Abstract
Purpose
To develop a large animal model for acute central cervical spinal cord injury syndrome (ACCSCIS.
Methods
Twenty-four adult male goats were randomized into four groups including group A with acute compression injury, group B with anterior chronic compression, group C as the test group that received anterior chronic compression by screw and acute compression by posterior balloon insertion, and group D as normal controls that received sham surgery. Neurological function (modified Tarlov motor function), CT, MRI, cortical somatosensory evoked potentials (CSEP), and pathological analysis were evaluated. The data were analyzed statistically.
Results
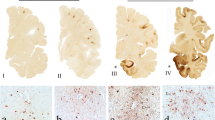
The motor function of the goats in group C was significantly lower than other groups. CSEP before spinal cord compression showed a stable pattern. Spinal cord compression resulted in a gradual decrement in the peak latency and significant increment in the peak amplitude. Cervical spinal canal occupying ratio was significantly lower in group C than the other groups. MRI revealed focal low signal in T1 weighted images and focal high signal in T2 weighted images in group C. Pathological analysis showed more severe lesions in the gray matter than that in the white matter in group C.
Conclusions
The model well simulated the pathogenesis and resembled the clinical characteristics of ACCSCIS. This model seems to have the potential to contribute to the development of effective therapies for ACCSCIS.




Similar content being viewed by others
References
McKinley W, Santos K, Meade M, Brooke K (2007) Incidence and outcomes of spinal cord injury clinical syndromes. J Spinal Cord Med 30:215
Bosch A, Stauffer ES, Nickel VL (1971) Incomplete traumatic quadriplegia: a ten-year review. JAMA 216:473–478
Fong W, Eismont FJ (2007) Controversies in the treatment of central cord injuries. Seminars in spine surgery. Elsevier, London, pp 260–271
Molliqaj G, Payer M, Schaller K, Tessitore E (2014) Acute traumatic central cord syndrome: a comprehensive review. Neurochirurgie 60:5–11. doi:10.1016/j.neuchi.2013.12.002
Bennett AD, Everhart AW, Hulsebosch CE (2000) Intrathecal administration of an NMDA or a non-NMDA receptor antagonist reduces mechanical but not thermal allodynia in a rodent model of chronic central pain after spinal cord injury. Brain Res 859:72–82
Vanický I, Urdzíková L, Saganová K, Cízková D, Gálik J (2001) A simple and reproducible model of spinal cord injury induced by epidural balloon inflation in the rat. J Neurotrauma 18:1399–1407
Thompson C, Gonsalves JF, Welsh D (2015) Hyperextension injury of the cervical spine with central cord syndrome. Eur Spine J 24:195–202
Khan M, Griebel R (1983) Acute spinal cord injury in the rat: comparison of three experimental techniques. Can J Neurol Sci 10:161–165
Blight AR (1991) Morphometric analysis of a model of spinal cord injury in guinea pigs, with behavioral evidence of delayed secondary pathology. J Neurol Sci 103:156–171
Tarlov IM, Klinger H (1954) Spinal cord compression studies. II. Time limits for recovery after acute compression in dogs. AMA Arch Neurol Psychiatry 71:271–290
Aarabi B, Koltz M, Ibrahimi D (2008) Hyperextension cervical spine injuries and traumatic central cord syndrome. Neurosurg Focus 25:E9
Schneider RC, Cherry G, Pantek H (1954) The syndrome of acute central cervical spinal cord injury: with special reference to the mechanisms involved in hyperextension injuries of cervical spine*. J Neurosurg 11:546–577
Quencer R, Bunge R, Egnor M, Green B, Puckett W, Naidich T, Post M, Norenberg M (1992) Acute traumatic central cord syndrome: MRI-pathological correlations. Neuroradiology 34:85–94
Koyanagi I, Iwasaki Y, Hida K, Akino M, Imamura H, Abe H (2000) Acute cervical cord injury without fracture or dislocation of the spinal column. J Neurosurg 93:15–20
Fukuda S, Nakamura T, Kishigami Y, Endo K, Azuma T, Fujikawa T, Tsutsumi S, Shimizu Y (2005) New canine spinal cord injury model free from laminectomy. Brain Res Brain Res Protoc 14:171–180. doi:10.1016/j.brainresprot.2005.01.001
Fehlings MG, Tator CH, Linden RD (1989) The relationships among the severity of spinal cord injury, motor and somatosensory evoked potentials and spinal cord blood flow. Electroencephalogr Clin Neurophysiol 74:241–259
Brown RH, Nash CL Jr, Berilla JA, Amaddio MD (1984) Cortical evoked potential monitoring. A system for intraoperative monitoring of spinal cord function. Spine (Phila Pa 1976) 9:256–261
Laschinger JC, Izumoto H, Kouchoukos NT (1987) Evolving concepts in prevention of spinal cord injury during operations on the descending thoracic and thoracoabdominal aorta. Ann Thorac Surg 44:667–674
Koyanagi I, Iwasaki Y, Hida K, Akino M, Imamura H, Abe H (2000) Acute cervical cord injury without fracture or dislocation of the spinal column. J Neurosurg Spine 93:15–20
Koyanagi I, Iwasaki Y, Hida K, Imamura H, Fujimoto S, Akino M (2003) Acute cervical cord injury associated with ossification of the posterior longitudinal ligament. Neurosurgery 53:887–892
Hubscher CH, Fell JD, Gupta DS (2010) Sex and hormonal variations in the development of at-level allodynia in a rat chronic spinal cord injury model. Neurosci Lett 477:153–156. doi:10.1016/j.neulet.2010.04.053
Sharp K, Boroujerdi A, Steward O, Luo ZD (2012) A rat chronic pain model of spinal cord contusion injury. Methods Mol Biol 851:195–203. doi:10.1007/978-1-61779-561-9_14
Purdy PD, Duong RT, White CL 3rd, Baer DL, Reichard RR, Pride GL Jr, Adams C, Miller S, Hladik CL, Yetkin Z (2003) Percutaneous translumbar spinal cord compression injury in a dog model that uses angioplasty balloons: MR imaging and histopathologic findings. AJNR Am J Neuroradiol 24:177–184
Courtine G, Bunge MB, Fawcett JW, Grossman RG, Kaas JH, Lemon R, Maier I, Martin J, Nudo RJ, Ramon-Cueto A, Rouiller EM, Schnell L, Wannier T, Schwab ME, Edgerton VR (2007) Can experiments in nonhuman primates expedite the translation of treatments for spinal cord injury in humans? Nat Med 13:561–566. doi:10.1038/nm1595
Assina R, Sankar T, Theodore N, Javedan SP, Gibson AR, Horn KM, Berens M, Sonntag VK, Preul MC (2008) Activated autologous macrophage implantation in a large-animal model of spinal cord injury. Neurosurg Focus 25:E3. doi:10.3171/FOC.2008.25.11.E3
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jiang, H., Wang, J., Xu, B. et al. A model of acute central cervical spinal cord injury syndrome combined with chronic injury in goats. Eur Spine J 26, 56–63 (2017). https://doi.org/10.1007/s00586-016-4573-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4573-6