1261–1270. doi:10.1302/0301-620X.90B10.20910
Duance VC, Crean JKG, Sims TJ et al (1998) Changes in collagen cross-linking in degenerative disc disease and scoliosis. Spine 23:2545–2551. doi:10.1097/00007632-199812010-00009
Osti OL, Vernon-Roberts B, Fraser RD (1990) Anulus tears and intervertebral disc degeneration: an experimental study using an animal model. Spine 15:762–767
Acknowledgement
The authors would like to thank W. Petzold and M. Menzel for technical assistance. Our special thanks go to the staff of the Animal Core Unit of the Translational Centre of Regenerative Medicine TRM, our keeper S. Köhler and veterinary assistant M. Köberle. The work presented in this paper was made possible by funding from the German Federal Ministry of Education and Research (BMBF, 1315883), the DAAD internship program RISE, and the Whitaker Biomedical Engineering Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Schwan, S., Ludtka, C., Wiesner, I. et al. Percutaneous posterolateral approach for the simulation of a far-lateral disc herniation in an ovine model. Eur Spine J 27, 222–230 (2018). https://doi.org/10.1007/s00586-017-5362-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-017-5362-6