Abstract
Background
To present the strategies and preliminary findings of the first 3 years after implementing a Helicobacter pylori screening and eradication program to prevent gastric cancer in Saga Prefecture.
Methods
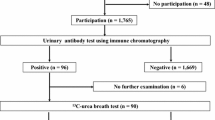
A screening and treatment program to eradicate H. pylori from third-grade junior high students was started in Saga Prefecture in 2016, using local governmental grants. Screening was with urinary anti-H. pylori antibody tests, followed by H. pylori stool antigen tests for students who were antibody positive. Those positive on both tests underwent H. pylori eradication by triple therapy based on a potassium-competitive acid blocker.
Results
From 2016 to 2018, the participation rate was 83.1% and the H. pylori infection rate was 3.1% (660/21,042). The participation rates were higher in 2017 (85.4%) and 2018 (85.9%) compared with 2016 (78.5%) (P < 0.0001), and the infection rate also decreased in a time-dependent manner (2016: 3.6%, 2017: 3.3%, 2018: 2.5%, P = 0.0001). In total, 501 students positive for H. pylori received eradication therapy (85.1% success) and adverse events occurred in 20 of these (4.0%). However, no serious complications occurred.
Conclusions
The H. pylori screening and eradication project for school students in Saga Prefecture has started successfully and we have seen both a steady increase in the participation rate and a steady decrease in the infection rate, without major safety concerns.


Similar content being viewed by others
References
Uemura N, Okamoto S, Yamamoto S, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–9.
Park JY, Forman D, Waskit LA, et al. Epidemiology of Helicobacter pylori and CagA-positive infections and global variations in gastric cancer. Toxins. 2018;10:e163.
Suzuki H, Mori H. World trends for H. pylori eradication therapy and gastric cancer prevention strategy by H. pylori test-and-treat. J Gastroenterol. 2018;53:354–61.
Hatakeyama H. Helicobacter pylori CagA and cancer: a paradigm for hit-and-run carcinogenesis. Cell Host Microbe. 2014;15:306–16.
Kikuchi S, Nakajima T, Kobayashi O, et al. Effect of age on the relationship between gastric cancer and Helicobacter pylori. Jpn J Cancer Res. 2000;91:774–9.
Ekstom AM, Held M, Hansson LE, et al. Helicobacter pylori in gastric cancer established by CagA immunoblot as a marker of past infection. Gastroenterology. 2001;121:784–91.
Horiuchi Y, Fujisaki J, Yamamoto N, et al. Biological behavior of the intramucosal Helicobacter pylori-negative undifferentiated-type early gastric cancer: comparison with helicobacter pylori-positive early gastric cancer. Gastric Cancer. 2016;19:160–5.
Tsuda M, Asaka M, Kato M, et al. Effect on Helicobacter pylori eradication therapy against gastric cancer in Japan. Helicobacter. 2017;22:e12415.
Sugano K. Effect of Helicobacter pylori eradication on the incidence of gastric cancer: a systematic review and meta-analysis. Gastric Cancer. 2018. https://doi.org/10.1007/s10120-018-0876-0
Asaka M, Mabe K, Matsushima R, et al. Helicobacter pylori eradication to eliminate gastric cancer: the Japanese strategy. Gastroenterol Clin N Am. 2015;44:639–48.
Tsutsumi K, Kusano C, Suzuki S, et al. Diagnostic accuracy of latex agglutination turbidimetric immunoassay in screening adolescents for Helicobacter pylori infection in Japan. Digestion. 2018;98:75–80.
Take S, Mizuno M, Ishiki K, et al. Baseline gastric mucosal atrophy is a risk factor associated with the development of gastric cancer after Helicobacter pylori eradication therapy in patients with peptic ulcer diseases. J Gastroenterol. 2007;42:21–7.
Wong BC, Lam SK, Wong WM, et al. Helicobacter pylori eradication to prevent gastric cancer in a high-risk region of China: a randomized controlled trial. JAMA. 2004;291:187–94.
Sugano K, Tack J, Kuipers EJ, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015;64:1353–67.
Kato S, Kikuchi S, Nakajima S. When does gastric atrophy develop in Japanese children? Helicobacter. 2008;13:278–81.
Nozaki K, Shimizu N, Ikehara Y, et al. Eradication diminishes enhancing effects of Helicobacter pylori infection on glandular stomach carcinogenesis in Mongolian gerbils. Cancer Sci. 2003;94:235–9.
Furuta T, El-Omar EM, Xiao F, et al. Interleukin 1beta polymorphisms increase risk of hypochlorhydria and atrophic gastritis and reduce risk of duodenal ulcer recurrence in Japan. Gastroenterology. 2002;123:92–105.
Akamatsu T, Ichikawa S, Okada S, et al. Introduction of an examination and treatment for Helicobacter pylori infection in high school health screening. J Gastroenterol. 2011;46:1353–60.
Kusano C, Gotoda T, Ishikawa H, et al. The administrative project of Helicobacter pylori infection screening among junior high school students in an area of Japan with a high incidence of gastric cancer. Gastric Cancer. 2017;20:S16–9.
Okuda M, Kamiya S, Booka M, et al. Diagnostic accuracy of urine-based kits for detection of Helicobacter pylori antigen in children. Pediatr Int. 2013;55:337–41.
Yamamoto T, Ishii T, Kawakami T, et al. Reliability of urinary tests for antibody to Helicobacter pylori in patients with pulmonary tuberculosis. World J Gastroenterol. 2005;11:412–4.
Tamamura T, Morita E, Kondo T, et al. Prevalence of Helicobacter pylori infection measured with urinary antibody in an urban area of Japan, 2008–2010. Nagoya J Med Sci. 2012;74:63–70.
Shimoyama T. Stool antigen tests for the management of Helicobacter pylori infection. World J Gastroenterol. 2013;19:818–91.
Fukuda Y, Tomita T, Hori K, et al. Evaluation of a novel Helicobacter pylori stool antigen detection kit, Testmate rapid pylori antigen, for rapid diagnosis of Helicobacter pylori infection. Jpn J Med Pharm Sci. 2004;52:469–74.
Zebala Torrres B, Luceno Y, Lagomarcino AJ, et al. Review: prevalence and dynamics of Helicobacter pylori infection during childhood. Helicobacter. 2017;22:e12399.
Tanaka Y, Sakata Y, Hara M, et al. Risk factors for Helicobacter pylori infection and endoscopic reflux esophagitis in healthy young Japanese volunteers. Intern Med. 2017;56:2979–83.
Takeshita E, Sakata Y, Hara M, et al. Higher frequency of reflux symptoms and acid-related dyspepsia in females than males regardless of endoscopic esophagitis: cross sectional analysis of 3,505 Japanese subjects undergoing medical health checkups. Digestion. 2016;93:266–71.
Fujimoto Y, Furusyo N, Toyoda K, et al. Intrafamilial transmission of Helicobacter pylori among the population of endemic areas in Japan. Helicobacter. 2007;12:170–6.
Asaka M, Kimura T, Kudo M, et al. Relationship of Helicobacter pylori to serum pepsinogens in an asymptomatic Japanese population. Gastroenterology. 1992;102:760–6.
Okuda M, Osaki T, Lin Y, et al. Low prevalence and incidence of Helicobacter pylori infection in children: a population-based study in Japan. Helicobacter. 2014;20:133–8.
Schwarz S, Morelli G, Kusecek B, et al. Horizontal versus familial transmission of Helicobacter pylori. PLoS Pathog. 2008;4:e1000180.
Rogers AH. The source of infection in the intrafamilial transfer of Streptococcus mutans. Caries Res. 1981;15:26–31.
Seow WK. Early childhood caries. Pesiatr Clin N Am. 2019;65:941–54.
Murakami K, Sakurai Y, Shiino M, et al. Vonoprazan, a novel potassium-competitive acid blocker, as a component of first-line and second-line triple therapy for Helicobacter pylori eradication: a phase III, randomized, double-blind study. Gut. 2016;65:1439–46.
Tanabe H, Ando K, Sato K, et al. Efficacy of vonoprazan-based triple therapy for Helicobacter pylori eradication: a multicenter study and a review of the literature. Dig Dis Sci. 2017;62:3069–76.
Takara Y, Endo H, Nakano R, et al. Smoking and drinking did not increase the failure of therapeutic Helicobacter pylori eradication by vonoprazan, clarithromycin, and amoxicillin. Digestion. 2018;4:1–7.
Ozaki H, Harada S, Takeuchi T, et al. Vonoprazan, a novel potassium-competitive acid blocker, should be used for the Helicobacter pylori eradication therapy as first choice: a large sample study of vonoprazan in real world compared with our randomized control trial using second-generation proton pump inhibitors for Helicobacter pylori eradication therapy. Digestion. 2018;97:212–8.
Suzuki S, Gotoda T, Kusano C, et al. The efficacy and tolerability of a triple therapy containing a potassium-competitive acid blocker compared with a 7-day PPI-based low dose clarithromycin triple therapy. Am J Gastroenterol. 2016;111:949–56.
Khurana R, Fischbach L, Chiba N, et al. Meta-analysis: Helicobacter pylori eradication treatment efficacy in children. Aliment Pharmacol Ther. 2007;25:423–36.
Jones NL, Koletzko S, Goodman K, et al. Joint ESPGHAN/NASPGHAN guideline for the management of Helicobacter pylori in children and adolescents (update 2016). J Pediatr Gastroenterol Nutr. 2017;23:2453–8.
Kotilea K, Mekhael J, Salame A, et al. Eradication rate of Helicobacter pylori infection is directly influenced by adherence to therapy in children. Helicobacter. 2017;22:e12383.
Kato S, Konno M, Maisawa S, et al. Results of triple eradication therapy in Japanese children: a retrospective multicenter study. J Gastroenterol. 2004;39:838–43.
Kato S, Fujimura S. Primary antimicrobial resistance of Helicobacter pylori in children during the past 9 years. Pediatr Int. 2010;52:18–90.
Savoldi A, Carrara E, Graham DY, et al. Prevalence of antibiotic resistance in Helicobacter pylori: a systematic review and meta-analysis in world health organization regions. Gastroenterology. 2018;55:1372–82.
Montes M, Villalon FN, Eizaguirre FJ, et al. Helicobacter pylori infection in children. Antimicrobial resistance and treatment response. Helicobacter. 2015;20:169–75.
Ogata SK, Godoy AP, da Silva Patricio FR, et al. High Helicobacter pylori resistance to metronidazole and clarithromycin in Brazilian children and adolescents. J Pediatr Gastroenterol Nutr. 2013;56:645–8.
Okamura T, Suga T, Nagaya T, et al. Antimicrobial resistance and characteristics of eradication therapy of Helicobacter pylori in Japan: a multi-generational comparison. Helicobacter. 2014;19:214–20.
Okuda M, Kikuchi S, Mabe K, et al. Nationwide survey of Helicobacter pylori treatment for children and adolescents in Japan. Pediatr Int. 2017;59:57–61.
Mabe K, Okuda M, Kikuchi S, et al. Randomized controlled trial: PPI-based triple therapy containing metronidazole versus clarithromycin as first-line treatment for Helicobacter pylori in adolescents and young adults in Japan. J Infect Chemother. 2018;24:538–43.
Adachi T, Matsui S, Watanabe T, et al. Comparative study of clarithromycin- versus metronidazole-based triple therapy as first-line eradication for Helicobacter pylori. Oncology. 2017;93:15–9.
Kusano C, Gotoda T, Suzuki S, et al. Safety of first-line triple therapy with a potassium-competitive acid blocker for Helicobacter pylori eradication in children. J Gastroenterol. 2018;53:718–24.
Gibbons TE, Gold BD. The use of proton pump inhibitors in children: a comprehensive review. Paediatr Drugs. 2003;5:25–40.
Acknowledgements
This work is supported by public grants of Saga Prefecture. We would like to thank Mr. Daisuke Yoshihara, Mr. Shuichi Furukawa, and Ms. Aiko Ikegami of the department of Health and Welfare in Saga Prefecture. The authors thank Professor Takuji Gotoda, Dr. Tsugio Furukawa, Dr. Chika Kusano, Dr. Mototsugu Kato, Dr. Katsuhiro Mabe, and Mr. Hiroo Sashiyama for their excellent suggestions, as well as Professor Masahiro Asaka for their role as safety evaluation committee chairman for this project. We also want to acknowledge Dr. Yamaguchi D, Dr. Takeshita E, Dr. Hanada K, Dr. Kawakubo Y, Dr. Imamura I, Dr. Taniguchi K, Dr. Yamamoto K, Dr. Kajiwara T, Dr. Kuwahara A, Dr. Akiyama M, Dr. Yamaguchi S, Dr. Mizoe A, Dr. Muro E, Dr. Nishimura S, Dr. Noda T, Dr. Ogata S, Dr. Matsunaga K, Dr. Watanabe S, Dr. Inutsuka S, Dr. Fukuyama K, Dr. Ooi J, Dr. Fujioka Y, Takayanagi T and Dr. Fukuda K, the head of the department in charge of the cooperating medical institution. Finally, we thank Ms. Kozue Kakiuchi, Ms. Tomomi Ito, and Ms. Hiromi Beppu, the project business center’s staff, for their support.
Funding
This project was supported by public grants from the government of Saga Prefecture.
Author information
Authors and Affiliations
Contributions
Guarantor of the article: TK. Conceptualization, data analysis, and manuscript preparation: TK, HE, and KF. Data collection: TK. Critical review of the manuscript: MM, KA, and KF. All authors have read and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kakiuchi, T., Matsuo, M., Endo, H. et al. A Helicobacter pylori screening and treatment program to eliminate gastric cancer among junior high school students in Saga Prefecture: a preliminary report. J Gastroenterol 54, 699–707 (2019). https://doi.org/10.1007/s00535-019-01559-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-019-01559-9